- Hidden Phase Before Diagnosis
- What Is Thalassemia
- Epidemiology (Study of disease distribution and patterns in populations)
- Causes and Risk Factors
- Early Symptoms
- Conditions That May Be Mistaken for Thalassemia
- Advanced Symptoms
- Disease Progression and Staging
- Classification of Thalassemia
- Pathophysiology of Thalassemia
- Clinical Manifestations of Thalassemia
- Diagnostic Approach to Thalassemia
- Modern Medical Treatment of Thalassemia
- Complications and Long-Term Outcomes
- Ayurvedic Perspective
- Ayurvedic Treatment Principles
- Ayurvedic Management (Step-by-Step Clinical Protocol)
- Herbs and Natural Interventions
- Classical Formulation (Avaleha / Rasayana) Medicine
- Modern Scientific Research
- Global Medical Guidance
- Diet and Lifestyle
- Is There a Better Way to Approach Recovery
- Why This Approach Is Different
- What Should You Do Next
- Study Case Real-World Patient Outcomes and Clinical Evidence
- FAQs
- Preventive Strategies and Genetic Counseling
- References
Silent Progression Before Diagnosis
Thalassemia often progresses silently before diagnosis. Many patients live with persistent fatigue, weakness, and reduced stamina for years without a clear explanation. In early stages, the body compensates for defective hemoglobin production, masking the severity of the condition. This silent adaptation delays clinical suspicion, especially in regions where routine genetic screening is not performed. Studies in hematology literature confirm that mild to moderate forms of thalassemia can remain undetected until physiological stress or worsening anemia reveals the underlying disorder [3][8].
Lifelong Dependency on Medical Support
Patients diagnosed with moderate to severe forms of thalassemia often experience lifelong dependency on medical care. Regular blood transfusions, iron chelation therapy, and frequent hospital visits become essential to maintain survival. Over time, this dependency affects not only physical health but also emotional and social well-being. Clinical guidelines emphasize that transfusion-dependent thalassemia requires continuous intervention, as the body cannot independently maintain adequate hemoglobin levels [2][6].
Misdiagnosis and Delayed Recognition
Misdiagnosis and delay are common in the early stages of thalassemia. Initial symptoms such as fatigue, pallor, and low energy are frequently mistaken for iron deficiency anemia or nutritional deficiencies. As a result, patients may receive inappropriate supplementation rather than proper diagnostic evaluation. Genetic confirmation and specialized testing are often delayed, especially in primary care settings. Research highlights that overlapping clinical features with common anemia contribute significantly to delayed diagnosis and suboptimal early management [11][12].
Gap in Current Treatment Approaches
There is a clear gap in current treatment approaches for thalassemia. While modern medicine provides life-saving interventions such as transfusions and bone marrow transplantation, these strategies primarily focus on management rather than restoration of normal physiological function. Many patients continue to experience fatigue, complications from iron overload, and reduced quality of life despite ongoing treatment. This has led to increasing interest in integrative approaches that aim to address metabolic balance, systemic resilience, and long-term stability beyond symptom control [2][9].
Globally, Thalassemia affects millions, with the highest prevalence in regions like India, Southeast Asia, and the Middle East. According to recent epidemiological estimates, approximately 80–90 million people worldwide are carriers of Thalassemia, while more than 300,000 individuals are born each year with severe forms of the disease (Weatherall, 2018).
Early identification and regular medical intervention play crucial roles in mitigating Thalassemia’s impact on affected individuals, ensuring a better quality of life. Current treatments primarily focus on symptom management through regular blood transfusions, iron chelation therapies, and potentially curative approaches like bone marrow transplantation. Simultaneously, traditional medicine systems such as Ayurveda provide complementary approaches aimed at improving overall health and reducing disease severity through holistic, individualized treatment strategies.
In this comprehensive guide, we will explore the complexities of Thalassemia, integrating modern medical insights with Ayurvedic principles, to offer a holistic understanding that empowers both patients and healthcare providers.
In the early stages of thalassemia, a no-symptom phase often exists where the condition remains clinically silent. The body initially compensates for reduced or defective hemoglobin by increasing bone marrow activity and adjusting oxygen delivery mechanisms. This adaptive response can mask the underlying disorder, allowing the disease to progress unnoticed for extended periods [3][8].
Subtle early signs are frequently ignored or misunderstood. Mild fatigue, pale skin, low stamina, and reduced exercise tolerance are often attributed to lifestyle factors, stress, or minor nutritional deficiencies. Because these symptoms develop gradually and are non-specific, patients rarely seek specialized evaluation at this stage, further contributing to diagnostic delay [11][12].
Normal or borderline laboratory reports can also delay diagnosis. Routine blood tests may show only mild anemia or microcytosis, which can be misinterpreted as iron deficiency. Without advanced investigations such as hemoglobin electrophoresis or genetic testing, the underlying hemoglobinopathy may remain undetected, especially in primary care settings [3][11].
These delay factors significantly increase disease burden over time. Late diagnosis often results in progression to moderate or severe forms, requiring regular transfusions and long-term medical management. As complications such as iron overload and organ stress begin to develop, patients enter a cycle of dependency that could have been better managed with earlier identification and intervention [2][9].
What Is Thalassemia
Thalassemia is a hereditary blood disorder characterized by defective production of hemoglobin, the oxygen-carrying protein inside red blood cells. Hemoglobin is made of alpha and beta globin chains, and genetic mutations affecting these chains lead to reduced or absent synthesis of one component. This imbalance disrupts normal hemoglobin structure, producing fragile and ineffective red blood cells that fail to transport oxygen efficiently throughout the body [1][3].
At a deeper clinical level, thalassemia is not only a condition of low hemoglobin but a disorder of ineffective erythropoiesis. The bone marrow continuously attempts to produce red blood cells, but many of these cells are abnormal and destroyed before entering circulation. This creates a dual burden where the body faces both reduced production of functional cells and increased destruction of defective ones. In response, the bone marrow becomes hyperactive and expands, which in severe cases can alter bone structure and lead to skeletal changes [8][10].
The disease primarily originates in the bone marrow, but its impact extends across multiple organ systems. Chronic anemia forces the heart to increase its workload to maintain oxygen delivery, placing long-term strain on cardiac function. The liver and spleen enlarge as they work to filter abnormal blood cells and compensate for ongoing destruction. In patients requiring regular transfusions, excess iron accumulates in the body, depositing in vital organs such as the heart, liver, and endocrine glands, eventually leading to organ dysfunction if not carefully managed [9][13].
From a systemic perspective, thalassemia creates a state of ongoing physiological stress. Reduced oxygen delivery affects cellular metabolism, energy production, and overall organ performance. Patients commonly experience persistent fatigue, reduced physical capacity, delayed growth, and hormonal imbalances. This reflects that thalassemia is not merely a blood disorder, but a multi-system condition influencing long-term health, development, and quality of life [3][8].
A clinically complete approach therefore requires going beyond hemoglobin correction and addressing the broader biological imbalance, including bone marrow function, organ protection, metabolic stability, and overall physiological resilience.
Epidemiology (Study of disease distribution and patterns in populations)

Thalassemia is one of the most widely distributed genetic blood disorders globally, affecting millions of individuals either as carriers or as patients with clinically significant disease. It is especially prevalent in regions historically referred to as the “thalassemia belt,” which includes South Asia, the Middle East, the Mediterranean region, and parts of Africa. Countries such as India and Pakistan have particularly high carrier frequencies, with estimates suggesting that 3% to 17% of the population may carry thalassemia traits depending on the region and community. This high carrier burden leads to a significant number of affected births each year, making thalassemia a major inherited health challenge in these populations [5][4].
On a global scale, it is estimated that over 300,000 children are born annually with severe hemoglobin disorders, including thalassemia syndromes. The condition contributes substantially to pediatric morbidity and mortality in low- and middle-income countries, where access to regular transfusion therapy, chelation, and advanced treatments such as bone marrow transplantation remains limited. This creates a dual burden where both disease prevalence and treatment gaps coexist, increasing long-term complications and reducing life expectancy in underserved regions [4][3].
In Western countries such as United States, United Kingdom, Canada, and Australia, thalassemia was historically considered rare. However, over recent decades, the epidemiological pattern has shifted significantly due to global migration, interethnic marriages, and increased population diversity. As a result, healthcare systems in these countries are encountering a growing number of patients with both thalassemia minor and major forms. This has led to the expansion of screening programs, genetic counseling services, and specialized hematology care centers to manage the rising demand [3][5].
Regions such as Singapore and Gulf countries including United Arab Emirates, Saudi Arabia, and Qatar are also experiencing increasing incidence rates. This trend is driven by a combination of high carrier prevalence in certain ethnic groups and the presence of diverse expatriate populations. Many of these regions have implemented premarital and antenatal screening programs to reduce disease transmission, yet new cases continue to emerge due to incomplete awareness and variable access to preventive strategies [4][5].
From a public health perspective, thalassemia represents not only a genetic disorder but also a socio-economic challenge. The lifelong need for medical care, including transfusions, iron chelation, and monitoring for complications, places a significant burden on healthcare systems and families. This growing global distribution highlights the urgent need for early detection, preventive screening, and integrative approaches that address both disease management and long-term quality of life [3][2].
Causes and Risk Factors
Genetic Inheritance as the Primary Cause
Thalassemia originates from inherited mutations in the genes responsible for producing alpha or beta globin chains, which are essential components of hemoglobin. When one parent carries the mutation, the individual may remain a carrier with mild or no symptoms. However, when both parents carry defective genes, the risk of developing moderate to severe thalassemia increases significantly. These mutations disrupt normal hemoglobin synthesis, leading to unstable red blood cells and chronic anemia. This genetic basis explains the clustering of thalassemia within families and high-prevalence populations [1][12].
Metabolic Stress and Bone Marrow Burden
A major contributor to disease severity is ongoing metabolic stress caused by ineffective erythropoiesis. The bone marrow continuously attempts to compensate for anemia by increasing red blood cell production, but many of these cells are defective and destroyed prematurely. This creates a cycle of excessive production and destruction, placing a constant energy demand on the body. Over time, this leads to fatigue, organ stress, and enlargement of the liver and spleen as they attempt to manage the excess breakdown of abnormal cells [8][3].
Environmental Factors and Nutritional Influence
Although thalassemia is genetic in origin, environmental factors such as nutritional status play a critical role in symptom expression. Deficiencies in nutrients like folate, vitamin B12, and proteins can further impair red blood cell production and worsen anemia. In many cases, especially in resource-limited settings, poor nutrition accelerates disease progression and increases symptom severity. External stressors such as infections can also weaken the body’s ability to cope with chronic anemia, amplifying the overall disease burden [11][3].
Internal Imbalance, Inflammation, and Oxidative Stress
Chronic inflammation and oxidative stress significantly influence the progression of thalassemia. Unstable hemoglobin leads to increased production of reactive oxygen species, which damage red blood cell membranes and shorten their lifespan. In patients receiving repeated transfusions, excess iron accumulates in the body, further intensifying oxidative damage. This iron overload affects vital organs including the heart, liver, and endocrine system, contributing to long-term complications and reduced quality of life [9][13].
Variation in Disease Expression and Severity
Thalassemia presents in multiple forms, ranging from asymptomatic carrier states to severe transfusion-dependent conditions. This variation depends on the type of genetic mutation, co-inherited conditions, and individual physiological response. Some individuals may live with minimal symptoms, while others require lifelong medical support. Factors such as access to healthcare, early diagnosis, and lifestyle also influence disease progression, making thalassemia a highly variable condition in clinical practice [10][3].
Systemic Interaction of Genetic and Modifiable Factors
While the genetic mutation is the root cause, the overall clinical picture of thalassemia is shaped by the interaction between inherited defects and modifiable factors such as metabolism, nutrition, and oxidative stress. Addressing these contributing elements can influence disease progression, improve quality of life, and reduce complications, even though the underlying genetic mutation remains unchanged.
Early Symptoms

Fatigue and Persistent Weakness
Fatigue is one of the earliest and most consistent clinical features of thalassemia. Patients often experience persistent tiredness, reduced stamina, and easy exhaustion despite adequate rest. This occurs because defective hemoglobin reduces oxygen delivery to tissues, particularly muscles and the brain. As a result, even routine physical or mental activities may feel disproportionately exhausting. In milder forms, this fatigue may remain unnoticed for years, while in more severe cases it becomes evident early in life [3][8].
Pale Skin and Reduced Vitality
Pallor is a key early clinical sign caused by decreased hemoglobin levels and reduced red blood cell mass. Patients may appear visibly pale, especially in the face, palms, and conjunctiva. Along with this, there is a noticeable decline in energy levels, mental alertness, and overall vitality. These changes reflect the body’s inability to maintain adequate oxygenation at the cellular level, leading to a generalized sense of weakness and lethargy [1][3].
Subtle Onset in Mild and Intermediate Forms
In many individuals, particularly those with thalassemia trait or intermedia, symptoms begin gradually and may remain mild for long periods. Patients may only notice decreased exercise tolerance, mild fatigue, or occasional weakness without any severe clinical signs. Because the onset is slow and non-specific, these symptoms are often normalized or attributed to lifestyle factors, delaying proper evaluation and diagnosis [3][10].
Early Growth and Developmental Concerns in Children
In more clinically significant forms, especially in children, early symptoms may include poor weight gain, delayed growth, irritability, and reduced appetite. These signs indicate that chronic anemia is affecting metabolic processes and development. Over time, untreated anemia can interfere with normal growth patterns, making early recognition critical in pediatric cases [3][8].
Occasional Susceptibility to Infections
Some patients, particularly those with moderate to severe anemia, may experience increased susceptibility to infections or slower recovery from illness. This is related to reduced physiological resilience and overall systemic stress rather than a primary immune deficiency. While not always a dominant early symptom, it can become more noticeable as the disease progresses [11][3].
