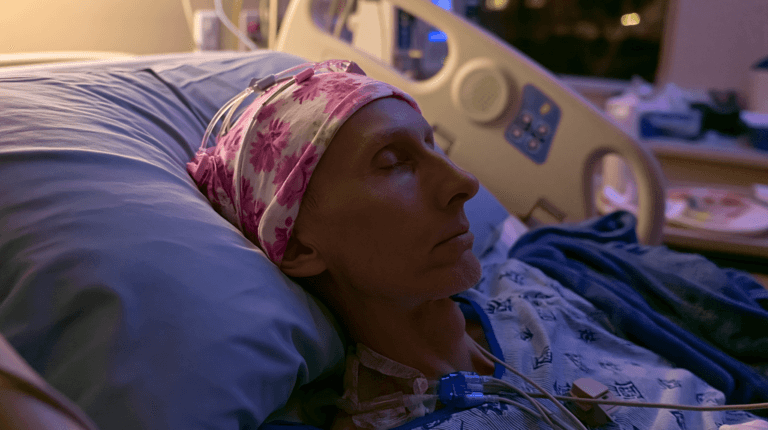- Why Patients Confuse Glioma and Glioblastoma
- Quick Answer: Is Glioblastoma a Type of Glioma?
- What Is a Glioma?
- What Is Glioblastoma?
- Glioma vs Glioblastoma: Simple Comparison Table
- Difference in Grade: Low-Grade Glioma vs High-Grade Glioma vs Glioblastoma
- Difference in Molecular Markers
- Difference in Growth Pattern
- Difference in Symptoms
- Difference in Diagnosis
- Difference in Treatment
- Treatment Side Effects and Quality of Life
- Difference in Survival and Prognosis
- Recurrence: Why Glioblastoma Comes Back More Often
- Ayurvedic Perspective: How This Difference Matters in Holistic Care
- Safety Section: What Patients Should Not Do
- Patient Decision Guide: Questions to Ask Your Doctor
- FAQs
- Reference
Why Patients Confuse Glioma and Glioblastoma
Glioma vs glioblastoma is one of the most common areas of confusion for patients and families after a brain tumor diagnosis. Many people hear both words during MRI review, biopsy discussion, or neuro-oncology consultation, and they assume they mean the same thing. This confusion is understandable because glioblastoma does belong to the glioma family. However, the two terms are not equal. Glioma is a broad medical category that includes several tumors arising from glial-supporting cells of the brain and spinal cord, while glioblastoma is one specific, highly aggressive form of glioma classified as grade 4 [2].
A simple way to understand the difference is this: all glioblastomas are gliomas, but not all gliomas are glioblastomas. Some gliomas grow slowly, remain stable for a period of time, and may be managed with surgery, monitoring, radiation, chemotherapy, targeted therapy, or a combination depending on the case. Glioblastoma, on the other hand, is considered the most aggressive adult-type diffuse glioma. It usually grows rapidly, infiltrates nearby brain tissue, and has a higher risk of recurrence even after standard treatment [6].
This distinction is important because the word “glioma” alone does not tell the full story. A patient with a low-grade glioma may have a very different treatment plan and survival outlook compared with a patient diagnosed with glioblastoma. Modern neuro-oncology now looks beyond the tumor name and studies the tumor grade, MRI pattern, microscopic appearance, and molecular markers such as IDH status, 1p/19q codeletion, MGMT promoter methylation, and other genetic features. These details help doctors understand how the tumor may behave and which treatment path may be more suitable [2].
For patients, the confusion also becomes emotional. When someone searches online for glioma and finds glioblastoma survival statistics, fear can rise immediately. But every glioma is not glioblastoma, and every patient’s case must be understood individually. Age, tumor location, neurological condition, extent of surgery, molecular profile, recurrence risk, and overall strength all influence the treatment journey and prognosis [7].
This article explains the difference between glioma and glioblastoma in a clear, patient-friendly way, covering grade, growth pattern, symptoms, diagnosis, treatment, recurrence risk, survival, and integrative Ayurvedic support. For readers who want a deeper explanation of glioma symptoms, diagnosis, modern treatment limitations, Rasayana support, diet, safety, and Ayurvedic treatment principles, the complete guide on gliomas is available here: https://panaceayur.com/disease-cure-oncology-brain-gliomas-ayurvedic-cure/ [1].
Quick Answer: Is Glioblastoma a Type of Glioma?
Quick Answer
Yes. Glioblastoma is a type of glioma, but it represents the most aggressive end of the glioma spectrum. Glioma is a broad medical term used for tumors that arise from glial cells in the brain or spinal cord. Glioblastoma, also called GBM, is a specific high-grade glioma classified as CNS WHO grade 4. This means it has rapid growth potential, infiltrates surrounding brain tissue, and usually requires urgent neuro-oncology care. [2,3,6,7]
In practical terms, all glioblastomas are gliomas, but not all gliomas are glioblastomas. Some gliomas are low-grade and may grow slowly over years, while others are high-grade and behave aggressively. Glioblastoma belongs to the most serious category because it is grade 4 by definition. [2,6,7]
Clinical Meaning of the Difference
For a patient or family, the word “glioma” alone is not enough to understand the seriousness of the disease. The more important questions are: What type of glioma is it? What grade is it? Is it IDH-mutant or IDH-wildtype? Does it have 1p/19q codeletion? What is the MGMT promoter methylation status?
These details are not technical extras; they directly influence prognosis, treatment planning, recurrence risk, and expected response to therapy. A grade 2 glioma, grade 3 glioma, and glioblastoma may all fall under the larger glioma family, but they often differ greatly in growth speed, treatment urgency, and survival outlook. [2,3,6,7]
Modern WHO Classification
Under the modern WHO classification of central nervous system tumors, glioblastoma is diagnosed using both microscopic appearance and molecular features. In adults, the term glioblastoma is now mainly used for glioblastoma, IDH-wildtype, CNS WHO grade 4. This distinction is important because some tumors that previously may have been called “IDH-mutant glioblastoma” are now more accurately classified as astrocytoma, IDH-mutant, CNS WHO grade 4. [2,3]
This change in terminology helps doctors separate biologically different tumors. Two tumors may both appear aggressive, but if one is IDH-mutant and another is IDH-wildtype, their behavior, prognosis, and treatment expectations may not be the same. Therefore, molecular testing is now essential in glioma diagnosis, not optional. [2,3]
Patient-Friendly Explanation
Think of “glioma” as the larger family name. Within that family, there are different members, such as astrocytoma, oligodendroglioma, and glioblastoma. Glioblastoma is the most aggressive member of this family. It grows faster, invades nearby brain tissue more extensively, and has a higher risk of recurrence than many lower-grade gliomas. [6,7]
This is why patients should avoid comparing survival numbers or treatment stories unless the exact tumor type, grade, and molecular profile are known. A patient with a low-grade IDH-mutant glioma may have a very different clinical course from a patient with glioblastoma, IDH-wildtype, CNS WHO grade 4. [2,3,7]
Key Takeaway for Readers
Glioblastoma is not separate from glioma; it is a severe form of glioma. The main difference is that glioma is a broad category, while glioblastoma is a specific grade 4 tumor with aggressive growth and a more challenging prognosis. Before making treatment decisions or exploring supportive Ayurvedic care, the diagnosis should be confirmed through MRI, histopathology, and molecular testing. [2,3,6,7]
Once the exact diagnosis is clear, patients can better understand the role of surgery, radiation, chemotherapy, targeted therapy where appropriate, rehabilitation, nutrition, and supervised integrative support. This clarity protects patients from confusion and helps families make informed decisions at the right time.
What Is a Glioma?
Medical Definition
A glioma is a tumor that develops from glial cells, the supportive cells of the brain and spinal cord. These cells normally help protect, nourish, and maintain nerve cells. When abnormal growth begins in these cells, different types of gliomas can form, depending on the cell lineage, tumor grade, and molecular profile. [2,4,6]
Glioma is not a single disease. It is a broad diagnostic category that includes tumors such as astrocytoma, oligodendroglioma, ependymoma, and glioblastoma. This is why two patients may both be told they have a “glioma,” yet their disease behavior, treatment plan, and prognosis may be very different. [2,4,6]
Why the Word “Glioma” Can Be Misleading
From a patient’s point of view, the word glioma often creates immediate fear because it is associated with brain tumors. Clinically, however, the term needs further clarification. Some gliomas are low-grade and may grow slowly. Others are high-grade, fast-growing, and more difficult to control. Glioblastoma is the most aggressive form within this larger group. [2,6]
Therefore, the first step after hearing the diagnosis is not panic, but precision. The treating team must identify the exact tumor type, CNS WHO grade, location, and molecular markers. These details help decide whether the patient may need surgery alone, surgery followed by radiation or chemotherapy, active surveillance, targeted therapy, rehabilitation, or additional supportive care. [2,4]
Types of Gliomas
Gliomas are commonly classified according to the type of glial cell involved and their molecular behavior. Astrocytomas arise from astrocytic cells and may range from lower-grade tumors to grade 4 astrocytoma. Oligodendrogliomas are usually associated with IDH mutation and 1p/19q codeletion, and they often behave differently from astrocytic tumors. Ependymomas arise from cells lining the ventricles or spinal canal. Glioblastoma is a grade 4 diffuse glioma and represents the most aggressive end of this disease spectrum. [2,4,6]
This classification is important because treatment is no longer based only on the tumor’s name. Modern neuro-oncology combines MRI findings, histopathology, and molecular testing to understand how the tumor may behave and which treatment strategy is most appropriate. [2,4]
Low-Grade and High-Grade Gliomas
Gliomas are also described as low-grade or high-grade. Low-grade gliomas usually grow more slowly and may present with symptoms such as seizures, mild headaches, subtle cognitive changes, or changes in speech or behavior. However, “low-grade” does not mean harmless. These tumors can still infiltrate brain tissue, recur, or transform into higher-grade disease over time. [4,6]
High-grade gliomas grow more aggressively and often produce faster neurological symptoms. They may cause worsening headache, weakness, speech difficulty, confusion, vision problems, seizures, or symptoms related to increased pressure inside the skull. Glioblastoma falls into this high-grade category and is always considered grade 4. [4,6]
Why Molecular Testing Matters
In earlier decades, gliomas were classified mainly by microscopic appearance. Today, molecular testing has become central to diagnosis. Markers such as IDH mutation, 1p/19q codeletion, MGMT promoter methylation, TERT promoter mutation, ATRX status, TP53 mutation, EGFR amplification, and chromosome 7 gain/10 loss can help doctors understand tumor biology more accurately. [2,4]
This is especially important because molecular findings can influence prognosis and treatment decisions. For example, an IDH-mutant glioma may behave differently from an IDH-wildtype glioma. Similarly, an oligodendroglioma with 1p/19q codeletion may have a different clinical course from an astrocytic tumor. Without this information, the diagnosis remains incomplete. [2,4]
Clinical Relevance for Patients
For patients and families, understanding glioma as a spectrum is empowering. It prevents unnecessary comparison with other patients, especially those who may have completely different tumor biology. A person with a grade 2 IDH-mutant glioma should not assume the same outlook as someone with glioblastoma. At the same time, a low-grade diagnosis should not lead to neglect, because regular follow-up and timely treatment remain essential. [2,4,6]
This is also where integrative and Ayurvedic support must be placed correctly. Before considering any supportive plan, the tumor must be clearly diagnosed through MRI, histopathology, and molecular testing. The Panacea Ayur glioma pillar article discusses gliomas from both modern and Ayurvedic perspectives, including symptoms, diagnostic clarity, Rasayana support, safety warnings, and quality-of-life care. [1]
Practical Clinical Message
A glioma diagnosis should always be followed by four essential questions: What type of glioma is it? What is the CNS WHO grade? What are the molecular markers? What treatment is needed now?
Once these answers are clear, the patient and family can make better decisions about surgery, radiation, chemotherapy, surveillance, rehabilitation, nutrition, and supervised Ayurvedic supportive care. The goal is not only to name the disease, but to understand its behavior and respond with the right strategy at the right time. [1,2,4,6]
What Is Glioblastoma?
Medical Definition
Glioblastoma is the most aggressive form of diffuse glioma in adults. It is classified as CNS WHO grade 4, which means it has features of highly malignant behavior, rapid growth, invasion into surrounding brain tissue, and a strong tendency to recur even after treatment. [2,3,4,7]
In modern classification, adult glioblastoma is generally referred to as glioblastoma, IDH-wildtype, CNS WHO grade 4. This terminology is important because brain tumor diagnosis has moved beyond older descriptions based only on microscopic appearance. Today, molecular markers are essential for defining the tumor correctly and predicting its likely behavior. [2,3]
Why Glioblastoma Is Considered Grade 4
Glioblastoma is grade 4 by definition. It is not a low-grade tumor, and the phrase “low-grade glioblastoma” is medically incorrect. Lower-grade gliomas may grow more slowly and may remain stable for a period of time, but glioblastoma behaves differently from the beginning. It tends to multiply rapidly, infiltrate nearby brain tissue, stimulate abnormal blood vessel formation, and create areas of tissue breakdown or necrosis. [2,3,4]
This grade 4 status explains why glioblastoma usually requires urgent evaluation by a neurosurgeon, neuro-oncologist, radiation oncologist, and supportive care team. The aim is not only to remove or reduce the visible tumor, but also to control microscopic disease that may extend beyond what is seen on imaging. [3,4]
How Glioblastoma Behaves in the Brain
Glioblastoma does not usually grow like a neatly separated mass that can be removed completely with clear borders. Instead, it spreads into the surrounding brain tissue through microscopic infiltration. This is one of the main reasons treatment is challenging. Even when surgery removes the visible tumor, small numbers of tumor cells may remain in nearby tissue and later contribute to recurrence. [3,4]
The clinical behavior of glioblastoma can also be fast. Patients may develop headaches, seizures, weakness, speech difficulty, memory changes, personality changes, confusion, vision problems, or worsening neurological function. The symptoms depend on the tumor location, the amount of swelling, and how quickly pressure effects develop inside the brain. [4,7]
Modern Diagnostic Identity
In the current WHO framework, glioblastoma in adults is primarily an IDH-wildtype diffuse astrocytic glioma with grade 4 features. Molecular findings such as TERT promoter mutation, EGFR amplification, and combined chromosome 7 gain/chromosome 10 loss can support the diagnosis in the appropriate setting. [2,3]
This matters because a tumor with IDH mutation and grade 4 features may no longer be called glioblastoma in the same way under modern classification. It may be diagnosed as astrocytoma, IDH-mutant, CNS WHO grade 4. Although both are serious, their biology and prognosis may differ. This is why a complete pathology report should include both histological and molecular details. [2,3]
Treatment Direction in Glioblastoma
The usual treatment approach for newly diagnosed glioblastoma includes maximal safe surgical resection when feasible, followed by radiation therapy with temozolomide chemotherapy, and then further temozolomide cycles. This combined approach is used because glioblastoma is not only a local mass but an infiltrative disease. [4]
In selected patients, other options may be considered, including Tumor Treating Fields, clinical trials, treatment for recurrence, rehabilitation, seizure control, steroid management, and palliative or supportive care. Treatment is individualized according to age, performance status, tumor location, extent of surgery, MGMT promoter methylation status, neurological condition, and patient preference. [3,4,7]
Prognosis: Why Honest Communication Matters
Glioblastoma has a more difficult prognosis than many other gliomas. Mayo Clinic reports that median life expectancy for many patients is commonly around 12 to 18 months after diagnosis with standard treatment, while a smaller group survives longer. Five-year survival is limited, but outcomes vary from patient to patient. [7]
These numbers should be explained with care. Survival statistics describe groups, not individuals. A patient’s outlook may be influenced by age, general health, neurological function, tumor location, extent of resection, molecular markers such as MGMT promoter methylation, ability to complete treatment, and access to multidisciplinary care. [3,4,7]
Why This Distinction Helps Patients Make Better Decisions
For patients and families, understanding glioblastoma clearly prevents two common mistakes. The first is underestimating the disease by thinking it is just another “glioma.” The second is losing hope because survival statistics are misunderstood as fixed predictions. A precise diagnosis helps the family plan treatment, supportive care, rehabilitation, nutrition, emotional support, and follow-up with better clarity. [3,4,7]
This is also where supervised integrative and Ayurvedic support should be positioned responsibly. Glioblastoma requires modern neuro-oncology care. Ayurvedic support, where appropriate, should be considered as a complementary strategy for strength, appetite, sleep, fatigue, emotional balance, treatment tolerance, and quality of life—not as a replacement for surgery, radiation, chemotherapy, or urgent medical care.
Clinical Bottom Line
Glioblastoma is a grade 4 glioma with aggressive growth, microscopic infiltration, and a high recurrence risk. It is now understood through both histology and molecular testing, especially IDH status. Once the diagnosis is confirmed, the priority is timely multidisciplinary treatment, followed by careful monitoring and supervised supportive care that protects the patient’s function, dignity, and quality of life. [2,3,4,7]
Glioma vs Glioblastoma: Simple Comparison Table
At-a-Glance Comparison
| Feature | Glioma | Glioblastoma |
|---|---|---|
| Basic meaning | A broad group of tumors arising from glial cells in the brain or spinal cord | A specific, highly aggressive type of glioma |
| Disease category | Umbrella term that includes astrocytoma, oligodendroglioma, ependymoma, and glioblastoma | High-grade diffuse glioma, usually described as glioblastoma, IDH-wildtype, CNS WHO grade 4 in adults |
| WHO grade | Can be grade 1, grade 2, grade 3, or grade 4 depending on the tumor type | Always CNS WHO grade 4 |
| Growth pattern | May be slow-growing, moderately aggressive, or fast-growing | Rapid-growing, infiltrative, and difficult to completely remove |
| Common presentation | Seizures, headache, speech difficulty, weakness, memory change, personality change, or symptoms depending on tumor location | Similar symptoms, but often faster progression with more aggressive neurological decline |
| Molecular markers | May include IDH mutation, 1p/19q codeletion, ATRX alteration, TP53 mutation, MGMT status, TERT promoter mutation, and other markers | Commonly IDH-wildtype in adults; MGMT promoter methylation, EGFR amplification, TERT promoter mutation, and chromosome 7/10 changes may be relevant |
| Treatment approach | Depends on grade, location, symptoms, surgical safety, and molecular profile | Usually maximal safe surgery when possible, followed by radiation therapy with temozolomide; other options may be considered in selected patients |
| Recurrence risk | Variable; lower-grade gliomas may recur or transform over time | High recurrence risk, even after aggressive treatment |
| Survival outlook | Highly variable; lower-grade and IDH-mutant gliomas often have a better outlook than aggressive high-grade tumors | Generally poorer prognosis; median survival is commonly discussed in months rather than years, though individual outcomes vary |
| Supportive care need | Long-term monitoring, seizure control, rehabilitation, nutrition, mental health support, and supervised integrative care when appropriate | Intensive multidisciplinary support for fatigue, cognition, appetite, sleep, neurological function, emotional distress, and quality of life |
Glioma vs Glioblastoma: Simple Comparison Table
At-a-Glance Comparison
| Feature | Glioma | Glioblastoma |
|---|---|---|
| Basic meaning | A broad group of tumors arising from glial cells in the brain or spinal cord | A specific, highly aggressive type of glioma |
| Disease category | Umbrella term that includes astrocytoma, oligodendroglioma, ependymoma, and glioblastoma | High-grade diffuse glioma, usually described as glioblastoma, IDH-wildtype, CNS WHO grade 4 in adults |
| WHO grade | Can be grade 1, grade 2, grade 3, or grade 4 depending on the tumor type | Always CNS WHO grade 4 |
| Growth pattern | May be slow-growing, moderately aggressive, or fast-growing | Rapid-growing, infiltrative, and difficult to completely remove |
| Common presentation | Seizures, headache, speech difficulty, weakness, memory change, personality change, or symptoms depending on tumor location | Similar symptoms, but often faster progression with more aggressive neurological decline |
| Molecular markers | May include IDH mutation, 1p/19q codeletion, ATRX alteration, TP53 mutation, MGMT status, TERT promoter mutation, and other markers | Commonly IDH-wildtype in adults; MGMT promoter methylation, EGFR amplification, TERT promoter mutation, and chromosome 7/10 changes may be relevant |
| Treatment approach | Depends on grade, location, symptoms, surgical safety, and molecular profile | Usually maximal safe surgery when possible, followed by radiation therapy with temozolomide; other options may be considered in selected patients |
| Recurrence risk | Variable; lower-grade gliomas may recur or transform over time | High recurrence risk, even after aggressive treatment |
| Survival outlook | Highly variable; lower-grade and IDH-mutant gliomas often have a better outlook than aggressive high-grade tumors | Generally poorer prognosis; median survival is commonly discussed in months rather than years, though individual outcomes vary |
| Supportive care need | Long-term monitoring, seizure control, rehabilitation, nutrition, mental health support, and supervised integrative care when appropriate | Intensive multidisciplinary support for fatigue, cognition, appetite, sleep, neurological function, emotional distress, and quality of life |
How to Interpret This Comparison
The most important difference is that glioma is a category, while glioblastoma is a specific diagnosis within that category. Calling a tumor “glioma” is similar to naming the broad disease family. It does not yet tell the patient how fast the tumor may grow, how urgent treatment is, or what survival expectations should be. For that, doctors need the exact tumor type, CNS WHO grade, and molecular profile. [2,4,6]
Glioblastoma, on the other hand, immediately indicates a grade 4 tumor. It is aggressive by definition and usually requires prompt multidisciplinary treatment. In adult patients, modern classification places strong emphasis on IDH status, because glioblastoma is now generally used for IDH-wildtype grade 4 diffuse astrocytic glioma. [2,4]
Why This Table Matters Clinically
This comparison protects patients from two common misunderstandings. The first is assuming that every glioma has the same poor prognosis as glioblastoma. That is not correct. Some gliomas, especially lower-grade and IDH-mutant tumors, may follow a slower course and may be managed over a longer period with surgery, monitoring, radiation, chemotherapy, targeted therapy, or a combination of these approaches. [2,4,6]
The second misunderstanding is assuming that a “glioma” diagnosis is automatically mild because the word glioblastoma is not used. That is also unsafe. Some non-glioblastoma gliomas can still be high-grade, infiltrative, recurrent, or difficult to treat depending on their location and biology. The name alone should never replace complete grading and molecular testing. [2,4]
Treatment Planning Perspective
For low-grade gliomas, the treatment conversation may include maximal safe surgery, observation in selected cases, radiation, chemotherapy, or newer targeted therapy options depending on IDH status and clinical risk. For glioblastoma, treatment is more urgent and usually begins with maximal safe resection when feasible, followed by radiation therapy and temozolomide chemotherapy. [4]
This difference is important for families because treatment intensity, side effects, follow-up frequency, and supportive care needs are not the same. A patient with a slow-growing glioma may need long-term surveillance and seizure control. A patient with glioblastoma may need rapid coordination between neurosurgery, radiation oncology, medical oncology, rehabilitation, nutrition, and palliative/supportive care services. [4,7]
Prognosis Perspective
Survival in glioma cannot be explained with one number. It depends on age, performance status, tumor location, extent of surgical removal, grade, IDH status, 1p/19q codeletion, MGMT promoter methylation, treatment response, and recurrence pattern. Population statistics are useful for general understanding, but they should not be applied mechanically to an individual patient. [2,7,14]
Glioblastoma has a more guarded prognosis than most lower-grade gliomas. However, even in glioblastoma, prognosis varies. Factors such as younger age, good functional status, greater extent of resection, MGMT promoter methylation, and ability to complete standard therapy may influence outcome. [4,7]
Integrative Care Perspective
From an integrative and Ayurvedic care viewpoint, this distinction is essential. A patient with a low-grade glioma may need long-term support for neurological stability, digestion, sleep, mental strength, seizure control coordination, and treatment recovery. A patient with glioblastoma often needs more intensive supportive care focused on strength, fatigue, appetite, cognition, emotional resilience, treatment tolerance, and quality of life.
The clinical priority remains clear: the exact diagnosis must come first. Ayurvedic or integrative support should be planned only after understanding the tumor type, grade, molecular profile, ongoing medicines, surgery status, radiation schedule, chemotherapy plan, steroid use, and anti-seizure medication. This approach keeps supportive care safe, individualized, and aligned with neuro-oncology treatment. [2,4,7]
Practical Message for Patients
If the diagnosis says “glioma,” the next step is not to compare it with another patient’s story. The next step is to ask for precision: What type of glioma is it? What grade is it? Is it IDH-mutant or IDH-wildtype? Is there 1p/19q codeletion? What is the MGMT promoter methylation status? What is the safest and most effective treatment plan now?
A clear diagnosis creates a clear pathway. It helps the patient understand whether the condition is slow-growing or aggressive, whether treatment is urgent or staged, how recurrence risk should be monitored, and where supervised supportive care may add value. [2,4,6,7]
Difference in Grade: Low-Grade Glioma vs High-Grade Glioma vs Glioblastoma
Why Tumor Grade Matters
Tumor grade is one of the most important pieces of information in a glioma report. It tells us how aggressive the tumor is expected to be, how quickly treatment may be needed, and how closely the patient should be monitored after treatment. In simple clinical language, grade reflects the biological behavior of the tumor, not merely its size.