Frequent Misinterpretation as Common Anemia
A major clinical challenge in early thalassemia is misinterpretation. Symptoms such as fatigue, pallor, and low energy closely resemble iron deficiency anemia or general nutritional deficiencies. As a result, many patients are initially treated with iron supplements without further investigation. This delays accurate diagnosis, as definitive identification requires specialized tests such as hemoglobin electrophoresis or genetic analysis. Such diagnostic delays allow the condition to progress silently before appropriate management begins [11][12].
Conditions That May Be Mistaken for Thalassemia

Iron Deficiency Anemia
Iron deficiency anemia is the most common condition confused with thalassemia because both present with microcytic anemia, fatigue, pallor, and low hemoglobin levels. In early stages, routine blood tests may not clearly differentiate between the two, leading to frequent misdiagnosis. Patients are often prescribed iron supplements, which may not improve symptoms in thalassemia. A key distinction is that thalassemia is genetic and does not respond to iron therapy unless a true deficiency coexists. Specialized tests such as hemoglobin electrophoresis are required for accurate differentiation [11][12].
Chronic Fatigue Syndrome
Chronic fatigue syndrome presents with persistent tiredness, reduced stamina, and impaired daily functioning, which can closely resemble the fatigue experienced in thalassemia. Because early thalassemia may not show severe anemia, patients can be misclassified under functional or unexplained fatigue conditions. However, thalassemia has an identifiable hematological basis, whereas chronic fatigue syndrome is a complex condition with multifactorial causes and no single definitive diagnostic marker [3][8].
Vitamin B12 Deficiency
Vitamin B12 deficiency can also present with fatigue, weakness, pallor, and reduced energy levels. In some cases, neurological symptoms such as tingling or cognitive changes may coexist, which can further complicate the clinical picture. While B12 deficiency typically causes macrocytic anemia, overlapping symptoms may still lead to confusion, especially when laboratory findings are borderline or mixed. Proper differentiation requires detailed blood indices and biochemical testing [11][3].
Bone Marrow Disorders
Certain bone marrow disorders, including aplastic anemia and myelodysplastic syndromes, may present with anemia, fatigue, and reduced blood cell production. These conditions can mimic thalassemia in terms of clinical presentation, particularly in more severe cases. However, bone marrow disorders involve failure or dysfunction of marrow production, whereas thalassemia involves defective hemoglobin synthesis despite active marrow. Advanced investigations such as bone marrow studies and genetic testing help distinguish between these conditions [3][8].
Why This Confusion Happens
The primary reason for diagnostic confusion is the overlap of non-specific symptoms such as fatigue, pallor, and low energy, which are common across multiple conditions. In addition, reliance on basic blood tests without detailed analysis can lead to incorrect assumptions. This overlap often delays accurate diagnosis, allowing thalassemia to progress untreated or improperly managed in its early stages. Early recognition and appropriate testing are therefore essential to avoid prolonged misdiagnosis and unnecessary treatments [11][12].
Advanced Symptoms
Severe Anemia and Transfusion Dependency
As thalassemia progresses, anemia becomes more severe and clinically significant. Patients often experience extreme fatigue, breathlessness, dizziness, and reduced functional capacity. At this stage, the body is no longer able to compensate for defective hemoglobin production, and regular blood transfusions become necessary to maintain adequate oxygen levels and survival. This transition marks the shift from a compensated state to transfusion-dependent disease, particularly in beta-thalassemia major [3][6].
Organ Enlargement Due to Compensatory Stress
Chronic destruction of abnormal red blood cells leads to increased workload on the liver and spleen, resulting in hepatosplenomegaly, or enlargement of these organs. The spleen becomes overactive in filtering defective cells, while the liver participates in both blood processing and iron storage. Over time, this enlargement can cause abdominal discomfort, reduced appetite, and further complications such as hypersplenism, which worsens anemia by destroying even healthy blood cells [8][3].
Bone Changes and Skeletal Deformities
In response to chronic anemia, the bone marrow becomes hyperactive in an attempt to produce more red blood cells. This continuous stimulation leads to marrow expansion, particularly in the skull and facial bones. As a result, patients with severe thalassemia may develop characteristic skeletal changes, including facial bone prominence and thinning of bones. These deformities reflect the body’s prolonged effort to compensate for ineffective erythropoiesis [8][10].
Growth Delay and Developmental Impact in Children
In children, long-standing anemia and metabolic stress can significantly affect normal growth and development. Delayed height gain, underweight status, delayed puberty, and hormonal imbalances are commonly observed in untreated or poorly managed cases. These changes are not only due to reduced oxygen supply but also related to chronic illness burden and endocrine dysfunction, especially in patients with iron overload from repeated transfusions [3][9].
Systemic Complications from Iron Overload
With ongoing transfusion therapy, excess iron accumulates in the body because there is no natural mechanism to eliminate it efficiently. This iron overload deposits in vital organs such as the heart, liver, and endocrine glands, leading to complications including cardiac dysfunction, liver disease, diabetes, and hormonal disturbances. These systemic effects represent one of the most serious long-term consequences of advanced thalassemia and require continuous monitoring and management [9][13].
Disease Progression and Staging
Spectrum of Disease Severity
Thalassemia exists as a spectrum rather than a single uniform condition, ranging from asymptomatic carrier states to severe, life-threatening disease. This variation depends on the type of genetic mutation, the number of affected globin genes, and the body’s compensatory capacity. As a result, patients may present with very different clinical courses, even within the same family or population [1][10].
Mild Forms: Thalassemia Minor
In mild forms, often referred to as thalassemia trait or minor, individuals may have little to no symptoms. Hemoglobin levels are slightly reduced, and patients may experience occasional fatigue or mild anemia, but daily functioning is generally unaffected. Many individuals remain undiagnosed unless identified through routine screening or family history evaluation. These cases typically do not require regular medical intervention but are important from a genetic counseling perspective [3][12].
Moderate Forms: Thalassemia Intermedia
Moderate forms, commonly termed thalassemia intermedia, present with more noticeable symptoms such as persistent anemia, fatigue, and reduced physical capacity. Patients may not require regular transfusions initially but may need them during periods of stress, illness, or physiological demand. Over time, complications such as organ enlargement, bone changes, and metabolic disturbances may develop, requiring closer medical monitoring and supportive care [10][3].
Severe Forms: Thalassemia Major
Severe forms, particularly beta-thalassemia major, are characterized by profound anemia that manifests early in life. These patients are unable to maintain adequate hemoglobin levels without regular blood transfusions, often starting in infancy or early childhood. Lifelong transfusion dependency becomes necessary to sustain survival, along with iron chelation therapy to manage iron overload. Without appropriate treatment, severe complications and reduced life expectancy are common [3][6].
Clinical Importance of Staging
The stage or severity of thalassemia plays a critical role in determining treatment strategy, monitoring requirements, and long-term prognosis. Early identification of disease severity allows for timely intervention, prevention of complications, and better quality of life. It also helps guide decisions regarding advanced therapies such as bone marrow transplantation or emerging genetic treatments, making accurate staging an essential component of comprehensive care [2][3].
Classification of Thalassemia
Thalassemia includes a spectrum of inherited blood disorders caused by mutations affecting hemoglobin chain production. It is classified based on whether the alpha or beta chains are deficient, and how many genes are involved.
Alpha Thalassemia arises from deletions in the HBA1 and HBA2 genes on chromosome 16. Since humans inherit two alpha-globin genes from each parent (total of four), the severity depends on how many genes are affected. A single gene deletion results in a silent carrier state—clinically asymptomatic but detectable through genetic testing. Deletion of two genes causes Alpha Thalassemia Trait, leading to mild microcytic anemia. Deletion of three genes leads to Hemoglobin H Disease, presenting with moderate anemia, jaundice, splenomegaly, and occasional bone changes. Complete deletion of all four genes results in Hydrops Fetalis, a fatal condition causing stillbirth due to severe anemia and generalized edema.
Beta Thalassemia is caused by mutations in the HBB gene on chromosome 11, resulting in reduced (β⁺) or absent (β⁰) synthesis of beta-globin chains. Individuals inherit one beta gene from each parent. When only one gene is mutated, the person has Beta Thalassemia Minor (Trait), which presents as mild microcytic anemia, often asymptomatic but with elevated HbA2 levels on electrophoresis. If both beta genes are mutated with mild variants, the result is Beta Thalassemia Intermedia, characterized by moderate anemia that may require occasional transfusions and carries a risk of iron overload from increased absorption. Homozygous severe mutations cause Beta Thalassemia Major (Cooley’s Anemia), leading to severe anemia manifesting in early infancy. These patients are transfusion-dependent and develop bone deformities, hepatosplenomegaly, and iron overload complications without curative intervention.
Other rare types include Delta-Beta Thalassemia, which combines deficiency of delta and beta chains and results in mild to moderate anemia, and E-beta Thalassemia, a compound heterozygous state combining beta-thalassemia and Hemoglobin E mutation, common in Southeast Asia with clinical severity ranging from intermediate to major depending on alleles.
For clarity, the following table summarizes the types of Thalassemia:

A real-life example involves a 5-year-old child from eastern India presenting with pallor, jaundice, and abdominal swelling. Lab tests showed hemoglobin 5.2 g/dL, MCV 62 fL, elevated HbF at 80%, and HbA2 at 6.2% on HPLC. Molecular analysis confirmed homozygous β⁰ mutation. The child was diagnosed with Beta Thalassemia Major and began regular transfusions and iron chelation therapy.
Reference:
Colah, R. B., Gorakshakar, A. C., & Nadkarni, A. H. (2010). Global burden, distribution and prevention of beta-thalassemias and hemoglobin E disorders. Expert Review of Hematology, 3(1), 103–117. https://doi.org/10.1586/ehm.09.74
Pathophysiology of Thalassemia
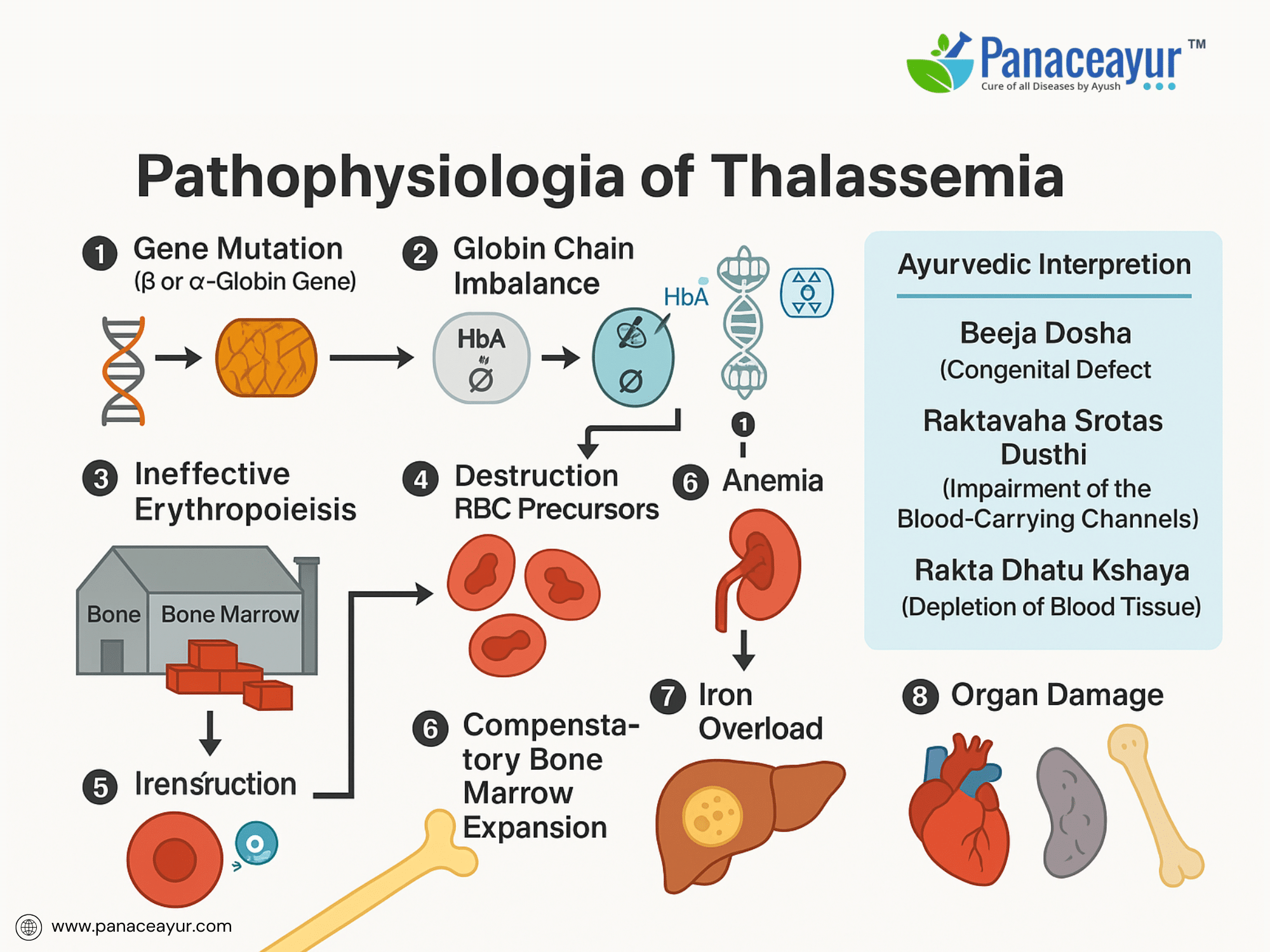
Thalassemia disrupts the body’s ability to produce functional hemoglobin, resulting in ineffective erythropoiesis and chronic anemia. The underlying pathology centers around an imbalance in globin chain synthesis, leading to a cascade of cellular and systemic effects.