Table : Low-Grade Glioma vs High-Grade Glioma vs Glioblastoma
| Category | Usual grade | Growth pattern | Clinical seriousness | Common treatment direction |
|---|---|---|---|---|
| Low-grade glioma | Usually grade 1 or grade 2 | Slower growth | Serious but often less aggressive than high-grade glioma | Surgery, observation, radiation, chemotherapy, or targeted therapy depending on risk |
| High-grade glioma | Usually grade 3 or grade 4 | Faster growth | More aggressive, higher recurrence risk | Surgery followed by radiation and/or chemotherapy in many cases |
| Glioblastoma | Grade 4 | Rapid, infiltrative growth | Most aggressive common adult glioma | Maximal safe surgery, radiation, temozolomide, and supportive care when appropriate |
A small tumor can still be serious if it has aggressive molecular features. Similarly, a larger low-grade tumor may grow slowly but still require careful treatment if it is located near speech, memory, movement, vision, or seizure-sensitive areas of the brain. This is why grading must always be interpreted along with MRI findings, histopathology, molecular markers, neurological symptoms, and the patient’s general condition. [2,3,4]
Understanding Low-Grade Gliomas
Low-grade gliomas usually refer to grade 1 or grade 2 tumors, though in adult diffuse gliomas, grade 2 is especially important. These tumors often grow more slowly than high-grade gliomas. Many patients first present with seizures, mild headache, subtle speech difficulty, memory changes, personality changes, or symptoms related to the tumor’s location. [4,6]
However, the term “low-grade” should never be misunderstood as “harmless.” Low-grade diffuse gliomas can infiltrate normal brain tissue, recur after treatment, and in some cases transform into higher-grade tumors over time. Their clinical journey may be long, but it still requires disciplined monitoring and timely intervention. [2,3,4]
For selected grade 2 IDH-mutant gliomas, treatment decisions may include maximal safe surgery, observation, radiation, chemotherapy, or targeted therapy depending on age, residual tumor, symptoms, molecular profile, and risk of progression. Recent guideline updates also include vorasidenib as an option in selected adults with IDH-mutant grade 2 astrocytoma or oligodendroglioma when radiation and chemotherapy can be deferred. [13]
Understanding High-Grade Gliomas
High-grade gliomas generally include grade 3 and grade 4 tumors. These tumors have more aggressive biological features and usually need more intensive treatment. They may grow faster, cause more rapid neurological decline, and have a greater risk of recurrence compared with many lower-grade gliomas. [2,3,4]
A grade 3 glioma is serious and usually requires active treatment. Depending on the exact diagnosis, treatment may include surgery, radiation therapy, chemotherapy, and long-term MRI surveillance. These tumors are not the same as glioblastoma, but they are also not mild tumors. They sit in an intermediate-to-aggressive category where treatment planning must be precise and proactive. [3,4]
Grade 4 tumors represent the most aggressive category. In this group, the tumor may show rapid cell division, necrosis, microvascular proliferation, or molecular features that indicate highly malignant behavior. Glioblastoma belongs to this grade 4 category. [2,3,4]
Where Glioblastoma Fits
Glioblastoma is always CNS WHO grade 4. It is not a lower-grade glioma and should not be described as “low-grade glioblastoma.” In adults, modern classification generally uses the term glioblastoma for IDH-wildtype diffuse astrocytic glioma with grade 4 histological or molecular features. [2,3]
This distinction is clinically meaningful. A patient with astrocytoma, IDH-mutant, CNS WHO grade 4 may have a different biological profile from a patient with glioblastoma, IDH-wildtype, CNS WHO grade 4. Both are serious, but they are not identical diseases. This is why the pathology report should clearly mention the tumor type, WHO grade, IDH status, and other relevant molecular findings. [2,3]
Glioblastoma grows quickly, infiltrates surrounding brain tissue, and has a high recurrence risk even after surgery, radiation, and chemotherapy. For this reason, it usually requires rapid multidisciplinary care involving neurosurgery, neuro-oncology, radiation oncology, rehabilitation, seizure management, nutrition, and supportive care. [3,4,7]
Molecular Features Can Change the Grade
In modern neuro-oncology, grade is not decided by microscope appearance alone. Molecular markers now play a central role. For example, IDH status helps separate biologically different gliomas. The presence of 1p/19q codeletion supports the diagnosis of oligodendroglioma when combined with IDH mutation. CDKN2A/B homozygous deletion can influence grading in IDH-mutant astrocytoma. In IDH-wildtype diffuse astrocytic tumors, markers such as TERT promoter mutation, EGFR amplification, and chromosome 7 gain/chromosome 10 loss may support aggressive classification in the right context. [2,3]
This is why a diagnosis such as “glioma” is incomplete unless molecular testing is included. The grade, molecular pattern, and tumor location together create the real clinical picture. They help doctors estimate prognosis, select treatment, plan follow-up, and counsel the patient more responsibly. [2,3,4]
How Grade Influences Treatment
In low-grade glioma, treatment may be staged. Some patients undergo maximal safe surgery followed by observation. Others may need radiation, chemotherapy, or targeted therapy depending on residual tumor, symptoms, age, molecular profile, and risk factors. The strategy is often designed to control the disease while preserving neurological function and quality of life. [3,4,13]
In high-grade glioma, treatment is usually more urgent and more intensive. Surgery is often followed by radiation and chemotherapy, with the exact regimen depending on tumor type and molecular findings. In glioblastoma, standard treatment commonly includes maximal safe resection when feasible, followed by radiation therapy with temozolomide, and then additional temozolomide cycles. [4,7]
This is the practical value of grading. It does not merely label the disease; it guides the entire clinical roadmap. It influences how fast treatment should begin, how aggressive the treatment should be, how often MRI follow-up is needed, and what type of supportive care the patient may require.
Why Patients Should Ask About Grade Clearly
Every patient with glioma should ask the treating team: What is the exact WHO grade? Is the tumor low-grade or high-grade? Is it IDH-mutant or IDH-wildtype? Is there 1p/19q codeletion? Is MGMT promoter methylation tested? Are there any molecular features that increase the grade or change the treatment plan?
These questions are not only scientific; they are deeply practical. They help the family understand whether the disease may follow a slower course or whether urgent aggressive treatment is needed. They also prevent confusion when patients search online and find survival statistics that may not apply to their exact tumor type. [2,3,4,6]
Clinical Perspective for Integrative Care
From an integrative and Ayurvedic support perspective, grade changes everything. A patient with a slow-growing grade 2 glioma may require long-term support for neurological stability, seizure coordination, sleep, digestion, mental strength, recovery after surgery, and careful surveillance. A patient with glioblastoma may require more intensive support for fatigue, appetite, treatment tolerance, cognition, mood, steroid-related side effects, and quality of life.
The responsible approach is to begin with diagnostic clarity. Ayurvedic support should be planned only after knowing the tumor grade, molecular profile, treatment schedule, current medicines, seizure history, steroid use, liver and kidney function, and overall strength of the patient. In this way, supportive care becomes personalized and clinically aligned rather than generic.
Practical Clinical Message
The difference between low-grade glioma, high-grade glioma, and glioblastoma is not just a technical classification. It is the foundation for treatment decisions, prognosis, follow-up planning, and supportive care. Glioma can belong to different grades, but glioblastoma is always grade 4. Once the grade and molecular profile are clear, the patient can move from fear and confusion toward a structured treatment plan with greater confidence. [2,3,4,6,13]
Difference in Molecular Markers
Why Molecular Markers Matter
In modern neuro-oncology, a glioma diagnosis is no longer complete if it only says “astrocytoma,” “oligodendroglioma,” or “glioblastoma.” The real clinical picture comes from combining MRI findings, microscopic examination, WHO grade, and molecular markers. These markers help doctors understand how the tumor is likely to behave, how aggressive it may become, and which treatment options may be more suitable. [2,3,4]
Table 3: Glioma Grades and What They Usually Mean
| CNS WHO grade | General meaning | Growth behavior | Important clinical note |
|---|---|---|---|
| Grade 1 | Less aggressive, often more circumscribed | Usually slow-growing | Some may be managed effectively with surgery, depending on type and location |
| Grade 2 | Lower-grade but often infiltrative | Slow to moderate growth | Can recur or progress; long-term MRI follow-up is important |
| Grade 3 | Malignant or anaplastic behavior | Faster growth | Usually needs active treatment and close monitoring |
| Grade 4 | Most aggressive category | Rapid growth with high recurrence risk | Glioblastoma is grade 4 by definition |
For patients and families, molecular testing is like the “biological identity card” of the tumor. Two tumors may look similar on MRI, and even appear similar under the microscope, but their molecular profile can lead to very different treatment decisions and survival expectations. This is why every glioma patient should ask for a complete pathology and molecular report, not just the tumor name. [2,3]
IDH Status
IDH status is one of the most important molecular markers in diffuse gliomas. IDH refers to isocitrate dehydrogenase, commonly IDH1 or IDH2. A glioma may be IDH-mutant or IDH-wildtype. This distinction strongly influences classification, prognosis, and treatment planning. [2,3,4]
In general, IDH-mutant gliomas often have a better prognosis than IDH-wildtype gliomas. Many adult low-grade diffuse gliomas are IDH-mutant, while adult glioblastoma is now generally classified as glioblastoma, IDH-wildtype, CNS WHO grade 4. This is why IDH testing is essential when differentiating lower-grade glioma, high-grade astrocytoma, and glioblastoma. [2,3]
A very important point for patients is this: a tumor with grade 4 features and IDH mutation may not be called glioblastoma under modern terminology. It is commonly classified as astrocytoma, IDH-mutant, CNS WHO grade 4. This difference is not just a change in language; it reflects different tumor biology and may influence prognosis. [2,3]
1p/19q Codeletion
The 1p/19q codeletion is another major marker, especially for oligodendroglioma. When a glioma has both an IDH mutation and 1p/19q codeletion, it supports the diagnosis of oligodendroglioma. These tumors often behave differently from astrocytic tumors and may respond better to certain treatments. [2,3,10]
This marker helps avoid vague or outdated labels such as “mixed glioma” or “oligoastrocytoma,” which are less commonly used in modern classification. Today, molecular testing allows doctors to define the tumor more accurately and choose a more rational treatment path. [2,3,10]
For patients, this means the report should not only mention “oligodendroglioma.” It should ideally confirm IDH mutation and 1p/19q codeletion. Without these details, the diagnosis may remain incomplete.
MGMT Promoter Methylation
MGMT promoter methylation is especially important in glioblastoma. MGMT is a DNA repair gene. When the MGMT promoter is methylated, the tumor may be less able to repair the DNA damage caused by temozolomide chemotherapy. Because of this, MGMT promoter methylation is often associated with better response to temozolomide and improved survival in glioblastoma patients. [4,9]
This does not mean that every MGMT-methylated tumor will respond perfectly, or that every unmethylated tumor will fail treatment. It means that MGMT status gives the oncology team useful information when discussing treatment expectations, prognosis, and chemotherapy benefit. [4,9]
From a counselling perspective, MGMT status helps families understand why two glioblastoma patients receiving similar treatment may have different outcomes. The treatment may look the same on paper, but the tumor biology may not be the same.
TERT Promoter Mutation
TERT promoter mutation is another marker often discussed in gliomas. It is commonly seen in certain glioma groups and can help refine molecular classification when interpreted with IDH status and 1p/19q codeletion. [2,10]
In glioblastoma, TERT promoter mutation may be one of the molecular features supporting aggressive IDH-wildtype tumor biology. In oligodendroglioma, TERT promoter mutation may also be seen along with IDH mutation and 1p/19q codeletion. Therefore, TERT should not be interpreted alone. It must be read as part of the full molecular pattern. [2,3,10]
EGFR Amplification and Chromosome 7/10 Changes
EGFR amplification is commonly associated with aggressive IDH-wildtype gliomas, including glioblastoma. EGFR is involved in cell growth signalling, and amplification of this gene can suggest more aggressive tumor behavior in the correct diagnostic setting. [2,3]
Another important molecular pattern is combined whole chromosome 7 gain and whole chromosome 10 loss, often written as +7/−10. Along with EGFR amplification and TERT promoter mutation, this pattern can support the diagnosis of glioblastoma, IDH-wildtype, in adult diffuse astrocytic tumors. [2,3]
For readers, the practical message is simple: glioblastoma is not diagnosed only because a tumor looks aggressive. Modern diagnosis also asks whether the molecular behavior fits glioblastoma biology.
ATRX and TP53
ATRX loss and TP53 mutation are commonly discussed in astrocytic tumors, especially IDH-mutant astrocytomas. These markers help distinguish astrocytoma from oligodendroglioma when interpreted with IDH mutation and 1p/19q status. [2,3]
For example, an IDH-mutant tumor with ATRX loss and TP53 mutation is more likely to belong to the astrocytoma pathway. In contrast, an IDH-mutant tumor with 1p/19q codeletion supports oligodendroglioma. This distinction matters because astrocytoma and oligodendroglioma may differ in prognosis, treatment planning, and long-term monitoring. [2,3]
CDKN2A/B Deletion
CDKN2A/B homozygous deletion is an important marker in IDH-mutant astrocytoma. Its presence may indicate more aggressive behavior and can influence grading. This is one example of how molecular findings can change the seriousness of the diagnosis, even when the tumor’s microscopic appearance does not tell the full story. [2,3]
This is why patients should not rely only on older biopsy language or incomplete reports. If molecular testing is missing, the treating team may need additional testing to define the tumor more accurately.
How Molecular Markers Influence Treatment
Molecular markers guide treatment in several ways. IDH status helps classify the tumor and estimate prognosis. 1p/19q codeletion helps identify oligodendroglioma and may influence chemotherapy planning. MGMT promoter methylation helps estimate likely benefit from temozolomide in glioblastoma. EGFR, TERT, and chromosome 7/10 changes help identify aggressive IDH-wildtype tumor biology. [2,3,4,9,10]
Newer treatment directions also depend on molecular profiling. For selected patients with grade 2 IDH-mutant astrocytoma or oligodendroglioma, updated guidance includes consideration of IDH-targeted therapy such as vorasidenib in appropriate clinical situations where radiation and chemotherapy may be deferred. [13]
This is an important shift. Treatment is becoming more personalized. The goal is no longer only to treat “a brain tumor,” but to treat the right tumor, in the right patient, at the right time, with the least avoidable harm.
What Patients Should Ask About Their Report
A patient with glioma should ask the treating doctor whether the pathology report includes IDH status, 1p/19q codeletion, MGMT promoter methylation, TERT promoter mutation, ATRX, TP53, EGFR amplification, chromosome 7 gain/chromosome 10 loss, and CDKN2A/B status where relevant. Not every marker is equally important in every case, but the treating team should be able to explain which markers matter for that specific tumor. [2,3,4]
This conversation should happen before comparing survival statistics, choosing treatment intensity, or planning long-term supportive care. Without molecular clarity, the patient may receive either too much fear or false reassurance.
Relevance for Integrative and Ayurvedic Support
From an integrative care perspective, molecular markers help define the level of urgency and support required. A patient with a slow-growing IDH-mutant glioma may need a long-term plan focused on neurological stability, digestion, sleep, seizure coordination, mental strength, and regular monitoring. A patient with glioblastoma, IDH-wildtype, may need more intensive supportive care during surgery, radiation, chemotherapy, steroid use, fatigue, cognitive decline, appetite loss, emotional distress, and recurrence monitoring.