In Beta Thalassemia, defective or absent synthesis of beta-globin chains causes an excess of unpaired alpha-globin chains. These free alpha chains are unstable and precipitate within developing erythroblasts in the bone marrow, forming intracellular inclusion bodies. The accumulated inclusions damage the erythroid precursors, leading to apoptosis and premature destruction, a process known as ineffective erythropoiesis. Consequently, despite increased erythropoietic activity, very few functional red blood cells enter the circulation.
In Alpha Thalassemia, depending on the number of gene deletions, there is a progressive deficiency of alpha-globin chains. The imbalance shifts toward excess beta or gamma chains. In cases like Hemoglobin H disease, excess beta chains form unstable tetramers (HbH) that have a high oxygen affinity but are ineffective in delivering oxygen to tissues. In the most severe form, Hydrops Fetalis, gamma chains aggregate into tetramers (Hemoglobin Bart’s), leading to severe hypoxia incompatible with fetal survival.
Both forms lead to chronic anemia, which stimulates increased erythropoietin production. This erythropoietic drive causes expansion of the bone marrow cavities (especially in the skull, maxilla, and long bones), manifesting as skeletal deformities and frontal bossing. The ineffective erythropoiesis also promotes iron absorption from the gastrointestinal tract, compounding iron overload, even in the absence of transfusions.
Another critical feature is extramedullary hematopoiesis, as the body attempts to compensate by producing blood cells outside the bone marrow, notably in the liver and spleen. This leads to hepatosplenomegaly, further complicating management.
On a molecular level, the abnormal red cells that escape the bone marrow exhibit reduced lifespan due to their membrane fragility and oxidative damage. They are prematurely cleared by the spleen, perpetuating the anemia.
A useful analogy is imagining a brick factory where one essential material (beta or alpha chains) is missing. As a result, incomplete or broken bricks accumulate inside the factory, causing machinery breakdown (ineffective erythropoiesis) and poor output (anemia), while the factory expands unsustainably to meet demand (marrow expansion), leading to structural deformities and further strain.
This pathophysiological cascade underscores why treatment in Thalassemia focuses not only on correcting anemia but also on controlling iron overload and preventing organ damage from chronic compensatory mechanisms.
Clinical Manifestations of Thalassemia
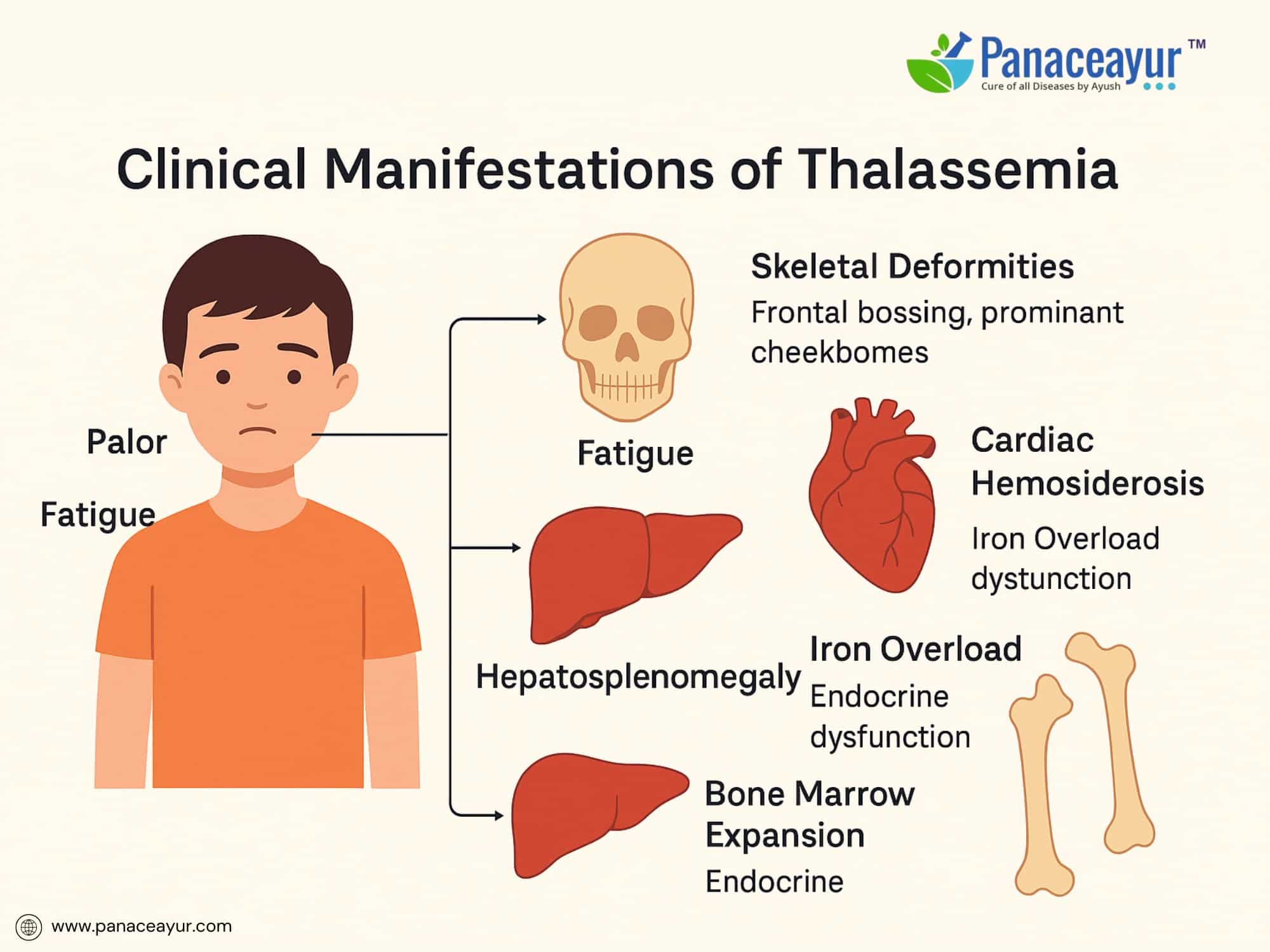
The clinical presentation of Thalassemia varies depending on the type and severity of the underlying genetic mutation. While individuals with minor forms may remain asymptomatic, those with more severe forms exhibit progressive, multisystem manifestations from early life.
Patients with Beta Thalassemia Major typically present between 6 to 12 months of age, once fetal hemoglobin declines and defective adult hemoglobin production becomes evident. Common symptoms include profound fatigue, pallor, and failure to thrive. The hallmark of the disease is severe anemia requiring regular transfusions.
As ineffective erythropoiesis continues, the bone marrow expands in an attempt to compensate, leading to characteristic skeletal changes such as frontal bossing, prominent cheekbones, and maxillary overgrowth causing dental malocclusion. Radiographs may reveal a “hair-on-end” appearance of the skull due to marrow hyperplasia.
Hepatosplenomegaly develops as extramedullary hematopoiesis shifts to the liver and spleen. Splenomegaly can worsen anemia by increasing red cell sequestration, leading to a vicious cycle of anemia and enlargement. Progressive enlargement may cause early satiety and abdominal discomfort.
Iron overload arises both from increased intestinal absorption (due to ineffective erythropoiesis) and recurrent transfusions. Over time, iron accumulates in critical organs including the heart, liver, and endocrine glands. This can result in complications such as cardiac hemosiderosis (leading to heart failure), hepatic fibrosis and cirrhosis, diabetes mellitus (due to pancreatic iron deposition), and hypogonadotropic hypogonadism causing delayed or absent puberty.
Children with Alpha Thalassemia Trait or Beta Thalassemia Minor are usually asymptomatic but may show mild microcytic anemia on routine blood testing. In Hemoglobin H disease, patients may experience mild to moderate anemia, jaundice, and splenomegaly, occasionally requiring transfusions during stress or infections.
A real-life example involves a 7-year-old boy from the Mediterranean region presenting with progressive pallor, irritability, abdominal distension, and frontal bossing. Examination revealed severe anemia (Hb 5.4 g/dL), palpable hepatosplenomegaly, and characteristic facial bone changes. Hemoglobin electrophoresis confirmed Beta Thalassemia Major with elevated HbF and absence of HbA. He was initiated on a regular transfusion protocol and iron chelation therapy to prevent long-term complications.
The psychological impact of Thalassemia must also be acknowledged. Children and adolescents undergoing lifelong transfusions and monitoring may develop emotional distress, social withdrawal, or academic difficulties due to frequent hospitalizations and visible physical differences.
The constellation of anemia, skeletal deformities, organomegaly, and iron-related complications underscores the need for a multidisciplinary approach in managing Thalassemia, aiming not only at correcting anemia but also at preventing the progression of end-organ damage and preserving quality of life.
Diagnostic Approach to Thalassemia
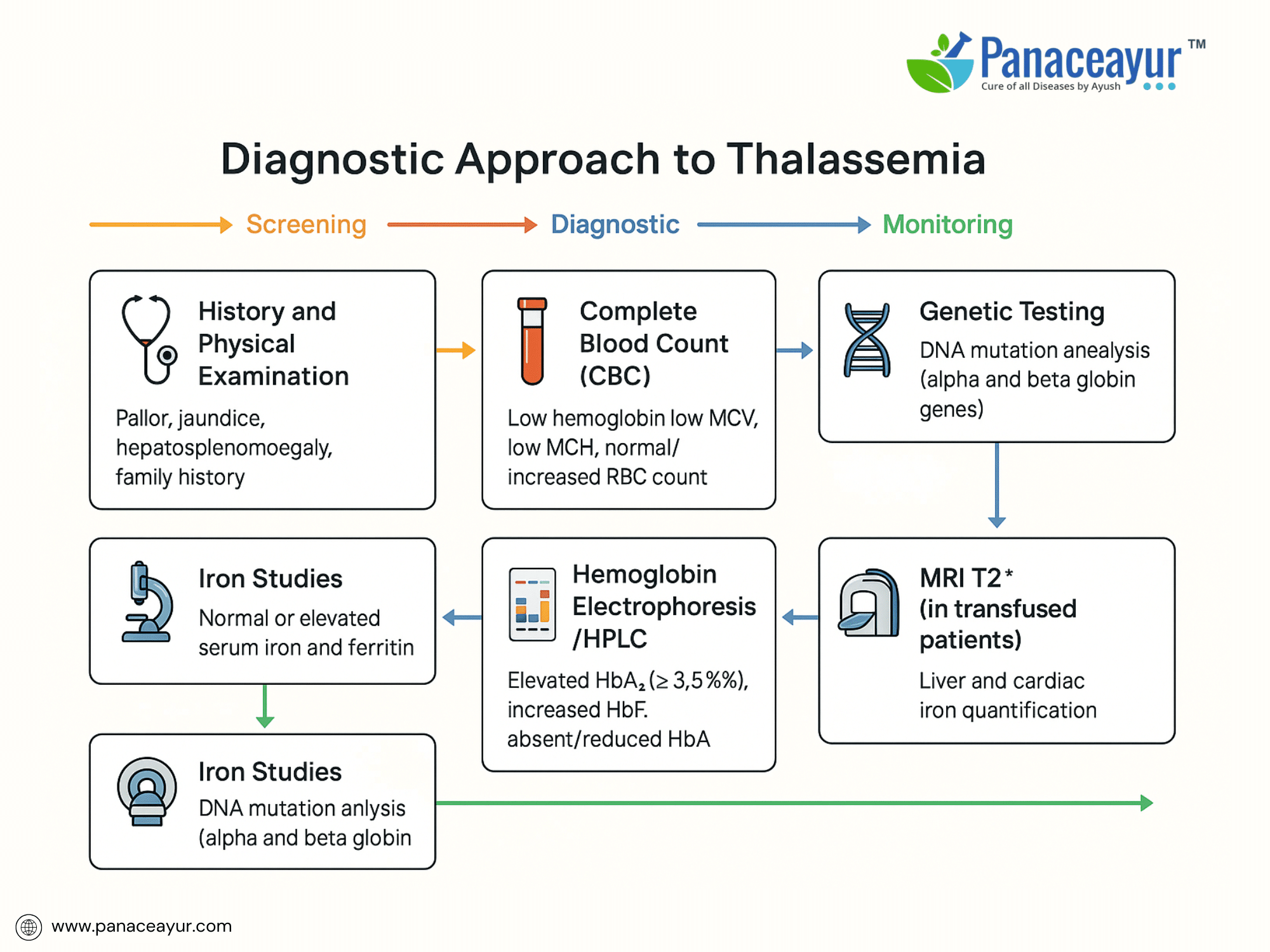
The diagnosis of Thalassemia involves a combination of clinical evaluation, hematological testing, and genetic analysis to confirm the type and severity of the disorder.
Initial suspicion arises in children presenting with chronic anemia, pallor, jaundice, or hepatosplenomegaly, particularly in regions with high Thalassemia prevalence or a family history of the disease.
The diagnostic workup begins with a complete blood count (CBC). This typically shows microcytic hypochromic anemia (low mean corpuscular volume [MCV], low mean corpuscular hemoglobin [MCH]), with a normal or elevated red blood cell count, helping differentiate Thalassemia from iron deficiency anemia. Peripheral blood smear reveals target cells, anisopoikilocytosis, and nucleated red blood cells.
Reticulocyte count is normal or mildly elevated, reflecting ineffective erythropoiesis. Serum iron and ferritin levels are usually normal or increased, further distinguishing it from iron deficiency.
A cornerstone of diagnosis is hemoglobin electrophoresis or high-performance liquid chromatography (HPLC) to quantify hemoglobin fractions. In Beta Thalassemia Minor, HbA2 is elevated (>3.5%), while in Beta Thalassemia Major, HbA is absent or markedly reduced with elevated HbF (>90%). Alpha Thalassemia Trait typically shows normal electrophoresis; diagnosis requires molecular analysis.
When Alpha Thalassemia is suspected, DNA-based tests such as gap-PCR or multiplex ligation-dependent probe amplification (MLPA) identify alpha-globin gene deletions. For Beta Thalassemia, sequencing the HBB gene confirms point mutations or small insertions/deletions.
For prenatal diagnosis, chorionic villus sampling or amniocentesis combined with molecular testing allows early detection in at-risk pregnancies. Carrier screening using HPLC or electrophoresis in parents is critical for genetic counseling.
Additional investigations include:
- Serum ferritin and liver iron quantification (MRI T2)* to monitor iron overload in transfused patients.
- Bone marrow examination (rarely needed) showing erythroid hyperplasia.
- Radiographs may show characteristic skeletal changes (hair-on-end skull appearance).
Differential diagnoses include iron deficiency anemia, sideroblastic anemia, lead poisoning, and anemia of chronic disease. The combination of microcytic anemia, normal iron stores, elevated RBC count, and abnormal hemoglobin fractions strongly supports Thalassemia.
Early and accurate diagnosis not only enables optimal treatment planning but also facilitates family counseling and prevention through carrier detection and prenatal screening.
A real-life case involved a 9-month-old infant with progressive pallor, jaundice, and abdominal swelling. Blood tests showed Hb 6.1 g/dL, MCV 65 fL, and target cells on smear. HPLC revealed HbF 85%, HbA absent, HbA2 4%. Genetic testing confirmed homozygous β⁰ mutation, establishing a diagnosis of Beta Thalassemia Major.
Modern Medical Treatment of Thalassemia
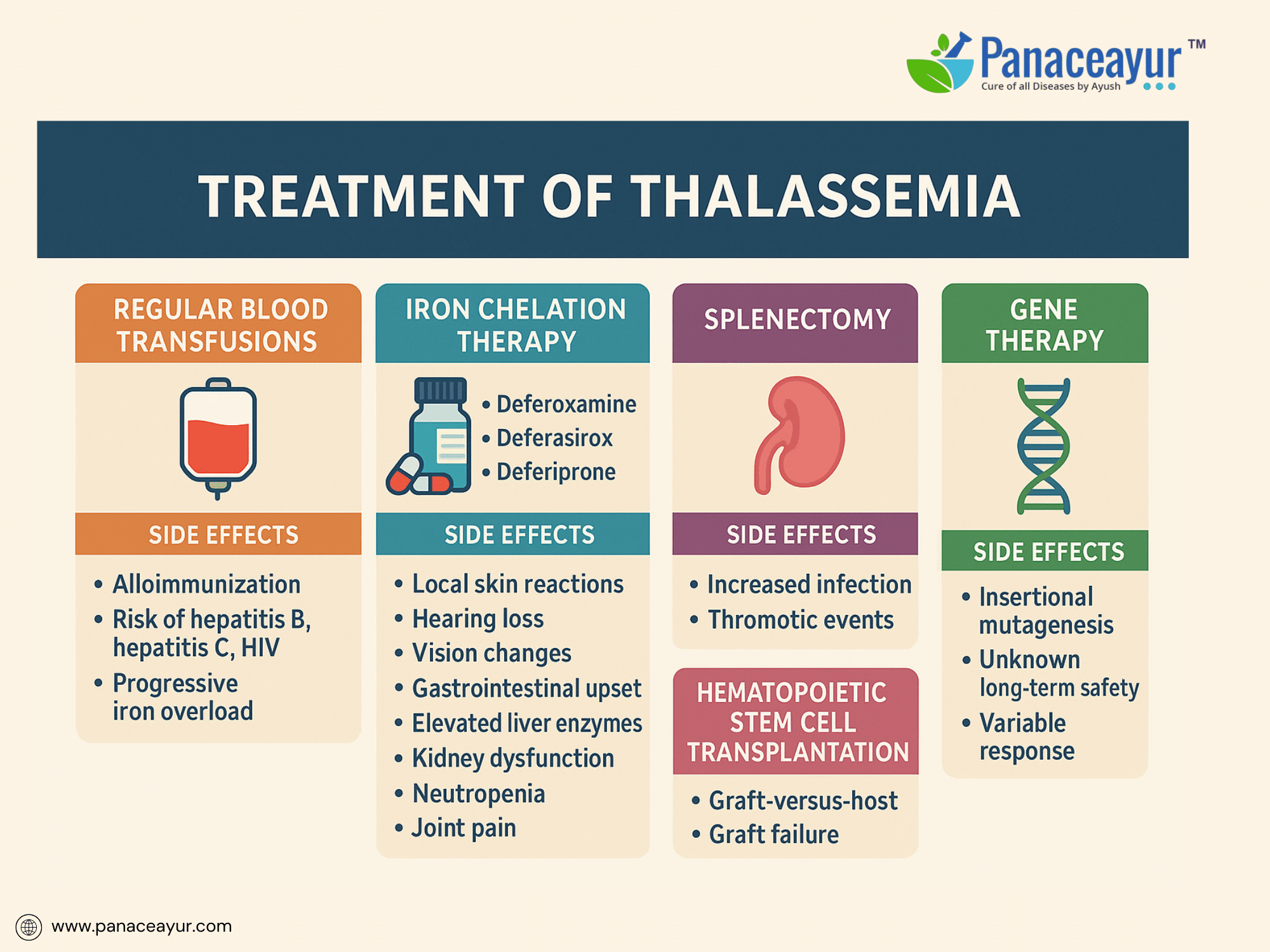
The treatment of Thalassemia is centered on correcting chronic anemia, preventing iron overload, and minimizing disease-related complications. Although curative therapies remain limited, advances in supportive care have dramatically improved both survival and quality of life for individuals living with Beta Thalassemia Major and other severe forms.
Regular blood transfusions remain the cornerstone of treatment, particularly for Beta Thalassemia Major. Lifelong transfusions, initiated in early childhood, aim to maintain pre-transfusion hemoglobin levels between 9 and 10 g/dL. This level effectively suppresses ineffective erythropoiesis, supports normal growth, and prevents skeletal deformities. Despite their lifesaving role, transfusions carry inherent risks. Patients may develop alloimmunization, forming antibodies against donor red cells that complicate future transfusions. Transfusion-transmitted infections, though minimized through rigorous screening, remain a concern for hepatitis B, hepatitis C, and HIV. Each transfusion introduces an iron load of approximately 200–250 mg, progressively contributing to iron accumulation. Early and extended red cell antigen typing is recommended to reduce the risk of alloimmunization, especially in transfusion-dependent patients.
Iron overload, a direct consequence of transfusion dependency, is inevitable without intervention. Excess iron deposits in vital organs such as the liver, heart, and endocrine glands, leading to progressive complications including cardiac failure, liver fibrosis, and endocrine dysfunction. Chelation therapy is essential to remove excess iron and protect against organ damage. Deferoxamine, administered as a subcutaneous or intravenous infusion over 8 to 12 hours for 5 to 7 nights per week, has been a mainstay of chelation. However, it is associated with notable side effects, including local skin reactions, hearing loss, vision impairment, and skeletal abnormalities such as bone dysplasia and growth retardation. The need for prolonged nightly infusions via pump significantly affects patient adherence and quality of life.
Oral chelators offer improved convenience and compliance. Deferasirox is taken once daily on an empty stomach and carries a side effect profile including gastrointestinal disturbances, elevated liver enzymes indicating hepatotoxicity, kidney dysfunction manifesting as proteinuria or rising creatinine, and occasional skin rashes. Regular liver and kidney function monitoring is critical during therapy. Deferiprone, another oral chelator requiring thrice-daily dosing, introduces the risk of neutropenia and potentially life-threatening agranulocytosis, necessitating weekly white blood cell counts. Joint pain, gastrointestinal symptoms, and elevated liver enzymes are additional adverse effects. In some cases, deferiprone is combined with deferoxamine for synergistic iron removal, particularly in patients with cardiac iron overload.
Splenectomy may be considered for patients with massive splenomegaly causing discomfort, hypersplenism resulting in excessive red cell destruction, or rising transfusion needs. However, splenectomy carries its own risks, including heightened susceptibility to life-threatening infections by encapsulated bacteria such as pneumococcus, meningococcus, and Haemophilus influenzae. It also increases the risk of thrombotic events such as portal vein thrombosis and pulmonary embolism. Therefore, splenectomy candidates require preoperative vaccination and, in some cases, lifelong antibiotic prophylaxis to mitigate infection risks.
For select patients, hematopoietic stem cell transplantation (HSCT) offers the potential for a lifelong cure. Ideally performed in childhood using an HLA-matched sibling donor, HSCT can free patients from transfusions and chelation. However, this procedure is not without significant risks, including graft-versus-host disease, graft failure, severe infections related to immunosuppression, and organ toxicity from conditioning regimens. Outcomes are best in younger patients without preexisting iron overload or liver damage, making early referral and preparation critical.
Exciting advancements in gene therapy are offering new hope. Clinical trials using lentiviral vectors to introduce functional beta-globin genes into a patient’s own hematopoietic stem cells have shown promising results, with some individuals achieving transfusion independence. Nonetheless, gene therapy remains experimental, with concerns about insertional mutagenesis and unknown long-term safety. Parallel research in gene editing technologies, such as CRISPR-Cas9, is targeting regulators of fetal hemoglobin to compensate for deficient adult hemoglobin production, though these approaches are still in investigational stages.
In addition to disease-specific treatments, supportive care plays a critical role. Comprehensive monitoring includes regular assessment of growth, pubertal development, endocrine function, and bone health to detect and manage complications such as diabetes, hypothyroidism, hypogonadism, and osteoporosis. Patients benefit from a multidisciplinary care approach involving hematology, endocrinology, cardiology, and psychosocial support services to address the multifaceted impact of chronic disease.
A real-life case involves a 15-year-old adolescent receiving deferasirox chelation therapy who developed rising liver enzymes and proteinuria, necessitating a dose adjustment and close monitoring of hepatic and renal function. Transitioning this patient to adult care required coordinated management across specialties, including cardiology and endocrinology, to monitor cardiac iron load, hormone status, and long-term organ health.
Modern treatment of Thalassemia has transformed a once fatal pediatric disease into a manageable chronic condition. Nevertheless, lifelong adherence to complex treatment regimens and vigilance for therapy-related side effects remain essential to preserve quality of life and prevent long-term complications.
Complications and Long-Term Outcomes
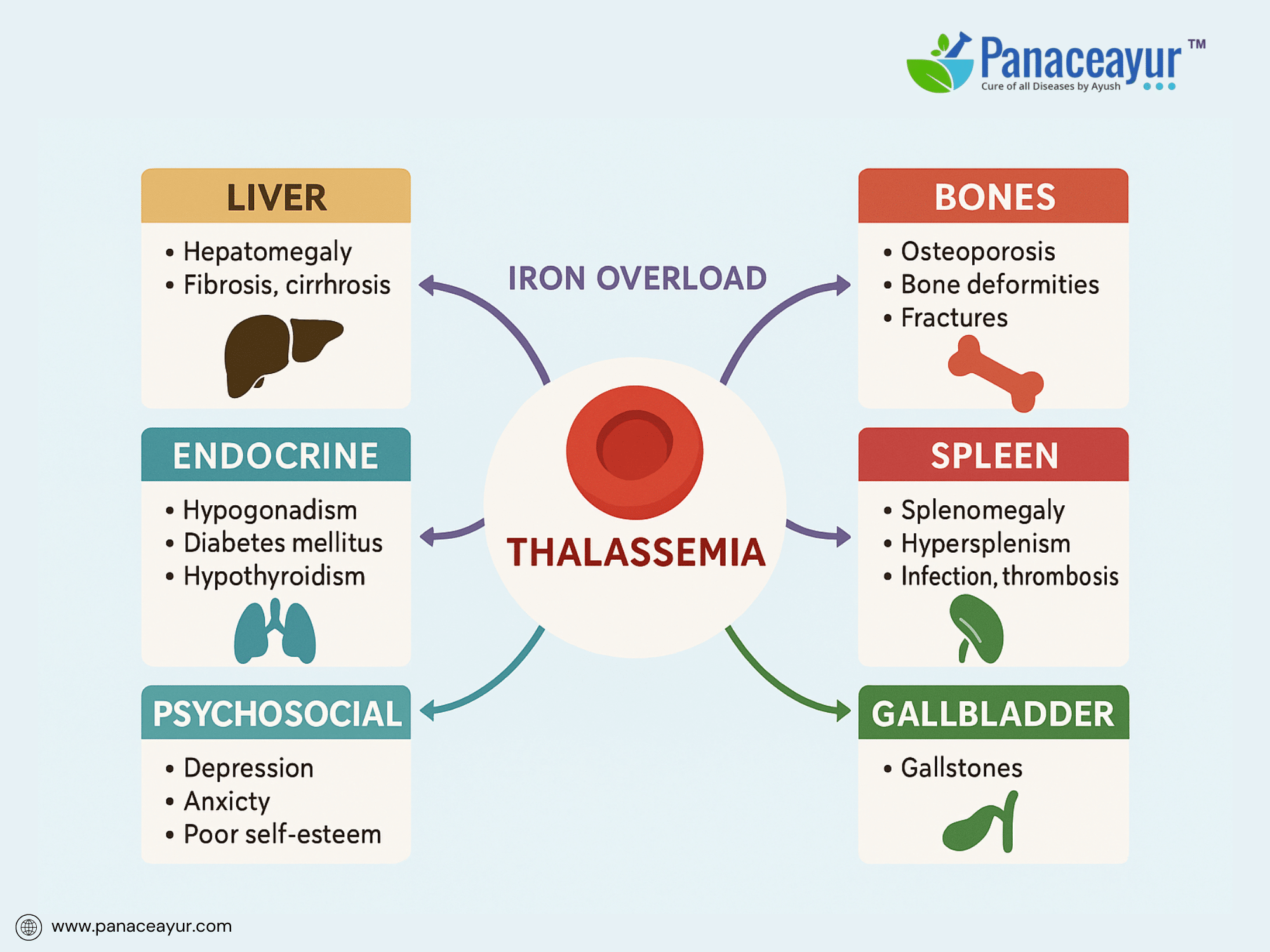
Despite advancements in treatment, Thalassemia remains associated with significant long-term complications due to chronic anemia, iron overload, and treatment side effects. These complications impact multiple organ systems and underscore the need for lifelong monitoring and multidisciplinary care.