Ayurvedic support should therefore not be generic. It should be aligned with the tumor type, grade, molecular profile, current treatment plan, medications, liver and kidney function, seizure history, and overall strength of the patient. This approach respects both modern neuro-oncology and individualized supportive care.
Clinical Message
Molecular markers are not minor laboratory details. They are central to modern glioma diagnosis and treatment planning. IDH status, 1p/19q codeletion, MGMT promoter methylation, TERT promoter mutation, EGFR amplification, chromosome 7/10 changes, ATRX, TP53, and CDKN2A/B can all influence how the disease is understood and managed. For patients, the most important step is to move from a broad label like “glioma” to a precise diagnosis that guides treatment, prognosis, follow-up, and supervised supportive care. [2,3,4,9,10,13]
Difference in Growth Pattern
Why Growth Pattern Is More Important Than Size Alone
When patients look at an MRI report, they often focus on the size of the tumor. Size is important, but in glioma care, growth pattern is equally important. A small tumor with aggressive biology may be more dangerous than a larger tumor that is slow-growing and well-monitored. The clinical question is not only “How big is it?” but also “How fast is it growing, how deeply is it infiltrating, and how is it affecting nearby brain function?” [3,4,6]
Gliomas do not always behave like tumors in other parts of the body. Many gliomas grow into the surrounding brain tissue rather than staying as a clearly separate lump. This infiltrative nature makes treatment complex because normal brain tissue controls speech, memory, movement, vision, personality, and consciousness. Even a small amount of tumor growth in a sensitive area can produce significant symptoms.
Growth Pattern in Lower-Grade Gliomas
Lower-grade gliomas usually grow more slowly than high-grade gliomas. Some may remain relatively stable for a period of time, especially when they are closely monitored and treated at the right stage. Patients may first come to medical attention because of seizures, mild headaches, subtle memory changes, speech difficulty, mood changes, or symptoms related to the tumor’s location. [4,6]
However, slow-growing does not mean harmless. Many lower-grade diffuse gliomas are infiltrative. They may extend beyond the visible MRI abnormality and gradually involve surrounding brain networks. Over time, some lower-grade gliomas can recur or transform into higher-grade tumors. This is why regular MRI follow-up, molecular testing, and timely treatment decisions are essential. [3,4]
From a clinical point of view, lower-grade glioma is often a long-term disease journey. The treatment strategy may focus on balancing tumor control with preservation of neurological function. Surgery, observation, radiation, chemotherapy, targeted therapy, seizure control, rehabilitation, and supportive care may all have a role depending on the individual case. [3,4]
Growth Pattern in High-Grade Gliomas
High-grade gliomas grow more aggressively. They tend to multiply faster, cause more swelling, and produce symptoms over a shorter period. A patient may notice worsening headache, repeated vomiting, weakness, speech difficulty, confusion, personality change, seizures, visual disturbance, or decline in daily functioning. [4,6]
These tumors often create a more urgent clinical situation because they can affect brain pressure and neurological function quickly. Treatment planning usually needs faster coordination between neurosurgery, neuro-oncology, radiation oncology, pathology, radiology, and supportive care teams. [3,4]
The challenge with high-grade gliomas is not only speed. It is also invasion. Even when the visible tumor is removed, microscopic tumor cells may remain in surrounding brain tissue. This is one of the major reasons high-grade gliomas require combined treatment rather than surgery alone in most cases. [3,4]
How Glioblastoma Grows
Glioblastoma has the most aggressive growth pattern among common adult gliomas. It is fast-growing, deeply infiltrative, and biologically heterogeneous. This means different areas of the same tumor may behave differently, respond differently to treatment, and contain cells with varying levels of resistance. [3,4,7]
Glioblastoma can stimulate new blood vessel formation to support its rapid growth. It may also develop areas of necrosis, where parts of the tumor outgrow their blood supply and break down. These features are part of why glioblastoma is classified as CNS WHO grade 4. [3,4]
On MRI, glioblastoma often appears as an enhancing mass with surrounding edema, but the visible abnormality does not always show the full extent of microscopic spread. Tumor cells may infiltrate beyond the contrast-enhancing region. This explains why complete cure through surgery alone is usually not possible, even when the operation successfully removes the main visible tumor. [3,4,7]
Why Glioblastoma Often Recurs
Recurrence is one of the defining challenges of glioblastoma. Surgery can reduce tumor burden, radiation can target the tumor region, and temozolomide can help control disease in selected patients, but glioblastoma cells may remain beyond the visible tumor margin. These remaining cells can later grow again. [3,4,7]
This is why glioblastoma treatment is usually multimodal. The goal is to remove as much tumor as safely possible, then treat the surrounding region where microscopic cells may remain. Even after aggressive treatment, regular MRI surveillance is necessary because recurrence is common. [3,4]
For families, this point must be explained with honesty and sensitivity. Recurrence does not always mean that treatment was wrong or surgery failed. It often reflects the natural biology of glioblastoma: fast growth, diffuse infiltration, cellular diversity, and treatment resistance.
Growth Speed and Symptoms
The speed of growth often influences how symptoms appear. A slow-growing glioma may give the brain time to adapt, so symptoms may be subtle in the beginning. Seizures may be the first sign. Some patients may continue normal activities for a long period before the diagnosis is made. [4,6]
Glioblastoma may produce symptoms more rapidly. Because it grows quickly and causes swelling, patients may develop neurological decline over weeks or months. Headache, confusion, weakness, speech difficulty, personality changes, memory problems, seizures, or reduced alertness may appear or worsen quickly. [4,7]
This difference is important for patient education. Any sudden neurological symptom, first-time seizure, progressive weakness, severe persistent headache, repeated vomiting, confusion, or reduced consciousness should be treated as urgent and should not be managed only with home remedies or delayed consultation.
Why Brain Location Changes the Clinical Picture
Growth pattern must always be interpreted with tumor location. A slow-growing tumor in a silent or less functional area may remain unnoticed for longer. A smaller tumor near the speech center, motor cortex, optic pathway, brainstem, or areas controlling memory and personality may cause symptoms earlier and may be more difficult to operate on safely. [3,4]
This is why treatment decisions are not made by grade alone. A neurosurgeon must consider whether maximal safe resection is possible without damaging essential brain function. In some cases, advanced imaging, functional MRI, tractography, awake surgery, or brain mapping may be considered to protect speech, movement, and cognition. [3,4]
Clinical Impact on Treatment Planning
Growth pattern directly affects treatment urgency. A slow-growing, lower-grade glioma may allow time for detailed planning, second opinion, molecular testing, functional mapping, and discussion of long-term treatment sequencing. In selected cases, observation after surgery may be appropriate. [3,4]
Glioblastoma usually requires a more immediate and coordinated plan. The common sequence is maximal safe surgery when feasible, followed by radiation therapy with temozolomide, and then further treatment based on the patient’s condition, MGMT status, MRI response, and overall goals of care. [4,7]
This difference is critical for families because it shapes expectations. Lower-grade glioma care may feel like a long-term strategic plan. Glioblastoma care often feels like a race against time, where each decision must be medically sound, timely, and aligned with the patient’s strength and dignity.
Integrative and Ayurvedic Support Perspective
From an integrative care viewpoint, growth pattern helps define the level and timing of support. A patient with a slower-growing glioma may benefit from a long-term supportive plan focused on sleep, digestion, neurological stability, emotional balance, seizure coordination, rehabilitation, and disciplined follow-up.
A patient with glioblastoma often needs more intensive support during surgery recovery, radiation, chemotherapy, steroid use, anti-seizure medication, fatigue, appetite loss, cognitive changes, emotional distress, and recurrence monitoring. In such cases, Ayurvedic support should be supervised, individualized, and coordinated with the oncology team.
The purpose of supportive Ayurvedic care in this context is not to ignore tumor biology, but to support the person living with the disease. The patient is not only carrying a diagnosis; they are dealing with fear, fatigue, treatment burden, neurological uncertainty, and family pressure. A responsible integrative plan should protect strength, clarity, comfort, and quality of life while respecting modern neuro-oncology treatment.
Practical Clinical Message
Glioma growth can range from slow and gradual to fast and aggressive. Glioblastoma grows rapidly, infiltrates nearby brain tissue, and has a high risk of recurrence because microscopic cells often extend beyond the visible tumor. Therefore, growth pattern must be understood along with tumor grade, molecular markers, MRI findings, symptoms, and patient condition. [3,4,6,7]
For patients, the most useful question is not only “Is it glioma or glioblastoma?” but “How is this tumor behaving in my brain right now?” That answer guides treatment urgency, surgical planning, follow-up frequency, supportive care, and the overall strategy for preserving function and quality of life.
Difference in Symptoms
Why Symptoms Differ from Patient to Patient
Symptoms of glioma and glioblastoma depend mainly on four factors: tumor location, tumor size, growth speed, and swelling around the tumor. A tumor near the speech area may cause difficulty speaking, while a tumor near the motor cortex may cause weakness on one side of the body. A tumor near the frontal lobe may first appear as personality change, poor concentration, emotional instability, or decision-making difficulty. [5,6]
Table: Symptoms of Glioma vs Glioblastoma
| Symptom area | Glioma | Glioblastoma |
|---|---|---|
| Symptom speed | May develop slowly, especially in lower-grade gliomas | Often progresses faster over weeks or months |
| Seizures | Common, especially in lower-grade gliomas | Can occur and may worsen with progression |
| Headache | May be mild, persistent, or progressive | May become severe or worsen with swelling and pressure |
| Weakness | Depends on tumor location | May progress more quickly |
| Speech difficulty | Can occur when language areas are involved | May worsen rapidly if tumor or swelling affects speech areas |
| Memory and cognition | May be subtle initially | Can decline more noticeably |
| Personality or mood change | Possible, especially with frontal lobe involvement | May be more rapid or severe |
| Emergency concern | First seizure, worsening headache, weakness, vomiting, confusion | Same symptoms, often with greater urgency |
This is why brain tumor symptoms should not be judged only by severity. A mild but persistent symptom can still be important if it is new, progressive, or associated with seizures, weakness, confusion, vomiting, vision changes, or worsening headache. The NCI notes that signs and symptoms of adult brain and spinal cord tumors are not the same in every person, and brain tumors can affect function by growing into or pressing on important areas of the brain. [5]
Common Symptoms Seen in Gliomas
Gliomas may present with symptoms that develop slowly or suddenly. In many lower-grade gliomas, seizures are one of the first warning signs. Some patients may otherwise feel healthy, and the tumor may be discovered after a first seizure or after subtle neurological complaints.
Common symptoms may include headache, seizures, nausea or vomiting, weakness in an arm or leg, speech difficulty, memory problems, personality change, vision disturbance, balance problems, fatigue, or reduced concentration. [5,6]
The pattern is often more important than a single symptom. A one-time headache is common in the general population, but a headache that is new, persistent, worsening, associated with vomiting, or accompanied by neurological changes needs medical evaluation. Similarly, a first-time seizure in an adult should never be ignored. [5,6]
Symptoms in Lower-Grade Gliomas
Lower-grade gliomas often grow more slowly. Because of this slower growth, the brain may partly adapt for some time, and symptoms may remain subtle in the early stage. A patient may experience occasional seizures, mild speech difficulty, changes in memory, mood fluctuation, reduced work efficiency, or a persistent but not dramatic headache. [5,6]
This slower presentation can create a false sense of safety. Families may think the condition is minor because the patient is still walking, speaking, working, or eating normally. Clinically, that assumption can be risky. Even a low-grade glioma can infiltrate nearby brain tissue, recur, or transform into a higher-grade tumor over time. [5,6]
The practical message is that a slower symptom pattern should not lead to delay. It should lead to proper evaluation, MRI, histopathology when needed, molecular testing, and planned follow-up.
Symptoms in Glioblastoma
Glioblastoma usually behaves more aggressively. Symptoms may appear faster, worsen over weeks or months, and interfere more quickly with daily life. Patients may develop progressive headache, seizures, weakness, speech difficulty, confusion, personality change, memory decline, drowsiness, visual symptoms, or loss of coordination. [5,7]
The speed of symptom progression is often a major difference between lower-grade glioma and glioblastoma. Glioblastoma can produce swelling around the tumor and pressure effects, which may cause rapid neurological decline. Mayo Clinic highlights that glioblastoma survival and clinical course are influenced by factors such as age, tumor location, surgery, MGMT status, and overall response to treatment, which is why symptoms and prognosis must be assessed individually. [7]
In simple clinical language, lower-grade glioma may give the family time to investigate and plan, while glioblastoma often demands quicker coordination between neurosurgery, oncology, radiation therapy, rehabilitation, and supportive care.
Location-Based Symptoms
Brain tumor symptoms often reflect the area of the brain involved. This helps patients understand why two people with glioma may have completely different complaints.
A frontal lobe tumor may cause personality change, poor judgement, lack of motivation, mood change, weakness, or speech difficulty. A temporal lobe tumor may cause seizures, memory problems, language difficulty, or unusual sensations. A parietal lobe tumor may affect sensation, spatial awareness, reading, writing, or coordination. An occipital lobe tumor may cause visual symptoms. A cerebellar tumor may affect balance, posture, and coordination. A brainstem tumor may cause swallowing difficulty, double vision, weakness, walking difficulty, or breathing-related concerns. [5]
The NCI explains that different parts of the brain control functions such as thinking, learning, speech, movement, balance, posture, breathing, heart rate, and cranial nerve-related activities. This is why tumor location can strongly influence the symptom pattern. [5]
Warning Signs That Need Urgent Attention
Some symptoms should be treated as urgent, especially in a known or suspected brain tumor patient. These include a first-time seizure, repeated seizures, sudden weakness, sudden speech difficulty, severe or worsening headache, repeated vomiting, confusion, reduced consciousness, new vision loss, increasing drowsiness, severe imbalance, or rapid personality change. [1,5,6]
These symptoms may indicate swelling, bleeding, increased intracranial pressure, seizure activity, or progression of disease. They should not be managed only with home remedies, over-the-counter medicines, or unsupervised herbal products.
This is also important from an Ayurvedic care perspective. Supportive treatment can be valuable when properly supervised, but emergency neurological symptoms require immediate modern medical evaluation. The role of integrative care is to support the patient’s strength, comfort, recovery, and quality of life—not to delay urgent diagnosis or treatment. [1]
Difference Between Slow Symptoms and Fast Symptoms
The symptom difference between glioma and glioblastoma is often a difference of tempo. Lower-grade gliomas may produce symptoms gradually, sometimes beginning with seizures or subtle cognitive changes. Glioblastoma may produce faster deterioration because of rapid growth, infiltration, edema, and pressure on surrounding brain tissue. [5,6,7]
For families, this difference is useful but not absolute. A low-grade glioma can still cause sudden seizures. A glioblastoma may sometimes begin with symptoms that look mild. Therefore, the final judgement should never be based only on symptoms. It must be based on MRI findings, histopathology, WHO grade, molecular markers, and clinical examination. [5,6]
Why Symptoms Alone Cannot Confirm the Diagnosis
Symptoms can raise suspicion, but they cannot tell whether the tumor is a low-grade glioma, high-grade glioma, or glioblastoma. Headache, seizure, weakness, speech difficulty, or memory change may occur in many types of brain tumors. The exact diagnosis requires imaging and tissue-based confirmation whenever clinically possible. [5,6]
This is where many patients make a mistake. They compare symptoms with online stories and assume either the best or the worst. A patient with seizures may assume it is a low-grade tumor. Another patient with headache may fear glioblastoma immediately. Both assumptions may be wrong. The correct next step is diagnostic clarity.
Relevance for Supportive and Ayurvedic Care
Symptoms also guide supportive care planning. A patient with seizures needs coordination with anti-seizure medicines and careful monitoring. A patient with weakness may need physiotherapy and fall prevention. A patient with speech difficulty may need speech therapy. A patient with poor appetite, insomnia, anxiety, fatigue, or treatment-related weakness may need structured supportive care.
In Ayurvedic supportive planning, the symptom pattern helps identify where the patient needs the most support: sleep, digestion, strength, cognition, mood, appetite, fatigue, or post-treatment recovery. However, this must be done only after understanding the diagnosis, tumor grade, molecular profile, ongoing treatment, steroid use, anti-seizure medication, liver and kidney function, and oncology plan. [1]
Clinical Action Point
Glioma and glioblastoma can share many symptoms, but glioblastoma often causes faster and more aggressive neurological decline. Symptoms such as seizures, progressive headache, weakness, speech difficulty, confusion, vomiting, vision changes, personality change, or reduced consciousness should not be ignored. The safest approach is early MRI evaluation, specialist consultation, accurate diagnosis, and a treatment plan that combines modern neuro-oncology with supervised supportive care where appropriate. [1,5,6,7]
Difference in Diagnosis
Diagnosis Begins With Imaging
The diagnosis of glioma or glioblastoma usually begins with a neurological examination and brain imaging. MRI brain with contrast is the preferred investigation because it gives detailed information about tumor location, size, swelling, enhancement pattern, and involvement of nearby brain structures. CT scan may be used in emergency situations, especially when the patient has sudden weakness, seizure, vomiting, altered consciousness, or suspected bleeding. [4,5]
Imaging can strongly suggest a glioma, but it cannot always confirm the exact tumor type or grade. A tumor that looks aggressive on MRI may need tissue diagnosis before the final treatment plan is made.
Tissue Diagnosis Is Essential
The exact diagnosis usually requires biopsy or surgical removal of the tumor, whenever medically safe. The tissue is then examined under a microscope to identify the tumor type and grade. This helps distinguish low-grade glioma, high-grade glioma, glioblastoma, lymphoma, metastasis, infection, or other brain lesions that may look similar on imaging. [2,3,4]
For glioblastoma, tissue diagnosis is especially important because treatment is urgent and intensive. Before radiation or chemotherapy is planned, doctors need to know whether the tumor is truly glioblastoma or another type of high-grade glioma.
Molecular Testing Completes the Diagnosis
Modern glioma diagnosis is incomplete without molecular testing. Important markers include IDH mutation status, 1p/19q codeletion, MGMT promoter methylation, TERT promoter mutation, EGFR amplification, ATRX, TP53, CDKN2A/B status, and chromosome 7 gain/chromosome 10 loss where relevant. [2,3,4]
These markers help doctors classify the tumor correctly, estimate prognosis, and choose treatment more precisely. For example, adult glioblastoma is generally classified as glioblastoma, IDH-wildtype, CNS WHO grade 4. A grade 4 tumor with IDH mutation may be classified differently, commonly as astrocytoma, IDH-mutant, CNS WHO grade 4. [2,3]
Diagnosis Guides Treatment Urgency
A lower-grade glioma may allow time for planned surgery, observation, molecular review, or staged treatment. Glioblastoma usually requires faster coordination between neurosurgery, radiation oncology, medical oncology, rehabilitation, and supportive care. [3,4]
This is why patients should not begin comparing survival numbers or treatment options until the diagnosis is complete. The words “brain tumor” or “glioma” are not enough. The real treatment roadmap depends on tumor type, WHO grade, molecular profile, location, symptoms, and the patient’s overall condition.
Relevance for Ayurvedic and Integrative Care
Ayurvedic support should begin only after diagnostic clarity. MRI, histopathology, and molecular testing are necessary before planning any supportive regimen. This is especially important when the patient is receiving steroids, anti-seizure medicines, chemotherapy, radiation therapy, or blood-thinning medicines. [1,4]
The role of supervised Ayurvedic care is to support strength, appetite, sleep, digestion, mood, fatigue, cognition, and quality of life. It should not delay biopsy, surgery, radiation, chemotherapy, or emergency neurological care when these are medically required. [1]
Practical Clinical Message
Glioma and glioblastoma cannot be reliably separated by symptoms alone. Accurate diagnosis needs MRI, tissue confirmation, histopathology, and molecular testing. Once the exact tumor identity is known, the patient can move from uncertainty to a structured treatment plan that includes modern neuro-oncology, rehabilitation, nutrition, and safe supportive care where appropriate. [1,2,3,4,5]
Difference in Treatment
Treatment Depends on the Exact Diagnosis
Treatment for glioma and glioblastoma is not decided by the word “brain tumor” alone. It depends on the tumor type, CNS WHO grade, molecular profile, location, symptoms, age, general health, neurological function, and how much tumor can be safely removed. A lower-grade glioma and glioblastoma may both belong to the glioma family, but their treatment pathways are often very different. [4]
The first goal is always diagnostic clarity. Once MRI, histopathology, and molecular testing confirm the tumor identity, the medical team can decide whether the patient needs surgery, observation, radiation therapy, chemotherapy, targeted therapy, Tumor Treating Fields, rehabilitation, supportive care, or clinical trial options. [4,13]
Treatment of Lower-Grade Gliomas
In lower-grade gliomas, treatment may be more planned and staged. Many patients undergo maximal safe surgical removal first. If the tumor is completely removed and the patient has favorable features, close MRI monitoring may be considered in selected cases. If there is residual tumor, higher-risk disease, symptoms, or progression, radiation therapy and chemotherapy may be advised. [4,13]
For some grade 2 IDH-mutant astrocytomas or oligodendrogliomas, newer targeted treatment options may also be relevant. Vorasidenib has been approved for selected patients with grade 2 astrocytoma or oligodendroglioma with susceptible IDH1 or IDH2 mutation after surgery, where immediate radiation and chemotherapy may be deferred in appropriate cases. [12,13]
The treatment aim in lower-grade glioma is long-term tumor control while preserving speech, memory, movement, cognition, fertility, work capacity, and quality of life as much as possible.
Treatment of Glioblastoma
Glioblastoma usually needs faster and more intensive treatment. The standard approach for newly diagnosed glioblastoma commonly includes maximal safe surgical resection when possible, followed by radiation therapy with temozolomide chemotherapy, and then additional temozolomide cycles. This approach is based on the landmark Stupp protocol and remains a major foundation of glioblastoma care. [4,8]
In selected patients, Tumor Treating Fields may be added during maintenance temozolomide. This option is not suitable for everyone, but it may be discussed with the neuro-oncology team depending on the patient’s condition, preferences, access, and treatment goals. [11]
Because glioblastoma is infiltrative, surgery alone is usually not enough. Even after good surgical removal, microscopic tumor cells may remain in surrounding brain tissue. This is why combined therapy and regular MRI follow-up are essential. [4,8]
Treatment at Recurrence
Recurrence is more common in glioblastoma than in many lower-grade gliomas. Treatment after recurrence is individualized. Options may include repeat surgery, re-irradiation in selected cases, chemotherapy, bevacizumab, clinical trials, targeted treatment if a relevant marker is present, and supportive or palliative care. [4]
The goal at recurrence is not only survival extension. It is also preservation of neurological function, reduction of swelling, seizure control, symptom relief, emotional support, and maintaining dignity and quality of life.
Supportive and Ayurvedic Care Position
Supportive care is important in both glioma and glioblastoma, but the intensity differs. Lower-grade glioma patients may need long-term support for seizures, sleep, cognition, fatigue, emotional stress, recovery after surgery, and regular follow-up. Glioblastoma patients often need more intensive support during surgery recovery, radiation, chemotherapy, steroid use, appetite loss, weakness, mood changes, cognitive decline, and family distress.
Ayurvedic support should be positioned responsibly. It may help support strength, digestion, sleep, appetite, fatigue, mental stability, and quality of life when planned by a qualified physician. However, it should not replace surgery, radiation therapy, chemotherapy, anti-seizure medicines, steroids, emergency care, or oncology follow-up when these are required.
Clinical Direction
The treatment difference is clear: many lower-grade gliomas are managed with a long-term strategy, while glioblastoma usually needs urgent multimodal treatment. The best outcomes come from timely diagnosis, maximal safe treatment, molecularly informed planning, rehabilitation, nutrition, psychological support, and carefully supervised integrative care. [4,8,11,12,13]
Treatment Side Effects and Quality of Life
Why Quality of Life Must Be Part of Treatment
In glioma and glioblastoma care, treatment success should not be measured only by MRI response. A good treatment plan must also protect the patient’s strength, speech, memory, sleep, appetite, mobility, mood, dignity, and daily independence. This is especially important because brain tumors affect the organ that controls personality, thinking, movement, emotion, and communication.
For a patient, the question is not only “How long can treatment control the tumor?” but also “How well can I live during and after treatment?” This is where quality of life becomes a central part of care, not an optional extra. [4,15,16]
Side Effects After Surgery
Surgery is often the first major treatment step when the tumor can be safely removed. After surgery, some patients recover quickly, while others may experience fatigue, headache, weakness, speech difficulty, memory changes, seizures, mood fluctuation, or reduced confidence in daily activities.
These effects depend on tumor location, the amount of tumor removed, swelling, bleeding risk, pre-existing neurological symptoms, and the patient’s general health. Rehabilitation, physiotherapy, speech therapy, occupational therapy, nutrition, and family support can make a significant difference in recovery. [4]
Side Effects of Radiation Therapy
Radiation therapy is commonly used in high-grade gliomas and glioblastoma, and in selected lower-grade gliomas when needed. Common issues may include tiredness, scalp irritation, hair loss in the treated area, headache, nausea, reduced appetite, sleep disturbance, and temporary worsening of neurological symptoms due to swelling.
Some patients may also experience delayed cognitive effects, especially when larger brain areas are treated or when the patient already has memory or concentration problems. For this reason, patients should be monitored not only for tumor control but also for thinking ability, mood, fatigue, and daily functioning. [4]
Side Effects of Chemotherapy
Temozolomide is commonly used in glioblastoma treatment and in selected glioma cases. Possible side effects include nausea, vomiting, appetite loss, constipation, fatigue, low blood counts, infection risk, liver enzyme changes, and general weakness. Blood tests are usually required during treatment to monitor safety. [4]
For families, chemotherapy-related fatigue can be difficult to understand because the patient may look physically normal but feel deeply exhausted. This fatigue is not ordinary tiredness. It may affect walking, eating, conversation, sleep, mood, and willingness to continue treatment.
Tumor Treating Fields and Practical Burden
Tumor Treating Fields may be considered in selected glioblastoma patients during maintenance therapy. While it may offer benefit for some patients, it also creates practical challenges. The patient has to wear the device for long hours, manage scalp care, handle skin irritation, and maintain discipline with daily use. [11]
This treatment is not only a medical decision; it is also a lifestyle decision. The patient and family should understand the expected commitment before choosing it.
Steroids, Anti-Seizure Medicines, and Daily Life
Many glioma and glioblastoma patients receive steroids to reduce brain swelling. Steroids may improve headache, weakness, or neurological symptoms, but they can also cause increased appetite, weight gain, high blood sugar, acidity, mood changes, sleep disturbance, muscle weakness, infection risk, and facial puffiness.
Anti-seizure medicines are also commonly used when seizures are present or risk is high. These medicines may cause drowsiness, dizziness, mood changes, fatigue, or coordination problems in some patients. They should not be stopped suddenly without medical advice.
This is why supportive care must include medicine review, symptom tracking, sleep support, fall prevention, diet planning, and regular communication with the treating team.
Emotional and Family Burden
Glioma and glioblastoma do not affect only the patient. They affect the entire family. Fear of recurrence, uncertainty about survival, financial pressure, treatment travel, caregiving responsibility, personality changes, and communication difficulties can place heavy emotional stress on caregivers.