Iron overload, whether from transfusions or increased intestinal absorption, is the primary driver of morbidity in transfusion-dependent patients. Excess iron deposits progressively in the liver, leading to hepatomegaly, hepatic fibrosis, and eventually cirrhosis. In the heart, iron deposition causes cardiomyopathy, manifesting as diastolic dysfunction progressing to congestive heart failure, a leading cause of mortality in Thalassemia Major. Endocrine complications result from iron-induced damage to the pituitary, pancreas, and thyroid, leading to hypogonadotropic hypogonadism, delayed puberty, infertility, hypothyroidism, and diabetes mellitus.
Skeletal complications arise from marrow expansion, particularly in undertreated or poorly transfused patients. Chronic ineffective erythropoiesis leads to bone marrow hyperplasia, causing characteristic skeletal deformities such as frontal bossing, maxillary overgrowth, and thinning of cortical bone, predisposing to fractures and osteoporosis. Radiological findings often include “hair-on-end” appearance of skull bones and widened medullary spaces in long bones.
Splenomegaly is a common consequence of extramedullary hematopoiesis and may lead to early satiety, discomfort, and increased red cell sequestration, further exacerbating anemia. Hypersplenism can worsen thrombocytopenia and leukopenia, increasing the risk of infections and bleeding. Post-splenectomy patients face lifelong increased susceptibility to severe infections from encapsulated bacteria, necessitating vaccination and prophylactic measures.
Endocrine complications extend to growth failure and bone mineral density loss. Many adolescents experience delayed or absent secondary sexual development due to hypogonadism. Osteoporosis and osteopenia are prevalent, partly due to iron toxicity in bone and hormonal deficiencies, increasing fracture risk.
Gallstones, often pigmented due to chronic hemolysis, are another complication, leading to cholecystitis or biliary colic. Chronic anemia also increases the risk of leg ulcers and thrombotic events, including pulmonary embolism and portal vein thrombosis, particularly in patients post-splenectomy.
Psychosocial complications are frequently overlooked but critically impact quality of life. Patients may experience anxiety, depression, poor self-esteem related to physical appearance (skeletal changes, short stature), and social stigma. Academic and employment challenges arise due to frequent hospitalizations and treatment burden.
The cumulative burden of these complications can reduce life expectancy and quality of life if not adequately managed. However, with optimal transfusion regimens, effective chelation, early detection of complications, and multidisciplinary care, survival into adulthood with preserved organ function is increasingly achievable. Regular screening using liver and cardiac MRI, bone density assessments, endocrine evaluations, and psychosocial support form the foundation of modern Thalassemia care.
A 22-year-old woman with Beta Thalassemia Major, well-chelated since childhood, presented with secondary amenorrhea and osteoporosis. Endocrine evaluation revealed hypogonadotropic hypogonadism requiring hormone replacement therapy. Her cardiac MRI showed mild myocardial iron overload, prompting chelation intensification. Despite these complications, she maintained normal liver function and preserved fertility options through assisted reproductive counseling.
Long-term outcomes are closely tied to the degree of iron overload control and early identification of organ dysfunction. Evolving therapies, including gene therapy, hold the promise of modifying the natural history of Thalassemia, potentially reducing long-term morbidity in future generation
Temporary Relief Rather Than True Recovery
One of the most important realities patients face is that conventional treatment primarily offers temporary correction rather than lasting recovery. Blood transfusions improve hemoglobin levels for a short period, but symptoms such as fatigue, weakness, and low stamina often return as levels decline. This creates a repetitive cycle where patients feel better briefly and then deteriorate again, leading to continuous dependence on medical intervention without achieving stable, long-term improvement [3][8].
Lifelong Dependency and Psychological Burden
Over time, many patients begin to recognize that their condition requires lifelong management rather than resolution. Regular hospital visits, repeated transfusions, ongoing medications, and constant monitoring become part of daily life. This dependency affects not only physical health but also emotional well-being, career choices, and social life. The realization that treatment must continue indefinitely can be mentally exhausting and leads many patients to search for more sustainable and comprehensive approaches to recovery [2][6].
Root Cause Remains Unaddressed
A critical limitation of current treatment approaches is that they do not correct the underlying defect in hemoglobin production. While advanced options like bone marrow transplantation aim to address this, they are not suitable or accessible for all patients. As a result, the core issue persists, and treatment continues to focus on managing symptoms and preventing complications rather than restoring normal physiological function. This gap becomes more evident as patients seek solutions that go beyond symptom control [2][7].
High Cost and Limited Accessibility Worldwide
The financial burden of thalassemia treatment is significant and ongoing. Lifelong transfusions, iron chelation therapy, diagnostic monitoring, and potential advanced treatments create a substantial economic strain on patients and families. In many regions, access to specialized care is limited, making consistent treatment difficult. Even in developed healthcare systems, advanced therapies such as gene therapy remain expensive and not widely accessible. This imbalance between available medical advancements and real-world access leaves many patients searching for alternative pathways that are both effective and sustainable [4][7].
Why Many Patients Begin to Seek Alternatives
As patients experience these challenges over time, a shift in perspective often occurs. The focus moves from short-term symptom control to long-term quality of life, independence, and stability. Many begin to ask deeper questions about whether it is possible to support the body in a way that reduces dependency, improves energy, and addresses the condition more holistically. This transition marks the point where patients actively explore integrative approaches that aim to complement or go beyond conventional care rather than replace it abruptly.
Ayurvedic Perspective

How Ayurveda Interprets Thalassemia
From an Ayurvedic clinical perspective, thalassemia can be interpreted as a chronic disorder of tissue nourishment and blood formation in which the body is unable to generate strong, stable, life-supporting Rakta Dhatu. The problem is not limited to low hemoglobin alone. It reflects a deeper disturbance in the chain by which food is digested, transformed, absorbed, and converted into progressively refined body tissues. When this chain is weakened, the earliest nutritive essence becomes defective, blood quality suffers, strength declines, and vitality gradually diminishes.
In simple terms, Ayurveda would not look at such a patient and say only that “the blood is low.” It would ask a deeper question: why is the body unable to continuously produce well-nourished, functionally strong blood tissue? This is where the concepts of Rasa Dhatu, Rakta Dhatu, Agni, and Ojas become clinically relevant.
Rasa Dhatu, the First Level of Nourishment
The first tissue formed after digestion is Rasa Dhatu, the circulating nutritive essence that nourishes the entire body and serves as the precursor for the formation of Rakta Dhatu and the remaining tissues. If Rasa is weak, impure, undernourished, or poorly transformed, the next tissue in sequence will also be affected.
Sanskrit
रसाद् रक्तं ततो मांसं मांसान्मेदः प्रजायते ।
मेदसोऽस्थि ततः मज्जा शुक्रं ततः प्रजायते ॥
Charaka Samhita, Chikitsa Sthana, Chapter 15, verse traditionally cited in Dhatu formation sequence
Transliteration
Rasat raktam tato mamsam, mamsan medah prajayate.
Medaso’sthi tatah majja, shukram tatah prajayate.
Translation
From Rasa arises Rakta, from Rakta comes Mamsa, from Mamsa comes Meda, from Meda comes Asthi, then Majja, and thereafter Shukra.
Urdu
رَس سے خون بنتا ہے، پھر جسم کے دوسرے دھات اور بافتیں بنتی ہیں۔
Arabic
من الرسا يتكوَّن الدم، ثم تتكوَّن الأنسجة الأخرى في الجسم.
Clinically, this principle is important because it shows that blood does not arise in isolation. It depends on prior nourishment. In a thalassemia patient, this sequence helps explain why fatigue, weakness, poor stamina, and reduced vitality cannot be addressed only by looking at laboratory numbers. The body’s entire nutritive pathway must be supported if one expects sustained improvement in strength and resilience.
Rakta Dhatu, the Blood Tissue That Sustains Life
Ayurveda gives special importance to Rakta because it is not merely a fluid tissue. It is the carrier of life, vitality, complexion, and functional activity. When Rakta is weak, impure, or insufficient, the patient develops tiredness, pallor, reduced endurance, and systemic weakness. This correlates remarkably well with the lived experience of many thalassemia patients.
Sanskrit
रक्तं जीव इति प्रोक्तं रक्तं धारयति प्रजाः ।
Sushruta Samhita, Sutra Sthana, Chapter 14, verse 44
Transliteration
Raktam jiva iti proktam, raktam dharayati prajah.
Translation
Rakta is called life itself, because it sustains living beings.
Urdu
خون کو زندگی کہا گیا ہے کیونکہ یہی جسم کو قائم رکھتا ہے۔
Arabic
يُقال إن الدم هو الحياة لأنه يحفظ بقاء الإنسان.
For a clinician, the importance of this verse is profound. It tells us that when blood is chronically compromised, the entire organism is placed under stress. Thus, from an Ayurvedic standpoint, a disorder like thalassemia is not a minor blood variation. It is a long-term impairment of one of the body’s most essential sustaining tissues. That is why patients experience not only anemia, but also low vitality, poor growth, reduced exercise tolerance, organ stress, and diminished overall life force.
Agni, the Metabolic Intelligence Behind Tissue Formation
Ayurveda repeatedly states that healthy tissue formation depends on healthy Agni. Agni is not limited to stomach digestion. It includes the body’s broader transformative intelligence by which nutrients are converted into usable tissue. If Agni is weak, irregular, or impaired, even good food cannot be properly transformed into high-quality Rasa and Rakta.
Sanskrit
आयुर्वर्णो बलं स्वास्थ्यं उत्साहोपचयप्रभाः ।
ओजस्तेजोऽग्नयः प्राणाः सर्वं देहेऽग्निनिर्भरम् ॥
Classical Ayurvedic teaching on the centrality of Agni, widely reflected in Charaka Samhita, Chikitsa Sthana, Chapter 15
Transliteration
Ayur varno balam svasthyam utsahopachaya prabhah,
Ojas tejo’gnayah pranah sarvam dehe’gninirbharam.
Translation
Longevity, complexion, strength, health, enthusiasm, nourishment, radiance, Ojas, Tejas, and life itself depend upon Agni.
Urdu
عمر، طاقت، صحت، توانائی، نشوونما اور زندگی کی بنیاد اگنی پر ہے۔
Arabic
العمر والقوة والصحة والحيوية والنمو وحتى الحياة نفسها تعتمد على الأجني.
This principle is central to clinical management. In thalassemia, the challenge is not only that blood is low, but that the process of tissue formation is chronically inefficient. Therefore, a physician trained in Ayurveda would not focus exclusively on supplementation. The first question would be whether the patient’s Agni is capable of receiving, digesting, assimilating, and transforming nourishment effectively. If Agni remains weak, long-term tissue support remains incomplete.
Ojas, the Essence of Vitality and Resistance
Ojas is the most refined essence of all Dhatus. It represents vitality, resistance, endurance, stability, and the body’s power to sustain itself under stress. In chronic disease states, especially those associated with weakness, repeated medical intervention, and long-standing tissue depletion, Ojas is often reduced.
Sanskrit
ओजः सर्वधातूनां सारम् ।
Charaka Samhita, Sutra Sthana, Chapter 17, verse 74
Transliteration
Ojah sarva-dhatunam saram.
Translation
Ojas is the essence of all the body tissues.
Urdu
اوجس تمام دھاتوں کا نچوڑ اور جسمانی قوت کی اصل ہے۔
Arabic
الأوجاس هو خلاصة جميع أنسجة الجسم ومصدر القوة والثبات.
In a thalassemia patient, low Ojas may be reflected clinically as chronic fatigue, poor immunity, low confidence in recovery, reduced resilience, and diminished quality of life. This is why many patients feel that even when treatment keeps them alive, they still do not feel truly strong. Ayurveda recognizes this distinction very clearly. Mere survival is not equal to restored vitality. A person may have ongoing medical support and still remain Ojas-depleted. This insight is especially relevant for patients who continue to feel weak despite conventional treatment.
Classical Ayurvedic Correlation with Pandu and Dhatu Kshaya
While thalassemia as a modern genetic diagnosis is not named in classical texts, its clinical features can be correlated with Ayurvedic descriptions of Pandu, Rakta Kshaya, and Dhatu Kshaya, where pallor, weakness, exertional fatigue, reduced strength, and poor tissue quality are emphasized.
Sanskrit
पाण्डुत्वमल्परक्तत्वं दौर्बल्यं श्रम एव च ।
Classical descriptions of Pandu in Charaka Samhita, Chikitsa Sthana, Chapter 16
Transliteration
Pandutvam alpa-raktatvam daurbalyam shrama eva cha.
Translation
Pallor, deficiency of blood, weakness, and easy exhaustion are features of the disorder.