Patients may experience anxiety, depression, irritability, anger, hopelessness, or withdrawal. These symptoms should not be dismissed as “weakness.” They may be part of the disease burden, treatment burden, or emotional shock of diagnosis. Counselling, caregiver education, spiritual support, structured routine, and compassionate communication are important parts of care.
Where Ayurvedic Support May Fit
This is one of the strongest reasons patients look for supervised Ayurvedic and integrative care. The aim should be supportive, not substitutive. Ayurvedic care may be considered to support digestion, appetite, sleep, fatigue, emotional balance, recovery strength, and overall quality of life when planned by a qualified physician and coordinated with oncology treatment. [1]
In Ayurvedic language, this support may be understood through Rasayana, Ojas protection, Agni correction, Vata-Kapha balance, Majja Dhatu support, and restoration of strength after major treatment. However, this must be individualized. A patient after surgery is not the same as a patient receiving radiation, chemotherapy, steroids, anti-seizure medicines, or recurrence treatment.
Safety With Herbs and Supplements
Patients should not start herbs, supplements, high-dose vitamins, or over-the-counter “brain tumor” products without informing the oncology team. Some products may interact with chemotherapy, radiation, steroids, anti-seizure medicines, blood thinners, diabetes medicines, or liver and kidney function. [15,16,18]
This is especially important during active treatment. Even natural products can be unsafe if the timing, dose, patient condition, or drug interaction risk is not considered. Responsible integrative care should protect the patient from both under-treatment and unsafe over-treatment.
Clinical Direction
Treatment for glioma and glioblastoma can extend life, reduce symptoms, and control disease, but it can also create physical, emotional, neurological, and family burden. A complete care plan should therefore include tumor-directed treatment, rehabilitation, nutrition, seizure control, sleep support, psychological care, caregiver guidance, and safe supervised integrative support.
The goal is not only to fight the tumor. The goal is to preserve the person behind the diagnosis. [1,4,11,15,16,18]
Difference in Survival and Prognosis
Why Survival Cannot Be Explained With One Number
Survival in glioma depends on the exact tumor type, CNS WHO grade, molecular profile, age, neurological condition, tumor location, extent of surgical removal, response to radiation or chemotherapy, and recurrence pattern. This is why the word “glioma” alone is not enough to estimate prognosis. A low-grade IDH-mutant glioma and glioblastoma, IDH-wildtype, may both belong to the glioma family, but their clinical course can be very different. [4,7,10,14]
For patients and families, survival statistics should be understood as population-level guidance, not as a fixed prediction for one individual. They help doctors discuss risk, but they do not decide the destiny of a particular patient.
Prognosis in Lower-Grade Gliomas
Lower-grade gliomas often have a better prognosis than glioblastoma, especially when favorable molecular markers are present. IDH-mutant gliomas and oligodendrogliomas with 1p/19q codeletion generally tend to behave more slowly than aggressive IDH-wildtype tumors. [4,10]
However, lower-grade does not mean risk-free. These tumors may recur, progress, or transform into higher-grade disease over time. Long-term MRI follow-up is therefore essential, even when the patient is clinically stable.
The main clinical aim in lower-grade glioma is to control the tumor while preserving brain function, cognition, speech, movement, fertility, work ability, and quality of life for as long as possible.
Prognosis in Glioblastoma
Glioblastoma has a more difficult prognosis than most other gliomas because it is grade 4, fast-growing, infiltrative, and commonly recurrent. Mayo Clinic reports that, in large studies, about 5% to 7% of people with glioblastoma are alive five years after diagnosis, and many patients have a median life expectancy of about 12 to 18 months with standard treatment. [7]
These numbers must be communicated with sensitivity. Some patients live shorter, while others live longer. Younger age, good functional status, safe maximal tumor removal, MGMT promoter methylation, access to standard treatment, and careful supportive care may influence outcomes. [4,7,9]
Role of Molecular Markers in Prognosis
Molecular markers are now central to prognosis. IDH status is one of the most important markers in diffuse gliomas. IDH-mutant tumors often have a more favorable course than IDH-wildtype aggressive gliomas. 1p/19q codeletion supports the diagnosis of oligodendroglioma and is generally associated with a better treatment response and longer disease course. [10]
In glioblastoma, MGMT promoter methylation is especially important because it may predict better response to temozolomide chemotherapy. The landmark MGMT study found that patients whose tumors had a methylated MGMT promoter benefited more from temozolomide than those without methylation. [9]
This is why two patients with the same tumor grade may still have different outcomes. The name of the tumor matters, but the biology behind the name matters even more.
Why Survival Statistics Can Mislead Patients
Many patients search online and immediately compare themselves with survival numbers. This can create either unnecessary fear or unrealistic confidence. A survival statistic does not know the patient’s age, MRI findings, surgical result, IDH status, MGMT status, neurological strength, treatment tolerance, or recurrence pattern.
Population reports such as CBTRUS are useful for understanding the overall burden of brain and CNS tumors, but individual prognosis must be discussed with the treating neuro-oncology team. CBTRUS reported that the five-year relative survival rate for malignant brain and other CNS tumors was 34.8%, while it was 91.7% for non-malignant tumors, showing how widely outcomes differ across tumor types. [14]
Prognosis Is Also About Function
In brain tumors, prognosis is not only about months or years. It is also about neurological function. A patient may ask: Will I be able to walk? Will I speak clearly? Will I remember my family? Will seizures be controlled? Will I tolerate treatment? Will I remain independent?
These questions are clinically important. Survival and quality of life must be discussed together, especially in glioblastoma, where treatment decisions often involve balancing tumor control, side effects, neurological function, and patient dignity.
Relevance for Ayurvedic and Integrative Support
From an integrative care perspective, prognosis helps define the goal of support. In lower-grade glioma, the focus may be long-term strength, seizure coordination, sleep, digestion, emotional stability, rehabilitation, and disciplined follow-up.
In glioblastoma, supportive care may need to be more intensive, especially during surgery recovery, radiation, chemotherapy, steroid use, appetite loss, fatigue, cognitive decline, mood changes, and recurrence monitoring. Ayurvedic care, when used, should be supervised and coordinated with oncology treatment. It should support the patient’s strength and quality of life, not replace urgent neuro-oncology care.
Practical Clinical Message
Glioma survival varies widely. Glioblastoma usually has a more guarded prognosis because it is grade 4, fast-growing, and highly recurrent. The most reliable prognosis comes from combining tumor grade, molecular markers, surgical result, treatment response, patient age, neurological condition, and follow-up findings. [4,7,9,10,14]
Patients should not judge their future from the word “glioma” alone or from one online statistic. The right question is: What is my exact tumor type, grade, molecular profile, treatment response, and current neurological status? That answer gives a more honest and useful prognosis.
Recurrence: Why Glioblastoma Comes Back More Often
Why Recurrence Is Common in Glioblastoma
One of the most difficult realities of glioblastoma is its high risk of recurrence. Even when the visible tumor is removed successfully during surgery, glioblastoma cells often extend microscopically into the surrounding brain tissue. These cells may not be clearly visible on MRI and may not be removable without damaging important brain functions such as speech, movement, memory, or vision. [3,4,7]
This is why glioblastoma is treated as an infiltrative disease, not simply as a single mass. Surgery reduces the main tumor burden, but radiation and chemotherapy are usually needed afterward to target residual microscopic disease. Despite this combined approach, recurrence remains common because of the tumor’s aggressive biology and treatment resistance. [3,4]
Why Surgery Alone Is Usually Not Enough
In many cancers, complete surgical removal can sometimes be curative. Glioblastoma behaves differently. It grows into the surrounding brain, making it difficult to define a clean border between tumor and healthy tissue. Neurosurgeons aim for maximal safe resection, which means removing as much tumor as possible while protecting essential neurological function. [3,4]
The word “safe” is important. Removing too much tissue in a critical brain area may leave the patient with permanent weakness, speech loss, memory damage, or poor quality of life. Therefore, even a technically successful surgery may leave behind microscopic cells that later cause recurrence. [3,4]
How Recurrence Is Detected
Recurrence is usually monitored through regular MRI scans and clinical assessment. Doctors look for new or increasing enhancement, swelling, tumor growth, worsening neurological symptoms, seizures, headache, weakness, speech difficulty, cognitive decline, or changes in daily functioning. [4]
However, MRI interpretation after treatment can be complex. Radiation effects, inflammation, treatment-related changes, and true tumor progression may sometimes look similar. In such cases, advanced imaging, repeated scans, clinical judgment, and multidisciplinary review may be needed. [3,4]
Recurrence in Lower-Grade Gliomas
Lower-grade gliomas can also recur, but the pattern is often different. Some may return slowly after months or years. Others may transform into a higher-grade tumor over time. This is why long-term follow-up is essential even when the patient feels well after surgery or initial treatment. [3,4]
A stable MRI does not mean follow-up can be ignored. Glioma care is a long-term strategy. The aim is early detection of progression, timely treatment, and preservation of neurological function.
Treatment Options After Recurrence
Treatment after recurrence is individualized. There is no single standard plan that fits every patient. Options may include repeat surgery, re-irradiation in selected cases, chemotherapy, bevacizumab, Tumor Treating Fields, targeted treatment if a relevant marker is present, clinical trials, rehabilitation, seizure control, steroid adjustment, and palliative or supportive care. [3,4,11]
The choice depends on tumor location, previous treatment, time since radiation, neurological condition, age, performance status, molecular profile, symptoms, and the patient’s goals. At recurrence, the medical team must balance disease control with quality of life.
Why Families Should Understand Recurrence Early
Families often feel shocked when glioblastoma returns after surgery, radiation, and chemotherapy. Many assume recurrence means the treatment failed. Clinically, this is not always true. Recurrence often reflects the natural behavior of glioblastoma: rapid growth, microscopic spread, cellular diversity, and resistance to treatment. [3,4,7]
When patients understand this early, they can plan better. They can ask about MRI follow-up, warning signs, rehabilitation, steroid side effects, seizure care, nutritional support, mental health support, and what treatment choices may be available if the tumor returns.
Integrative and Ayurvedic Support During Recurrence Risk
During follow-up or recurrence management, supportive care becomes very important. Patients may struggle with fear, fatigue, sleep disturbance, appetite loss, weakness, seizures, anxiety, memory problems, or treatment exhaustion. A supervised Ayurvedic and integrative plan may help support digestion, sleep, strength, emotional balance, and quality of life when coordinated with the oncology team.
However, recurrence should never be managed only with herbs, diet, or home remedies. New neurological symptoms, MRI progression, seizures, worsening headache, vomiting, confusion, or weakness require prompt medical evaluation. Ayurvedic care should support the patient’s resilience and comfort, not delay neuro-oncology treatment.
Practical Clinical Message
Glioblastoma comes back more often because it is fast-growing, deeply infiltrative, and difficult to remove completely at a microscopic level. Recurrence is not always due to poor treatment; it is often part of the disease biology. Regular MRI follow-up, early symptom reporting, multidisciplinary review, and safe supportive care are essential for protecting both survival and quality of life. [3,4,7,11]
Ayurvedic Perspective: How This Difference Matters in Holistic Care
Why the Difference Matters in Ayurveda
From an Ayurvedic and integrative care perspective, glioma and glioblastoma should not be approached as identical conditions. A slow-growing low-grade glioma, a high-grade glioma, and glioblastoma may all involve abnormal growth in the brain, but their clinical urgency, treatment intensity, recurrence risk, and supportive care needs are very different.
Table: Ayurvedic and Integrative Support by Treatment Stage
| Treatment stage | Common patient concerns | Supportive Ayurvedic/integrative focus |
|---|---|---|
| Before surgery | Anxiety, poor sleep, fear, weakness | Calm routine, digestion support, sleep support, physician-guided preparation |
| After surgery | Fatigue, weakness, wound recovery, speech or movement issues | Strength, nutrition, rehabilitation support, sleep, digestion, emotional stability |
| During radiation | Tiredness, scalp irritation, appetite loss, sleep disturbance | Fatigue support, appetite support, hydration, rest, gentle routine |
| During chemotherapy | Nausea, low appetite, low blood counts, weakness | Digestive support, strength support, careful monitoring, drug-interaction caution |
| During steroid use | High appetite, acidity, mood change, sleep issues, blood sugar changes | Diet discipline, sleep support, acidity management, medical monitoring |
| During follow-up | Fear of recurrence, seizure concern, fatigue | Long-term stability, regular MRI follow-up, supervised Rasayana support |
| Recurrence setting | Anxiety, neurological decline, treatment exhaustion | Symptom support, dignity, comfort, caregiver support, coordinated oncology care |
Ayurveda should therefore begin with diagnostic clarity. The patient must first know the exact tumor type, CNS WHO grade, molecular profile, MRI findings, neurological status, and current treatment plan. Only then can supportive Ayurvedic care be planned responsibly. In modern neuro-oncology, glioblastoma is a grade 4, aggressive, infiltrative tumor, while glioma is a broader category that may include slower or faster-growing tumors. This difference changes the entire care strategy. [2,3,6,7]
Ayurvedic Understanding of Brain Tumor Support
Classical Ayurvedic texts do not describe “glioma” or “glioblastoma” by modern pathological names. However, brain tumors may be clinically discussed through concepts such as Arbuda, Granthi, Vata-Kapha vitiation, Majja Dhatu involvement, Srotorodha, Ama accumulation, and Ojas depletion.
In Sushruta Samhita, Nidana Sthana, Chapter 11, Arbuda is discussed in the context of deep-seated abnormal growths involving aggravated Doshas and tissue derangement. This does not mean Arbuda is exactly equal to glioma, but it gives an Ayurvedic framework for understanding abnormal tissue growth, chronicity, obstruction, and systemic imbalance.