Urdu
پانڈو میں زردی یا پھیکا پن، خون کی کمی، کمزوری اور جلد تھک جانا شامل ہیں۔
Arabic
من علامات هذا الاضطراب الشحوب، قلة الدم، الضعف، وسرعة التعب.
This correlation matters because it gives the physician a therapeutic direction. Ayurveda would not treat the case as a single laboratory defect. It would assess the quality of nourishment, the strength of digestion and metabolism, the condition of Rakta Dhatu, the presence of depletion, and the state of Ojas. That creates a much broader and more personalized treatment framework.
Why This Perspective Matters Clinically
This Ayurvedic interpretation offers patients something important that many feel is missing in conventional care: a full-body explanation. Conventional medicine rightly addresses transfusion requirements, iron overload, and advanced interventions. However, many patients still ask why they continue to feel tired, weak, dependent, and physiologically fragile. Ayurveda addresses this gap by explaining that blood weakness is only one visible part of a deeper tissue-level disorder.
A professional Ayurvedic approach therefore aims to support the patient at multiple levels:
- It seeks to improve the quality of nourishment entering the tissues.
- It seeks to support Agni so that nutrition can be properly transformed.
- It seeks to strengthen Rakta Dhatu, not only numerically but functionally.
- It seeks to preserve or rebuild Ojas so that the patient feels stronger, more stable, and more resilient over time.
This does not mean ignoring the seriousness of thalassemia. On the contrary, it means approaching it with greater depth. A patient with chronic blood weakness requires more than short-term correction. He or she requires a structured plan that supports metabolism, tissue formation, vitality, and systemic stability in a sustained way.
Why Many Patients Find This Approach Meaningful
Many thalassemia patients eventually realize that managing reports alone is not enough. They want to feel better, function better, and live with more strength and less dependency. That is exactly where Ayurveda becomes meaningful. It offers a framework that explains why the body is weak, why energy remains low, and why deeper restoration must involve nourishment, metabolism, blood tissue, and vitality together.
For Gulf, South Asian, and international patients alike, this perspective often feels more complete because it does not reduce the person to a single test parameter. It sees the disease, but it also sees the patient’s strength, depletion, constitution, digestive capacity, resilience, and long-term recovery potential. That is the true clinical value of the Ayurvedic perspective.
Ayurvedic Treatment Principles

Root Cause Correction at the Level of Beejadushti and Dhatu Formation
From a clinically grounded Ayurvedic perspective, thalassemia is interpreted as a Beejadushtijanya condition, meaning a disorder arising at the level of genetic or cellular blueprint, leading to impaired formation of Rakta Dhatu. Classical Ayurveda explains that when the sequential transformation of tissues is disturbed, the quality of blood becomes compromised, resulting in chronic weakness, pallor, and reduced vitality. Therefore, treatment is not directed only at increasing hemoglobin temporarily but at restoring the physiological pathway through which nourishment is converted into functional blood tissue.
Sanskrit
रसाद् रक्तं ततो मांसं मांसान्मेदः प्रजायते ।
— Charaka Samhita, Chikitsa Sthana 15
Transliteration
Rasād raktam tato māṁsaṁ māṁsān medaḥ prajāyate
Translation
From properly formed nourishment arises blood, followed by other tissues
Urdu
صحیح رَس سے خون بنتا ہے اور پھر جسم کے باقی حصے بنتے ہیں
Arabic
من التغذية السليمة يتكوَّن الدم ثم تتكوَّن باقي أنسجة الجسم
This principle establishes a critical clinical insight that sustainable improvement in blood quality depends on correcting the foundational nutritive processes rather than relying solely on external correction.
Agni Restoration as the Central Metabolic Strategy
Agni represents the body’s metabolic intelligence responsible for digestion, absorption, and transformation of nutrients into tissues. In chronic disorders like thalassemia, impaired Agni leads to inefficient conversion of nutrition into usable biological substrates, resulting in poor Dhatu quality and persistent fatigue despite adequate intake or treatment.
Sanskrit
अग्निर्देहे सर्वकर्मणां मूलम्।
— Charaka Samhita, Sutra Sthana 12/1
Transliteration
Agnir dehe sarva-karmaṇāṁ mūlam
Translation
Agni is the root of all physiological functions
Urdu
اگنی جسم کے تمام افعال کی بنیاد ہے
Arabic
الأجني هو أساس جميع وظائف الجسم
From a clinical standpoint, restoring Agni improves digestion, enhances nutrient assimilation, and supports proper tissue formation. This explains why patients who only rely on external supplementation may not achieve lasting improvement unless metabolic efficiency is addressed.
Ama Reduction and Functional Clearance of Srotas
Ama refers to improperly processed metabolic byproducts that accumulate due to impaired digestion and metabolism. In chronic conditions, Ama obstructs microchannels or Srotas, reducing the delivery of nutrients to tissues and impairing physiological function. In thalassemia, long-term metabolic inefficiency and systemic stress create a similar state of internal obstruction.
Sanskrit
आमं दोषैः समायुक्तं रोगाणां मूलमुच्यते।
Transliteration
Āmaṁ doṣaiḥ samāyuktaṁ rogāṇāṁ mūlam uchyate
Translation
Ama, combined with disturbed doshas, is the root cause of disease
Urdu
اما اور خراب دوش مل کر بیماری کی جڑ بنتے ہیں
Arabic
الأما مع اختلال الدوشا يُعد أصل الأمراض
Clinically, reducing Ama improves microcirculation, enhances tissue responsiveness, and increases the effectiveness of all therapeutic interventions. It allows the body to utilize nutrition and medicine more efficiently.
Ojas Enhancement for Long-Term Stability and Vitality
Ojas is considered the final essence of all Dhatus and represents vitality, strength, immunity, and resilience. In chronic conditions such as thalassemia, prolonged physiological stress, repeated interventions, and tissue depletion lead to Ojas Kshaya, or reduction in vital strength.
Sanskrit
ओजः सर्वधातूनां सारः।
— Charaka Samhita, Sutra Sthana 17/74
Transliteration
Ojaḥ sarva-dhātūnāṁ sāraḥ
Translation
Ojas is the essence of all body tissues
Urdu
اوجس جسم کی طاقت اور مدافعت کی اصل ہے
Arabic
الأوجاس هو خلاصة الأنسجة ومصدر القوة والمناعة
Restoration of Ojas leads to improved energy, better recovery, enhanced endurance, and overall physiological stability. This is particularly important for patients who continue to experience weakness despite ongoing conventional treatment.
Integrated Clinical Perspective
When these principles are applied together, the treatment approach shifts from symptomatic management to systemic correction. The focus is on improving tissue quality, restoring metabolic efficiency, clearing physiological obstruction, and rebuilding vitality. This creates a more comprehensive therapeutic model where the patient is supported not only at the level of blood parameters but also in terms of energy, resilience, and long-term stability.
Ayurvedic Management (Step-by-Step Clinical Protocol)

Step 1: Comprehensive Clinical Assessment and Individualization
Management begins with a detailed evaluation of the patient’s condition, including Prakriti, Dosha imbalance, Dhatu status, Agni strength, and overall disease severity. In thalassemia, patients often notice that symptoms vary significantly despite similar diagnoses. Some feel extreme fatigue, others struggle with digestion, while some face recurrent weakness even after transfusions. This variation occurs because the internal state of the body differs in each individual.
A personalized assessment helps identify why the body is not responding optimally. It allows the treatment to be tailored according to the patient’s strength, metabolic capacity, and disease burden, rather than applying a uniform approach. This forms the foundation for meaningful and sustained improvement.
Step 2: Correction of Agni and Digestive Stabilization
A common concern among patients is that despite eating well or receiving treatment, energy levels remain low. Ayurveda explains this through impaired Agni, where the body is unable to properly convert food and nutrients into usable energy and tissue.
Sanskrit
अग्निर्देहे सर्वकर्मणां मूलम्।
— Charaka Samhita, Sutra Sthana 12/1
Transliteration
Agnir dehe sarva-karmaṇāṁ mūlam
Translation
Agni is the root of all physiological functions
Urdu
اگنی جسم کے تمام افعال کی بنیاد ہے
Arabic
الأجني هو أساس جميع وظائف الجسم
Restoring Agni improves digestion, enhances nutrient absorption, and supports the body’s ability to build strength. Clinically, patients begin to notice better appetite, improved digestion, and a gradual increase in energy levels.
Step 3: Reduction of Ama and Clearance of Srotas
Many patients experience a persistent sense of heaviness, low responsiveness, or sluggish recovery even after treatment. This is often due to the accumulation of Ama, which interferes with normal physiological processes and blocks proper nourishment at the tissue level.
Sanskrit
आमं दोषैः समायुक्तं रोगाणां मूलमुच्यते।
Transliteration
Āmaṁ doṣaiḥ samāyuktaṁ rogāṇāṁ mūlam uchyate
Translation
Ama combined with disturbed doshas is the root cause of disease
Urdu
اما اور خراب دوش مل کر بیماری کی جڑ بنتے ہیں
Arabic
الأما مع اختلال الدوشا يُعد أصل الأمراض
Reducing Ama improves circulation and allows nutrients and medicines to reach tissues more effectively. Patients often report feeling lighter, more responsive, and better able to recover from daily fatigue.
Step 4: Dhatu Nourishment with Focus on Rasa and Rakta
A major frustration for patients is that while treatments may maintain hemoglobin levels, they do not always restore strength or vitality. Ayurveda addresses this by focusing on improving the quality of Rasa and Rakta Dhatu, which are responsible for nourishment and blood formation.
Sanskrit
रसाद् रक्तं ततो मांसं मांसान्मेदः प्रजायते ।
— Charaka Samhita, Chikitsa Sthana 15
Transliteration
Rasād raktam tato māṁsaṁ māṁsān medaḥ prajāyate
Translation
From nourishment arises blood and subsequent tissues
Urdu
رَس سے خون بنتا ہے اور پھر جسم کے باقی حصے بنتے ہیں
Arabic
من الرسا يتكوَّن الدم ثم تتكوَّن باقي الأنسجة
At this stage, patients begin to notice more meaningful changes such as improved stamina, reduced fatigue, and better overall functional capacity. The focus shifts from temporary correction to gradual strengthening.
Step 5: Ojas Restoration and Strength Enhancement
Even with ongoing treatment, many patients feel mentally and physically exhausted. This reflects depletion of Ojas, which represents the body’s core vitality and resilience.
Sanskrit
ओजः सर्वधातूनां सारः।
— Charaka Samhita, Sutra Sthana 17/74
Transliteration
Ojaḥ sarva-dhātūnāṁ sāraḥ
Translation
Ojas is the essence of all body tissues
Urdu
اوجس جسم کی طاقت اور مدافعت کی اصل ہے
Arabic
الأوجاس هو خلاصة الأنسجة ومصدر القوة والمناعة
Restoring Ojas improves endurance, enhances recovery, and supports overall stability. Patients often report better mental clarity, improved strength, and a greater sense of well-being during this phase.
Step 6: Rasayana Therapy for Long-Term Stability
After initial improvement, Rasayana therapy is introduced to maintain and strengthen the gains achieved. This phase focuses on long-term tissue support, improved metabolic efficiency, and prevention of further decline.
Patients often observe that their condition becomes more stable, with fewer fluctuations in energy and improved day-to-day functioning.
Step 7: Panchakarma (Optional and Case-Dependent)
In selected cases, Panchakarma may be recommended to enhance detoxification and systemic balance. It is not required for every patient and is advised only when the body is strong enough to tolerate it.
Step 8: Continuous Monitoring and Integrative Care
Regular monitoring of hemoglobin levels, iron status, and organ function remains essential. Ayurvedic management is applied alongside necessary medical care to ensure safety and optimize outcomes.
Clinical Summary
This approach focuses on correcting the internal processes responsible for weakness, rather than only managing external symptoms. Patients often begin to experience improved energy, better functional capacity, and enhanced resilience over time. The goal is not only to maintain stability but to support the body in functioning more effectively, leading to a better quality of life
Herbs and Natural Interventions

Ashwagandha (Withania somnifera): Support for Strength, Hematopoiesis, and Energy
Ashwagandha is one of the most clinically relevant Rasayana herbs used in conditions characterized by chronic weakness, fatigue, and tissue depletion. In thalassemia, patients frequently experience persistent low energy, poor stamina, and slow recovery even after medical support. Ashwagandha helps address this by improving overall strength, supporting bone marrow function, and enhancing the body’s ability to adapt to long-term physiological stress.
Modern research supports its role as an adaptogen with potential hematinic effects, showing improvement in hemoglobin levels, reduction in fatigue, and better overall physiological resilience.
Singh, N., Bhalla, M., de Jager, P., & Gilca, M. (2011). An overview on Ashwagandha.
https://pubmed.ncbi.nlm.nih.gov/22754076
From a patient perspective, this translates into improved daily energy, better endurance, and enhanced ability to function without constant exhaustion.
Guduchi (Tinospora cordifolia): Immunomodulation and Systemic Stability
Guduchi plays a critical role in stabilizing the internal environment of the body. In thalassemia, where ongoing physiological stress, oxidative burden, and repeated medical interventions weaken systemic balance, Guduchi helps regulate immune response and reduce chronic inflammation.
Scientific studies highlight its immunomodulatory and antioxidant effects, which are essential in reducing long-term disease burden and improving overall resilience.
Sharma, U., Bala, M., Kumar, N., et al. (2012). Immunomodulatory active compounds from Tinospora cordifolia.
https://pubmed.ncbi.nlm.nih.gov/22472109
Clinically, patients often feel more stable, experience fewer fluctuations in energy, and show improved tolerance to ongoing treatment stress.
Amalaki (Emblica officinalis): Antioxidant Protection and Blood Support
Amalaki is a cornerstone Rasayana herb known for its potent antioxidant properties. In thalassemia, oxidative stress caused by unstable red blood cells and iron overload contributes significantly to fatigue and tissue damage. Amalaki helps counter this process by protecting cells from oxidative injury and supporting overall metabolic health.
Research confirms its strong antioxidant activity and role in improving hematological and metabolic parameters.
Scartezzini, P., & Speroni, E. (2000). Antioxidant activity of Emblica officinalis.
https://pubmed.ncbi.nlm.nih.gov/16226416
For patients, this often results in better vitality, improved digestion, and support in maintaining blood quality over time.
Shatavari (Asparagus racemosus): Deep Nourishment and Tissue Restoration
Shatavari is a deeply nourishing Rasayana herb used in conditions involving long-term depletion and weakness. In thalassemia, where chronic disease leads to reduced vitality and systemic exhaustion, Shatavari helps rebuild tissue strength and support recovery.
Modern evidence suggests its adaptogenic and restorative properties contribute to improved physiological balance and endurance.
Goyal, R. K., & Singh, J. (2003). Asparagus racemosus—an update.
https://pubmed.ncbi.nlm.nih.gov/14515032
Clinically, patients may experience improved strength, reduced fatigue, and better recovery capacity with sustained use.
Why Single Herbs Are Not Enough in Clinical Practice
While these herbs are powerful individually, thalassemia is a complex, multi-system condition that cannot be effectively managed using single herbs alone. Each patient presents with a unique combination of factors including disease severity, metabolic strength, digestive capacity, iron overload status, organ involvement, and overall vitality.
Using isolated herbs without understanding this complexity often leads to minimal or inconsistent results. This is why classical Ayurveda emphasizes combination therapy rather than single-drug intervention.
Role of Multi-Ingredient Formulations in Real Treatment
In actual clinical practice, Ayurvedic formulations are carefully designed combinations that may include 40 or more ingredients, incorporating multiple herbs and, where appropriate, properly purified mineral preparations. These combinations are not random. Each ingredient is selected to perform a specific function such as improving digestion, enhancing tissue formation, reducing oxidative stress, supporting organ function, and restoring vitality.
The composition of these formulations varies from patient to patient. Factors such as age, severity of thalassemia, frequency of transfusions, digestive strength, and systemic condition determine the exact formulation. This individualized approach ensures that treatment is targeted, balanced, and effective.
Clinical Perspective and Patient Relevance
For many patients, a key concern is that current treatment helps maintain survival but does not fully restore strength or quality of life. Ayurvedic herbal and formulation-based interventions aim to address this gap by supporting how the body functions internally.
The goal is not immediate replacement of existing treatment, but gradual improvement in energy, resilience, and physiological stability. Over time, patients often notice better stamina, improved recovery, and a more consistent sense of strength, which directly impacts their daily life and long-term well-being.
Classical Formulation (Avaleha / Rasayana) Medicine

Medicine name: Physician-Customized Drakshadi Rasayana Avaleha
For international patients, it is important to explain this clearly. This medicine is not a tablet, syrup, or nutritional supplement in the Western sense. It is an Avaleha, which means a slow-cooked medicated herbal paste prepared from multiple herbs, a decoction base, a nutritive medium, and selected supportive substances. In practical terms, it is designed to work on digestion, tissue nourishment, blood support, strength, recovery, and vitality over time.
In thalassemia, many patients say the same thing: “My treatment is continuing, but I still feel weak.” That happens because survival support and deep functional recovery are not the same. A formulation like Drakshadi Rasayana Avaleha is used to support the body at a broader level. It helps improve how food is processed, how tissues are nourished, how fatigue is handled, and how strength is maintained from day to day. It is not presented as a genetic cure. It is used as a supportive, physician-guided Rasayana protocol to improve resilience, appetite, digestion, recovery, and quality of life.
The table below gives the patient a simple clinical snapshot.
| Parameter | Clinical explanation |
|---|---|
| Medicine form | Avaleha, a thick medicated herbal paste |
| Core purpose | To support digestion, nourishment, blood tissue quality, energy, and vitality |
| Who may benefit | Patients with chronic weakness, poor stamina, low appetite, poor recovery, and tissue depletion |
| Dose | 15 grams twice daily |
| Duration | 30 days as an initial supervised protocol |
| How it is taken | Usually with warm water or as advised according to digestion |
| Important note | This is physician-supervised and not meant for self-preparation |
How this medicine works inside the body
From a clinical Ayurvedic perspective, this type of formulation is used in layers. First, it improves Agni, meaning the body’s ability to digest and transform food properly. This matters because many weak patients eat but do not truly convert that food into strength. Second, it supports Rasa Dhatu, the nutritive layer that feeds all tissues. Third, it supports Rakta Dhatu, which is why patients gradually notice improvement in stamina, recovery, and day-to-day energy. Fourth, it reduces internal stress and helps the body cope better with chronic disease burden. Finally, it works as a Rasayana, meaning it is intended to rebuild resilience rather than produce a short-lived stimulant effect.
For that reason, patients should not expect it to work like a painkiller or instant tonic. The goal is steadier improvement, better tolerance to illness, better digestion, less exhaustion, and more stable energy across the day.
Thirty-day herbal composition
The next table shows the raw material used to prepare one 30-day batch. These are preparation weights, not the amount consumed directly. The patient does not eat these herbs separately. They are processed into a final Avaleha, from which the patient takes 15 grams twice daily.
| Group | Ingredient | 30-day raw weight | Why it is included |
|---|---|---|---|
| Primary Rasayana support | Ashwagandha | 100 g | Supports strength, recovery, fatigue resistance |
| Primary Rasayana support | Guduchi | 100 g | Supports resilience, immune balance, systemic stability |
| Primary Rasayana support | Amalaki | 150 g | Supports antioxidant defense, tissue nourishment, vitality |
| Primary Rasayana support | Shatavari | 100 g | Deep nourishing and restorative support |
| Primary Rasayana support | Draksha | 150 g | Helps weakness, low energy, and depleted states |
| Primary Rasayana support | Yashtimadhu | 80 g | Supports tissue healing and overall vitality |
| Rakta and organ support | Punarnava | 80 g | Supports fluid balance and organ stress reduction |
| Rakta and organ support | Bhumyamalaki | 80 g | Supports liver function and metabolic correction |
| Rakta and organ support | Manjistha | 60 g | Supports healthy blood and circulation |
| Rakta and organ support | Daruharidra | 50 g | Supports inflammation control and tissue protection |
| Rakta and organ support | Neem | 40 g | Supports internal cleansing and balance |
| Rakta and organ support | Sariva | 60 g | Supports cooling, blood support, and systemic calm |
| Rakta and organ support | Arjuna | 60 g | Supports cardiovascular endurance and tissue stability |
| Agni and absorption | Pippali | 40 g | Improves absorption and bioavailability |
| Agni and absorption | Shunthi | 40 g | Supports digestion and reduces heaviness |
| Agni and absorption | Maricha | 30 g | Improves uptake of the formulation |
| Agni and absorption | Chitrak | 30 g | Supports metabolic stimulation where needed |
| Agni and absorption | Ajwain | 30 g | Helps bloating, poor digestion, and heaviness |
| Agni and absorption | Jeeraka | 30 g | Supports appetite and digestive comfort |
| Agni and absorption | Dhanyaka | 30 g | Supports digestion and mild detoxifying action |
| Agni and absorption | Musta | 40 g | Helps poor appetite and unstable digestion |
| Strength and Ojas support | Vidarikand | 80 g | Nourishes depleted tissues and supports strength |
| Strength and Ojas support | Gokshura | 60 g | Supports tissue endurance and recovery |
| Strength and Ojas support | Bala | 60 g | Supports physical strength and stability |
| Strength and Ojas support | Atibala | 50 g | Supports convalescence and weakness recovery |
| Strength and Ojas support | Safed Musli | 60 g | Supports long-term vitality and rebuilding |
| Nervous system and recovery | Shankhpushpi | 40 g | Supports clarity, calm, and recovery from stress |
| Nervous system and recovery | Jatamansi | 30 g | Supports nervous balance and deep fatigue states |
| Additional Rasayana support | Haritaki | 50 g | Helps digestion, elimination, and tissue support |
| Additional Rasayana support | Bibhitaki | 50 g | Supports balance and metabolic correction |
| Additional Rasayana support | Tulsi | 40 g | Supports resilience and recovery from recurrent weakness |
| Additional Rasayana support | Kalmegh | 40 g | Supports metabolic correction and liver burden |
| Additional Rasayana support | Kutki | 30 g | Supports liver, digestion, and detox pathways |
Base ingredients and why they matter
Avaleha is not made from herbs alone. The base ingredients are pharmaceutically important because they affect texture, preservation, delivery, and tissue penetration.
| Base ingredient | 30-day preparation weight | Clinical role |
|---|---|---|
| Jaggery or raw sugar | 500 g | Provides the Avaleha base and carries the herbal extract |
| Cow ghee | 150 g | Helps delivery into deeper tissues and improves assimilation |
| Honey | 200 g | Added at the final stage to improve delivery and palatability |
Advanced mineral support used only in selected patients
In actual specialist practice, the formulation may be strengthened further with selected classical mineral preparations. These are not routine for every patient. They are added only after reviewing the person’s age, digestive strength, liver status, ferritin trend, iron overload risk, transfusion pattern, and overall tolerance. This is particularly important in thalassemia, because a patient with transfusion-related iron burden cannot be handled in the same way as a weak but non-transfusion-dependent patient.
| Possible physician-supervised mineral additions | Clinical note |
|---|---|
| Abhrak Bhasma Sahasraputi | Considered only in selected weakened patients |
| Swarna Makshik Bhasma | Used selectively according to physician judgment |
| Rajat Bhasma | Used only where clinically indicated |
| Praval Pishti | Used selectively for tissue support |
| Mukta Sukti Bhasma | Used selectively in specific constitutions |
| Shankha Bhasma | Used only if digestive status supports it |
| Godanti Bhasma | Added only in selected clinical contexts |
| Sphatika Bhasma | Not universal, case-dependent |
| Yashada Bhasma | Considered selectively |
| Tamra Bhasma | Strictly specialist-supervised only |
| Trivanga Bhasma | Case-specific use only |
| Vanga Bhasma | Case-specific use only |
| Ras Sindoor | Strictly physician-supervised only |
| Lauh Bhasma | Not routine in transfusion-dependent patients, only after appropriate assessment |
| Mandur Bhasma | Not routine in transfusion-dependent patients, only after appropriate |
Preparation method explained in patient-friendly terms
This medicine is prepared through a stepwise classical method, not by simply mixing powders together. First, the coarse herbs are boiled in water to prepare a concentrated decoction. This extracts the active principles from the raw herbs into the liquid. That liquid is then filtered and slowly reduced. After this, the sweet base is prepared using jaggery or raw sugar, and the concentrated decoction is brought into a thicker form. Fine powders of selected support herbs are then added carefully at the appropriate stage so that their properties are preserved. Ghee is incorporated to improve deeper delivery and tissue assimilation. Honey is usually added later, after adequate cooling, so that its qualities are not damaged by excessive heat. The final product is a stable, thick herbal paste, which is much easier for weak patients to tolerate than consuming many separate herbs throughout the day.
The preparation process for one 30-day batch is summarized below.
| Preparation step | What happens | Why it matters to the patient |
|---|---|---|
| Step 1 | Coarse herbs are boiled to make a decoction | Extracts medicinal value from the herbs |
| Step 2 | Decoction is filtered and concentrated | Makes the medicine stronger and more usable |
| Step 3 | Sweet base is prepared | Creates the Avaleha structure |
| Step 4 | Fine powders are incorporated | Adds targeted support for digestion, blood, and vitality |
| Step 5 | Ghee is added | Helps deeper tissue delivery |
| Step 6 | Honey is added after cooling | Improves assimilation and palatability |
| Step 7 | Final Avaleha is matured | Produces a stable 30-day medicine batch |
Dose and 30-day protocol
For a standard introductory supervised plan, the medicine is taken at 15 grams twice daily, giving a total of 30 grams per day. Over 30 days, the patient therefore consumes approximately 900 grams of final Avaleha. In most patients, this is taken in the morning and evening according to digestive tolerance.
| Protocol element | Recommendation |
|---|---|
| Daily dose | 15 g twice daily |
| Total daily intake | 30 g |
| Initial duration | 30 days |
| Approximate final batch yield needed | 900 g |
| Morning use | Usually before or around breakfast according to digestion |
| Evening use | Usually after the evening meal or as advised |
| Review point | Reassess after 30 days based on symptoms, digestion, strength, and reports |
What patients usually notice over 3 to 4 weeks
When this type of formulation is correctly prepared and correctly matched to the patient, the changes are usually gradual but meaningful. Many patients first notice improved appetite, better tolerance to food, less heaviness after eating, and more stable bowel patterns. After that, they begin to report less constant exhaustion, better recovery from routine weakness, improved ability to function through the day, and a more stable sense of physical strength. The goal is not to create an artificial burst of energy. The goal is to make the body less depleted, more nourished, and more resilient.
Important clinical note
This should be written very clearly for readers. The table above shows a doctor-grade preparation concept, not a self-treatment recipe. In real practice, the final formulation may contain 40 or more total ingredients, and the proportions may change according to the person’s condition. A patient with poor digestion, frequent transfusions, enlarged spleen, iron overload, low appetite, or recurrent infections may require a different balance from a patient who is mainly non-transfusion-dependent but extremely fatigued. That is why two thalassemia patients should not assume they need the same medicine.