Because the brain is closely related to Majja Dhatu, Manovaha Srotas, Prana Vayu, and higher neurological function, glioma care in Ayurveda should not be reduced to “tumor treatment” alone. The patient may also need support for sleep, digestion, cognition, mood, strength, appetite, fatigue, seizure-related vulnerability, and treatment recovery. [1]
Low-Grade Glioma: Long-Term Supportive Strategy
In lower-grade gliomas, the Ayurvedic focus is usually long-term support. These patients may live with the diagnosis for many years, but they still require regular MRI follow-up, neurological monitoring, and careful coordination with modern treatment.
The supportive goals may include maintaining Agni, reducing Ama, supporting sleep, stabilizing Vata, improving mental strength, supporting recovery after surgery, and protecting Ojas. If the patient has seizures, speech difficulty, anxiety, poor sleep, or fatigue, the supportive plan must be adjusted accordingly.
This is also where Rasayana therapy may have a role under supervision. In Charaka Samhita, Chikitsa Sthana, Chapter 1, Rasayana is described for promoting strength, vitality, longevity, tissue nourishment, and resistance. Medhya Rasayana herbs described in the same broad Rasayana context are traditionally used for intellect, memory, and mental stability. However, in glioma patients, such support must be individualized and should not replace MRI monitoring, surgery, radiation, chemotherapy, or targeted treatment when these are needed.
Glioblastoma: Intensive Support Alongside Neuro-Oncology
Glioblastoma needs a different level of caution. It is not a slow or mild condition. It is grade 4, fast-growing, infiltrative, and has a high recurrence risk. Therefore, Ayurvedic care in glioblastoma should be positioned as supportive and integrative, not as a substitute for surgery, radiation therapy, temozolomide, steroids, anti-seizure medicines, or emergency neurological care. [3,4,7]
In glioblastoma, the patient often faces multiple burdens at the same time: surgery recovery, radiation fatigue, chemotherapy-related nausea or weakness, steroid side effects, sleep disturbance, appetite loss, cognitive changes, emotional distress, and fear of recurrence. A supervised Ayurvedic plan may be useful for supporting digestion, appetite, sleep, energy, emotional steadiness, and overall quality of life.
The value of integrative care is not in competing with modern neuro-oncology. Its value is in supporting the patient while neuro-oncology addresses the tumor.
Rasayana and Medhya Support
Rasayana therapy is one of the most relevant Ayurvedic principles in supportive oncology care. The aim is to improve strength, tissue resilience, vitality, digestion, mental stability, and recovery capacity. In brain tumor patients, Medhya Rasayana may be considered when cognitive function, sleep, emotional balance, and neurological recovery are important concerns.
Classical references include Charaka Samhita, Chikitsa Sthana, Chapter 1, where Rasayana and Medhya Rasayana principles are described. Herbs traditionally discussed in Medhya Rasayana contexts include Mandukaparni, Yashtimadhu, Guduchi, and Shankhpushpi. In clinical practice, other herbs such as Brahmi, Ashwagandha, Guduchi, Yashtimadhu, and turmeric-derived curcumin are often discussed in integrative oncology settings.
A modern review by Newton discussed Ayurvedic medicine in relation to brain cancer and summarized interest in herbs such as ashwagandha, curcumin, guduchi, and other botanicals. However, much of this evidence remains preclinical, experimental, or supportive in nature. It should not be presented as proof that herbs alone can cure glioma or glioblastoma. [17]
Safety Comes Before Supplementation
Safety is especially important in brain tumor patients. Many patients are already taking steroids, anti-seizure medicines, chemotherapy, pain medicines, sleep medicines, anticoagulants, diabetes medicines, or blood pressure medicines. Herbal medicines and supplements may interact with these drugs or affect liver enzymes, bleeding risk, sedation, immunity, or treatment tolerance.
The National Cancer Institute advises patients to inform their cancer care team about complementary therapies, including herbs, vitamins, supplements, and special diets. NCCIH also cautions that complementary approaches should not replace proven cancer treatment, and Memorial Sloan Kettering highlights the importance of checking herb-drug interactions before using botanicals during cancer care. [15,16,18]
This is why unsupervised “brain tumor packages,” online formulations, or aggressive detox therapies can be risky. In glioma and glioblastoma, the wrong timing or wrong combination may harm the patient or interfere with treatment.
Personalized Care, Not Generic Protocols
A responsible Ayurvedic plan should be based on the individual patient, not only the disease name. The physician should assess tumor type, grade, molecular markers, treatment stage, surgery status, radiation schedule, chemotherapy plan, steroid use, anti-seizure medicines, digestion, bowel pattern, sleep, appetite, strength, liver and kidney function, emotional state, and family support.
A patient after surgery may need a different plan from a patient receiving radiation. A patient on temozolomide may need a different plan from a patient under MRI surveillance. A patient with glioblastoma recurrence requires a different level of caution from a patient with stable low-grade glioma.
This is the clinical difference between personalized integrative care and generic alternative treatment.
Practical Clinical Message
Ayurveda can play a meaningful supportive role in glioma care when used with medical clarity, safety, and proper supervision. In lower-grade glioma, the focus may be long-term stability, recovery, cognition, sleep, digestion, and strength. In glioblastoma, the focus is often treatment tolerance, fatigue, appetite, mood, sleep, steroid-related issues, neurological comfort, caregiver support, and quality of life.
The foundation must remain clear: glioma and glioblastoma require accurate diagnosis through MRI, histopathology, and molecular testing. Ayurvedic support should be individualized and coordinated with neuro-oncology care. The aim is to support the patient as a whole person while respecting the seriousness of the tumor. [1,15,16,17,18]
Safety Section: What Patients Should Not Do
Why Safety Must Come First
Glioma and glioblastoma are serious brain tumors that require accurate diagnosis, timely treatment, and continuous medical monitoring. Supportive care, including Ayurvedic and integrative care, may have a valuable role in improving strength, sleep, appetite, fatigue, emotional stability, and quality of life. However, safety must always come before experimentation.
The biggest risk for patients is not only the tumor itself, but delay, confusion, wrong advice, unsafe supplements, and missed emergency symptoms. A brain tumor patient should never be treated casually or with a generic protocol. Every decision should be guided by tumor type, grade, molecular markers, MRI findings, neurological condition, and current medicines. [1,4,15,16,18]
Do Not Delay Diagnosis
Patients should not delay MRI, biopsy, histopathology, or molecular testing when a glioma or glioblastoma is suspected. Symptoms such as seizures, persistent headache, weakness, speech difficulty, vomiting, confusion, vision changes, or personality change need proper neurological evaluation.
A glioma cannot be accurately understood by symptoms alone. The exact diagnosis depends on imaging, tissue examination, and molecular markers such as IDH status, 1p/19q codeletion, MGMT promoter methylation, EGFR amplification, TERT promoter mutation, and other relevant findings. Without this clarity, treatment planning remains incomplete. [4]
Do Not Replace Standard Treatment With Home Remedies
Patients should not replace surgery, radiation therapy, chemotherapy, anti-seizure medicines, steroids, or emergency care with home remedies, diet plans, supplements, or unsupervised herbal medicines.
This is especially important in glioblastoma, which is a grade 4, fast-growing, infiltrative tumor. Delaying neuro-oncology treatment may allow the tumor to progress, increase swelling, worsen neurological function, and reduce future treatment options. Supportive Ayurvedic care should be used responsibly alongside appropriate medical treatment, not as a substitute for it. [1,4]
Do Not Start Herbs or Supplements Without Medical Supervision
Many patients assume that herbs, supplements, vitamins, detox powders, or “natural cancer products” are automatically safe. This is not correct. Natural products can still interact with chemotherapy, radiation therapy, steroids, anti-seizure medicines, blood thinners, diabetes medicines, blood pressure medicines, sedatives, and liver or kidney function. [15,16,18]
Patients should inform their oncologist and Ayurvedic physician about every product they are taking, including herbal capsules, churnas, decoctions, oils, rasayana preparations, high-dose vitamins, minerals, protein powders, mushrooms, curcumin products, and over-the-counter supplements. Safety depends on timing, dose, formulation, treatment stage, and the patient’s condition.
Do Not Stop Prescribed Medicines Suddenly
Anti-seizure medicines, steroids, chemotherapy medicines, pain medicines, and other prescribed drugs should not be stopped suddenly without medical advice. Sudden withdrawal of anti-seizure medicines may trigger seizures. Stopping steroids abruptly may worsen brain swelling or cause withdrawal-related complications.
If side effects occur, the correct step is to inform the treating doctor. Dose adjustment, alternative medicines, supportive treatment, or monitoring may be needed. Self-adjusting medicines can be dangerous in brain tumor patients.
Do Not Ignore Emergency Warning Signs
Certain symptoms require urgent medical attention. These include first-time seizure, repeated seizures, sudden weakness, sudden speech difficulty, severe or worsening headache, repeated vomiting, confusion, reduced consciousness, new vision loss, severe imbalance, increasing drowsiness, or rapid personality change.
These symptoms may indicate tumor progression, brain swelling, bleeding, seizure activity, raised intracranial pressure, infection, or treatment complications. They should not be managed only with Ayurvedic medicines, home care, painkillers, or waiting for symptoms to settle. [1,4]
Do Not Buy Generic “Brain Tumor Cure” Packages
Patients should avoid generic online packages that claim to cure glioma, glioblastoma, brain tumors, or cancer without reviewing MRI reports, pathology, molecular markers, medicines, liver and kidney function, and current treatment status.
Glioma care must be individualized. A patient with stable low-grade glioma is different from a patient with glioblastoma recurrence. A patient after surgery is different from one receiving radiation and temozolomide. A patient on steroids and anti-seizure medicines needs a different level of caution. Generic treatment can be ineffective, unsafe, or misleading.
Do Not Compare Your Case With Another Patient
Two patients may both have “glioma,” but their disease may be completely different. One may have a grade 2 IDH-mutant glioma, another may have oligodendroglioma with 1p/19q codeletion, and another may have glioblastoma, IDH-wildtype, CNS WHO grade 4.
Comparing survival, treatment response, or recovery based only on the word “glioma” can create unnecessary fear or false confidence. The correct comparison must be based on tumor type, grade, molecular profile, treatment received, age, neurological condition, and MRI response. [4]
Do Not Neglect Nutrition, Rehabilitation, and Mental Health
Safety is not only about avoiding harmful medicines. It is also about not neglecting the basics of recovery. Poor nutrition, uncontrolled seizures, sleeplessness, falls, untreated depression, caregiver burnout, steroid side effects, and lack of rehabilitation can reduce quality of life.
Glioma and glioblastoma patients may need physiotherapy, speech therapy, occupational therapy, psychological counselling, seizure education, sleep support, caregiver training, and nutritional planning. These supportive measures can help protect independence and dignity during treatment.
Safer Clinical Approach
The safer approach is to build a coordinated plan. The patient should have a neuro-oncology team for diagnosis and tumor-directed treatment, and a qualified Ayurvedic physician for supportive care where appropriate. Both sides should know the patient’s medicines, treatment schedule, symptoms, and risks.
Ayurvedic care may support digestion, appetite, sleep, fatigue, emotional balance, strength, and quality of life, but it should be personalized and medically supervised. It should never delay urgent treatment or replace evidence-based neuro-oncology care. [1,15,16,18]
Clinical Safety Message
The safest path for glioma and glioblastoma patients is clarity, coordination, and caution. Do not delay diagnosis. Do not replace standard treatment when it is needed. Do not use herbs or supplements without supervision. Do not ignore emergency neurological symptoms. A responsible integrative plan should protect the patient from both disease progression and unsafe treatment choices. [1,4,15,16,18]
Patient Decision Guide: Questions to Ask Your Doctor
Why These Questions Matter
After a glioma or glioblastoma diagnosis, many families feel pressure to make quick decisions. This is understandable, but treatment should not begin from fear alone. It should begin from clarity. The most important information is the exact tumor type, CNS WHO grade, molecular profile, location, symptoms, and treatment goal. [2,3,4]
A patient who knows the right questions can have a more meaningful discussion with the neurosurgeon, neuro-oncologist, radiation oncologist, and supportive care team. This helps the family understand what is urgent, what can be planned, and where supervised integrative or Ayurvedic support may safely fit.
Questions About the Exact Diagnosis
Ask your doctor:
- What exact type of glioma do I have?
- Is it astrocytoma, oligodendroglioma, glioblastoma, or another tumor type?
- What is the CNS WHO grade?
- Is this a low-grade glioma, high-grade glioma, or glioblastoma?
- Is the tumor IDH-mutant or IDH-wildtype?
- Is there 1p/19q codeletion?
- Has MGMT promoter methylation been tested?
- Are TERT promoter mutation, EGFR amplification, ATRX, TP53, CDKN2A/B, or chromosome 7/10 changes relevant in my case?
- Is the diagnosis final, or are more tests needed?
These questions are important because modern glioma diagnosis depends on both histopathology and molecular testing. A broad label like “glioma” is not enough for accurate prognosis or treatment planning. [2,3]
Questions About Tumor Location and Surgical Safety
Ask:
- Where exactly is the tumor located?
- Is it near speech, memory, movement, vision, or brainstem areas?
- Can the tumor be removed safely?
- What does “maximal safe resection” mean in my case?
- Is biopsy safer than surgery?
- Will brain mapping, functional MRI, tractography, or awake surgery be needed?
- What neurological problems may improve or worsen after surgery?
- How much tumor is expected to remain after surgery?
These questions help patients understand the balance between removing the tumor and preserving brain function. In brain tumor care, aggressive treatment must still respect speech, mobility, cognition, and independence. [3,4]
Questions About Treatment Options
Ask:
- What is the recommended treatment plan?
- Do I need surgery, radiation therapy, chemotherapy, targeted therapy, Tumor Treating Fields, or observation?
- If radiation is advised, when should it start?
- If chemotherapy is advised, will it be temozolomide, PCV, or another medicine?
- Is vorasidenib relevant if I have a grade 2 IDH-mutant glioma?
- Are clinical trials available for my tumor type?
- What happens if I choose to delay treatment?
- What is the goal of treatment: cure, control, symptom relief, longer survival, or quality of life?
For lower-grade gliomas, treatment may sometimes be staged. For glioblastoma, treatment is usually more urgent and often includes surgery followed by radiation therapy and temozolomide when the patient is fit for this approach. [4,13]
Questions About Prognosis and Recurrence
Ask:
- What factors affect my prognosis?
- How does my age and general health influence outcome?
- How does IDH status affect my case?
- Does MGMT promoter methylation change the expected response to temozolomide?
- What is the risk of recurrence?
- How often should MRI follow-up be done?
- What symptoms may suggest progression or recurrence?
- If the tumor comes back, what options may still be available?
Survival estimates should be discussed carefully. A statistic cannot fully predict one patient’s outcome. Prognosis depends on tumor biology, treatment response, neurological function, molecular markers, and recurrence pattern. [3,4]
Questions About Side Effects and Quality of Life
Ask:
- What side effects should I expect from surgery?
- What side effects may occur during radiation therapy?
- What side effects are common with temozolomide or other chemotherapy?
- Will I need steroids, and what are their side effects?
- Do I need anti-seizure medicines?
- Can I work, travel, exercise, or continue normal activities?
- Should I meet a physiotherapist, speech therapist, dietitian, psychologist, or palliative care specialist?
- What symptoms should make us go to the hospital immediately?
These questions help the family prepare practically. Glioma care is not only tumor treatment; it is also function, comfort, safety, dignity, and daily living. [4]
Questions About Ayurvedic and Integrative Support
Ask both the oncology team and Ayurvedic physician:
- Can I safely take Ayurvedic medicines with my current treatment?
- Are there any herbs or supplements I should avoid during chemotherapy or radiation?
- Could any formulation interact with steroids, anti-seizure medicines, blood thinners, or other drugs?
- Should herbal medicines be paused before surgery?
- Can supportive care help with appetite, sleep, digestion, fatigue, mood, or recovery?
- How will liver function, kidney function, blood counts, and treatment side effects be monitored?
- Who will coordinate care if symptoms worsen?
Patients should always inform their cancer care team about herbs, supplements, vitamins, or special diets. Some products may interact with cancer treatment or other medicines, so supervision is essential. [15,18]
Questions for Family and Caregivers
Caregivers should also ask:
- What changes in behavior or consciousness should we watch for?
- How should we respond if seizures occur?
- What precautions are needed at home to prevent falls?
- What diet and hydration guidance should we follow?
- How do we manage sleep disturbance, confusion, irritability, or steroid-related mood changes?
- Who should we contact after hours in an emergency?
- How can we support the patient without creating fear or pressure?
Glioma and glioblastoma affect the whole family. A prepared caregiver can reduce panic, improve safety, and help the patient follow treatment with more confidence.
Practical Clinical Message
The best consultation is not the one where the patient simply receives instructions. It is the one where the patient and family understand the diagnosis, treatment goal, risks, alternatives, and supportive care plan. Before deciding on treatment or Ayurvedic support, ask for clarity on tumor type, grade, molecular markers, surgical safety, treatment sequence, recurrence monitoring, and drug interaction risks.
Good questions create better decisions. In glioma and glioblastoma care, informed decisions protect both survival and quality of life. [2,3,4,13,15,18]
FAQs
Are glioma and glioblastoma the same?
No. Glioma is a broad group of brain and spinal cord tumors. Glioblastoma is one specific type of glioma and is the most aggressive form.
Is glioblastoma a type of glioma?
Yes. Glioblastoma is a type of glioma. All glioblastomas are gliomas, but not all gliomas are glioblastomas.
Is glioblastoma always grade four?
Yes. Glioblastoma is always classified as CNS WHO grade four. A low-grade tumor should not be called glioblastoma.
Which is more serious, glioma or glioblastoma?
Glioblastoma is usually more serious because it grows faster, spreads into nearby brain tissue, and has a higher risk of recurrence. Glioma can be low-grade or high-grade, so seriousness depends on the exact type and grade.
Can a low-grade glioma become aggressive?
Yes. Some low-grade gliomas can grow, recur, or transform into higher-grade tumors over time. Regular MRI follow-up and molecular testing are important.
What is the main difference between glioma and glioblastoma?
Glioma is the larger tumor category. Glioblastoma is a specific grade four glioma with rapid growth, deep tissue infiltration, and a more difficult prognosis.
How is glioblastoma diagnosed?
Glioblastoma is diagnosed using MRI, biopsy or surgical tissue examination, histopathology, and molecular testing. Important markers include IDH status, MGMT promoter methylation, EGFR amplification, and TERT promoter mutation.
Why is IDH status important in glioma?
IDH status helps doctors classify gliomas more accurately and estimate prognosis. In adults, glioblastoma is generally defined as glioblastoma, IDH-wildtype, CNS WHO grade four.
What is MGMT promoter methylation?
MGMT promoter methylation is a molecular marker that may help predict response to temozolomide chemotherapy in glioblastoma. Patients with MGMT-methylated tumors may respond better, though outcomes still vary.
Is glioblastoma curable?
Glioblastoma is very difficult to cure because it grows quickly and spreads microscopically into surrounding brain tissue. Treatment may control the tumor, reduce symptoms, extend survival, and support quality of life.
What is the survival rate of glioblastoma?
Glioblastoma survival varies by age, tumor location, surgery result, MGMT status, treatment response, and overall health. Many patients are quoted a median survival of around twelve to eighteen months with standard treatment, but some live longer.
Can Ayurveda help in glioma or glioblastoma?
Ayurveda supports strength, digestion, sleep, appetite, fatigue, mood, recovery, and quality of life when supervised by a qualified physician. It should not replace MRI diagnosis, surgery, radiation, chemotherapy, steroids, anti-seizure medicines, or emergency care.
Can Ayurvedic medicine be taken during chemotherapy or radiation?
Only with medical supervision. Some herbs, supplements, vitamins, or Rasayana formulations may interact with chemotherapy, radiation, steroids, anti-seizure medicines, blood thinners, or other drugs.
What symptoms need urgent medical attention?
A first-time seizure, repeated seizures, sudden weakness, speech difficulty, severe headache, vomiting, confusion, reduced consciousness, vision loss, severe imbalance, or rapid personality change needs urgent medical evaluation.
What should patients ask after a glioma diagnosis?
Patients should ask the exact tumor type, WHO grade, IDH status, 1p/19q codeletion, MGMT status, surgical options, treatment plan, recurrence risk, and MRI follow-up schedule.
Reference
- Louis, D. N., Perry, A., Wesseling, P., et al. (2021). The 2021 WHO Classification of Tumors of the Central Nervous System: A summary. Neuro-Oncology, 23(8), 1231–1251. https://pubmed.ncbi.nlm.nih.gov/34185076/
Brief: Use for WHO CNS 2021 classification, glioma grading, IDH status, molecular diagnosis, and the updated meaning of glioblastoma. - Weller, M., van den Bent, M., Preusser, M., et al. (2021). EANO guidelines on the diagnosis and treatment of diffuse gliomas of adulthood. Nature Reviews Clinical Oncology, 18, 170–186. https://pubmed.ncbi.nlm.nih.gov/33293629/
Brief: Use for adult diffuse glioma diagnosis, MRI, molecular testing, treatment planning, follow-up, and recurrence management. - National Cancer Institute. (2025). Central Nervous System Tumors Treatment (PDQ®): Health Professional Version. https://www.cancer.gov/types/brain/hp/adult-brain-treatment-pdq
Brief: Use for standard treatment of gliomas and glioblastoma, including surgery, radiation, chemotherapy, surveillance, recurrence, and clinical decision-making. (Cancer.gov) - National Cancer Institute. (2024). Adult Central Nervous System Tumors Treatment (PDQ®): Patient Version. https://www.cancer.gov/types/brain/patient/adult-brain-treatment-pdq
Brief: Use for patient-friendly explanations of brain tumor symptoms, diagnosis, and treatment options. - Mayo Clinic. (2026). Glioblastoma survival rates. https://www.mayoclinic.org/diseases-conditions/glioblastoma/survival-rates/gnc-20596050
Brief: Use for glioblastoma survival discussion, including median survival, age factors, treatment response, MGMT status, and realistic patient counselling. (Mayo Clinic) - Cleveland Clinic. (2025). Glioma: What it is, causes, symptoms, treatment and prognosis. https://my.clevelandclinic.org/health/diseases/21969-glioma
Brief: Use for simple explanations of glioma, glioblastoma, tumor grades, symptoms, and prognosis for general readers. - Stupp, R., Mason, W. P., van den Bent, M. J., et al. (2005). Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. New England Journal of Medicine, 352(10), 987–996. https://pubmed.ncbi.nlm.nih.gov/15758009/
Brief: Landmark study supporting the modern glioblastoma treatment standard of radiation plus temozolomide. (PubMed) - Hegi, M. E., Diserens, A. C., Gorlia, T., et al. (2005). MGMT gene silencing and benefit from temozolomide in glioblastoma. New England Journal of Medicine, 352(10), 997–1003. https://pubmed.ncbi.nlm.nih.gov/15758010/
Brief: Use for MGMT promoter methylation and why some glioblastoma patients respond better to temozolomide. (PubMed) - Eckel-Passow, J. E., Lachance, D. H., Molinaro, A. M., et al. (2015). Glioma groups based on 1p/19q, IDH, and TERT promoter mutations in tumors. New England Journal of Medicine, 372(26), 2499–2508. https://www.nejm.org/doi/full/10.1056/NEJMoa1407279
Brief: Use for molecular classification, especially IDH mutation, 1p/19q codeletion, and TERT promoter mutation. - Stupp, R., Taillibert, S., Kanner, A. A., et al. (2017). Effect of Tumor-Treating Fields plus maintenance temozolomide vs maintenance temozolomide alone on survival in patients with glioblastoma. JAMA, 318(23), 2306–2316. https://pubmed.ncbi.nlm.nih.gov/29260225/
Brief: Use for Tumor Treating Fields as an additional glioblastoma treatment option in selected patients. (PubMed) - U.S. Food and Drug Administration. (2024). FDA approves vorasidenib for grade 2 astrocytoma or oligodendroglioma with a susceptible IDH1 or IDH2 mutation. https://www.fda.gov/drugs/resources-information-approved-drugs/fda-approves-vorasidenib-grade-2-astrocytoma-or-oligodendroglioma-susceptible-idh1-or-idh2-mutation
Brief: Use for the latest targeted therapy update in selected grade 2 IDH-mutant gliomas after surgery. (U.S. Food and Drug Administration) - Blakeley, J., Mohile, N. A., Messersmith, H., et al. (2025). Therapy for diffuse astrocytic and oligodendroglial tumors in adults: ASCO-SNO guideline rapid recommendation update. Neuro-Oncology, 27(6), 1412–1418. https://pubmed.ncbi.nlm.nih.gov/40300117/
Brief: Use for updated ASCO-SNO treatment guidance, especially the role of vorasidenib and treatment deferral in selected IDH-mutant grade 2 gliomas. (PubMed) - Price, M., Ballard, C., Benedict, A., et al. (2025). CBTRUS Statistical Report: Primary brain and other central nervous system tumors diagnosed in the United States in 2018–2022. Neuro-Oncology, 27(Supplement 4), iv1–iv105. https://academic.oup.com/neuro-oncology/article/27/Supplement_4/iv1/8285946
Brief: Use for brain tumor incidence, mortality, survival statistics, and population-level context. (OUP Academic) - National Cancer Institute. (2024). Complementary and alternative medicine. https://www.cancer.gov/about-cancer/treatment/cam
Brief: Use for safety wording about complementary therapies, herbs, supplements, and the need to inform the oncology team. - National Center for Complementary and Integrative Health. (n.d.). Cancer and complementary health approaches: What you need to know. https://www.nccih.nih.gov/health/cancer-and-complementary-health-approaches-what-you-need-to-know
Brief: Use for integrative oncology safety, symptom-support framing, and caution around unproven cancer claims. (NCCIH) - Memorial Sloan Kettering Cancer Center. (n.d.). Herbs, botanicals and other products: FAQs. https://www.mskcc.org/cancer-care/diagnosis-treatment/symptom-management/integrative-medicine/herbs/herbs-botanicals-other-products-faqs
Brief: Use for herb-drug interaction warnings, especially during chemotherapy, radiation, anti-seizure therapy, or steroid use. (mskcc.org) - Newton, H. B. (2024). Indian Ayurvedic medicine: Overview and application to brain cancer. Journal of Ayurveda and Integrative Medicine, 15, 101013. https://pubmed.ncbi.nlm.nih.gov/39181067/
Brief: Use for the Ayurvedic brain cancer section, especially when discussing herbs and integrative possibilities. Frame carefully as supportive and research-oriented, not as standalone proof of cure. - Panacea Ayur. (2026). Gliomas: Symptoms, diagnosis, and Ayurvedic cure for brain tumors. https://panaceayur.com/disease-cure-oncology-brain-gliomas-ayurvedic-cure/
Brief: Use as the internal pillar reference for Ayurvedic glioma care, Rasayana support, diagnosis awareness, safety, and patient conversion pathway.