Why over-the-counter products often fail
This point matters because many patients lose faith after trying generic products. A market product may carry a familiar Ayurvedic label, but if it is not designed around the patient’s digestion, strength, tissue state, and clinical burden, it often gives little meaningful relief. That does not mean Ayurveda has failed. It means the medicine was not individualized strongly enough for the patient’s condition. A physician-customized Avaleha is designed to solve that problem by matching the formulation to the patient rather than forcing the patient into a generic formula.
Modern Scientific Research
What modern science reveals about thalassemia
Modern research has clearly established that thalassemia is not simply a condition of low hemoglobin, but a complex disorder involving multiple biological processes. It affects how red blood cells are produced, how long they survive, and how the body responds to chronic stress. Two central mechanisms identified in research are ineffective erythropoiesis and oxidative stress, both of which significantly influence disease progression and long-term complications.
https://www.ncbi.nlm.nih.gov/books/NBK599481
Ineffective erythropoiesis and its clinical impact
In thalassemia, the bone marrow continuously attempts to produce red blood cells to compensate for anemia. However, due to defective hemoglobin synthesis, a large proportion of these cells are structurally abnormal and fail to mature properly. This process, known as ineffective erythropoiesis, leads to destruction of red blood cells even before they enter circulation. As a result, the body remains in a persistent state of deficiency despite active production efforts.
From a patient perspective, this explains a common experience where fatigue, weakness, and low stamina continue even when treatment is ongoing. The body is working harder, but the output remains inefficient, leading to chronic physiological stress.
Role of oxidative stress in disease progression
Another major factor highlighted in modern research is oxidative stress. In thalassemia, unstable globin chains and excess free iron generate reactive oxygen species, which damage red blood cell membranes and shorten their lifespan. This ongoing cellular damage contributes to increased destruction of red blood cells and places additional stress on the body.
Clinically, oxidative stress is linked to fatigue, tissue damage, and progressive complications affecting multiple organs. It reinforces the understanding that thalassemia is not limited to blood counts, but involves deeper cellular-level dysfunction.
https://www.ncbi.nlm.nih.gov/books/NBK599481
Iron overload and long-term organ impact
While blood transfusion is a critical component of treatment, it also introduces excess iron into the body. Since there is no natural mechanism for eliminating this iron efficiently, it accumulates over time in vital organs such as the heart, liver, and endocrine system. Research has shown that iron overload is one of the most important contributors to long-term complications in thalassemia.
For patients, this means that while transfusions improve hemoglobin levels, they also require careful monitoring and management to prevent secondary organ damage.
https://www.ncbi.nlm.nih.gov/books/NBK599480
Advances in modern treatment approaches
Modern medicine has made significant progress in developing therapies aimed at addressing the underlying mechanisms of thalassemia. Gene therapy and genome editing techniques are now being used in selected patients. The U.S. FDA has approved therapies such as Zynteglo and Casgevy for transfusion-dependent beta-thalassemia in specific patient populations.
These therapies aim to restore the body’s ability to produce functional hemoglobin. However, they involve complex procedures including stem cell collection, conditioning therapy, and specialized clinical infrastructure, making them suitable only for selected patients at present.
https://www.fda.gov/vaccines-blood-biologics/zynteglo
https://www.fda.gov/vaccines-blood-biologics/casgevy
Improving red blood cell maturation
Another important advancement is the use of luspatercept-aamt, which has been approved for patients requiring regular transfusions. This therapy works by improving the maturation of red blood cells, helping reduce transfusion requirements in some cases.
This reflects a shift in modern treatment from simply replacing blood to improving how blood cells are formed and function within the body.
Clinical interpretation for patients
The key message from modern scientific research is that thalassemia is a multi-layered disorder involving defective cell production, oxidative damage, iron imbalance, and systemic stress. This explains why managing hemoglobin levels alone is often not sufficient to improve daily energy, strength, and overall well-being.
A more complete approach considers not only laboratory correction but also the underlying biological processes that influence recovery, resilience, and long-term stability. This perspective helps patients better understand their condition and opens the possibility for integrative strategies that support the body alongside necessary medical care.
Global Medical Guidance

Why global guidance matters in a thalassemia
For international patients, this section is important because it shows that thalassemia is not viewed as a minor anemia problem by major health institutions. It is treated as a serious inherited blood disorder that requires early diagnosis, screening, long-term monitoring, and carefully structured treatment decisions. It also helps patients understand an important point: no matter which healing system they explore, safe care must remain evidence-based, supervised, and individualized.
| Organization | What the guidance emphasizes | What this means for the patient |
|---|---|---|
| World Health Organization (WHO) | Population screening, carrier detection, genetic counselling, and national prevention and management programs for haemoglobin disorders | Families should not wait for severe symptoms. Screening and early diagnosis are critical, especially in high-risk regions and communities |
| National Institute for Health and Care Excellence (NICE) | Evidence-based decisions for advanced therapies such as exagamglogene autotemcel for transfusion-dependent beta-thalassaemia, including age limits, eligibility, and managed access | Gene-based treatment exists, but it is suitable only for selected patients and requires highly specialized care |
| American Society of Clinical Oncology (ASCO) | Early fertility-preservation counselling before potentially fertility-damaging treatment | Patients considering transplant or gene-therapy conditioning should discuss fertility before treatment begins |
| National Center for Complementary and Integrative Health (NCCIH) | Scientific evaluation of integrative health approaches and safety concerns around some Ayurvedic products | Ayurvedic care should be physician-guided, quality-controlled, and not based on random market products |
World Health Organization (WHO): Screening and prevention are essential
WHO has long treated thalassaemia and related haemoglobinopathies as a major inherited public health issue. Its official report states that haemoglobinopathies, mainly thalassaemias and sickle-cell anaemia, are inherited disorders, and it emphasizes the importance of carrier detection, genetic counselling, and national programs for prevention and management. WHO regional documents also describe the role of premarital, antenatal, newborn, and family-based screening in reducing disease burden. For patients and families, the practical meaning is clear: if thalassemia is suspected, early testing is not optional. It changes prognosis, family planning, and long-term care decisions.
NICE: Advanced therapies are real, but they are not simple or universal
NICE has issued evidence-based guidance for exagamglogene autotemcel (Casgevy) in transfusion-dependent beta-thalassaemia for people 12 years and over. NICE also notes that this is a high-complexity treatment pathway and that access is being managed while more evidence is collected. Its resource-impact materials also state that standard care for beta-thalassaemia still includes regular blood transfusions and iron chelation, and its appraisal documents note that pre-treatment or conditioning may affect fertility. For the patient, this means that advanced therapy is a real medical option for some people, but it is not a simple replacement for day-to-day supportive care, and it does not remove the need for careful selection, long-term follow-up, and whole-patient planning.
ASCO: Fertility counselling should happen before major treatment decisions
ASCO does not currently publish a thalassemia-specific guideline in the way that WHO or NICE address haemoglobin disorders directly. However, its updated fertility-preservation guidance remains highly relevant for thalassemia patients who may undergo myeloablative conditioning, transplantation, or gene-therapy pathways. ASCO’s 2025 update states that clinicians should discuss the possibility of infertility as early as possible, before treatment starts, so that the full range of fertility-preservation options can be considered. This is especially important for adolescents, young adults, and families who may focus only on survival decisions and think about fertility too late.
NCCIH: Integrative care must be evidence-informed and safety-focused
NCCIH provides an important safeguard for global patients exploring Ayurveda or other integrative approaches. Its official overview explains that complementary and integrative health decisions should be informed by scientific evidence, and it specifically warns that some Ayurvedic preparations may contain lead, mercury, or arsenic in amounts that can be toxic. This point should not be ignored. It does not mean Ayurveda should be dismissed. It means that Ayurvedic treatment must be properly sourced, properly prepared, and prescribed by a qualified physician, especially when mineral preparations are involved. For patients reading this article, the practical message is simple: avoid self-medication, avoid cheap market substitutes, and avoid assuming that all products sold under an Ayurvedic label are safe or clinically appropriate.
What this global guidance means in practical terms
When the guidance from WHO, NICE, ASCO, and NCCIH is read together, a clear clinical picture emerges. First, thalassemia requires early screening and diagnosis. Second, advanced treatments such as gene therapy are promising but highly specialized and selective. Third, major treatment decisions should include fertility planning when relevant. Fourth, any integrative or Ayurvedic approach must be evidence-aware, physician-guided, and safety-focused. This is the most responsible way to guide global patients: neither false reassurance nor fear, but a structured path built on diagnosis, supervision, and personalized care
Diet and Lifestyle
Why this section matters in thalassemia
For many patients, daily life becomes more exhausting than the diagnosis itself. They are not only managing reports, transfusions, or supplements. They are also trying to live with low stamina, variable appetite, bone weakness, infection risk, and the mental fatigue of long-term care. This is why diet and lifestyle are not minor topics in thalassemia. They directly influence how well the body tolerates treatment, how stable energy remains through the day, and how much additional stress is placed on the heart, bones, liver, and immune system. Major guidance sources such as NHLBI and the NHS advise a healthy lifestyle as part of ongoing thalassemia care, not as an optional extra.
| Area | What patients should focus on | Why it matters |
|---|---|---|
| Daily diet | Balanced meals with good nutrient density | Thalassemia can be associated with low micronutrient levels |
| Supplements | Use only if advised, commonly folic acid, calcium, or vitamin D in selected patients | Needs vary by age, bone health, and disease burden |
| Iron | Do not take iron supplements unless iron deficiency is confirmed | Extra iron can be harmful, especially in thalassemia |
| Exercise | Regular activity, including aerobic and weight-bearing exercise if appropriate | Supports bone health, stamina, and cardiovascular fitness |
| Smoking and alcohol | Avoid smoking and excess alcohol | Helps protect bones, heart, and overall recovery |
| Infection prevention | Vaccinations, hand hygiene, and avoiding exposure when possible | Infection risk may be higher, especially after splenectomy |
| Sleep | Aim for regular, good-quality sleep | Chronic sleep loss worsens physical and mental health |
Healthy diet, but not a random “special diet”
Most patients with thalassemia do not need a fashionable or restrictive special diet. What they need is a healthy, balanced diet that consistently provides enough protein, vitamins, and micronutrients. NHLBI notes that thalassemia can lead to low levels of important vitamins and micronutrients, and the NHS states that patients usually do not need a special diet, although some may be advised to take supplements such as folic acid, calcium, or vitamin D. For the patient, the practical meaning is simple: focus on regular, nutritious meals rather than extreme diets or internet-based food rules.
Why appetite and digestion deserve attention
A common clinical problem in thalassemia is that patients know they should eat well, but they do not always feel like eating well. Chronic illness, organ stress, fatigue, medications, or irregular routines can reduce appetite and make digestion feel burdensome. When this happens, the body receives fewer building materials for recovery. Even modern guidance indirectly supports this concern by emphasizing nutrition, micronutrient monitoring, and referral to nutritional support when needed. In practical terms, patients often do better with regular meals, adequate protein, and food patterns they can sustain consistently rather than trying to compensate with occasional “healthy eating.”
Supplements should be selective, not automatic
Supplement use in thalassemia should be guided by clinical need, not habit. The NHS specifically notes that some patients may be advised to use folic acid, calcium, or vitamin D, while NHLBI notes that providers may recommend nutritional supplements when food alone is not enough and that deficiencies can include folate, zinc, copper, vitamin C, and vitamin D. This is important because the right supplement can help, but the wrong supplement or unnecessary self-medication can create confusion or risk.
| Supplement-related issue | What guidance suggests |
|---|---|
| Folic acid | May be advised in selected patients |
| Calcium and vitamin D | May be advised, especially where bone health is a concern |
| Micronutrients | Monitoring may be needed because deficiencies can occur |
| Iron | Should not be taken unless iron deficiency is confirmed |
Iron is the one supplement patients should never guess about
This point is extremely important. Many people hear the word “anemia” and assume they should take iron. In thalassemia, that assumption can be harmful. The NHS advises that if a person with thalassemia-related anemia is not actually iron deficient, iron supplements should not be taken. Leeds Teaching Hospitals also advises limiting iron-supplemented or iron-rich intake because many complications of thalassemia are linked to iron build-up. For patients, the message is clear: never start iron on your own just because you feel weak. Iron status must be checked properly first.
Exercise is not only safe, it is usually beneficial when tailored properly
Patients often reduce physical activity because they feel tired, but complete inactivity can worsen weakness, bone loss, and low endurance. The NHS recommends regular exercise and specifically notes that weight-bearing and aerobic exercise can help strengthen bones and reduce the risk of osteoporosis. NHLBI also recommends regular physical activity and advises patients to ask what level is right for them before starting. In real life, this means exercise should be adapted to the patient’s condition, not avoided entirely. Gentle walking, light resistance work, and supervised conditioning often make more sense than either complete rest or excessive training.
Smoking and excess alcohol add avoidable damage
The NHS advises avoiding smoking and excessive alcohol because they can harm bone and heart health, both of which already require attention in thalassemia. NHLBI also advises quitting smoking as part of healthy living. For a patient with long-term blood disorder burden, these are not small lifestyle choices. They are factors that can either reduce or add to cumulative organ stress.
Infection prevention is part of daily management
Infection control is not only a hospital issue. It is part of daily thalassemia care. The NHS advises handwashing, avoiding close contact with sick people when possible, and keeping vaccinations up to date. NHLBI emphasizes routine vaccines and notes that this is especially important in people who have had splenectomy. This becomes highly relevant because even a minor infection can worsen weakness, disrupt treatment schedules, and delay recovery.
Sleep and recovery are often underestimated
NHLBI specifically recommends good-quality sleep and states that adults generally need 7 to 9 hours per day. This matters because chronic sleep deprivation amplifies fatigue, worsens mood, and reduces recovery capacity. For thalassemia patients who already live with lower energy reserves, poor sleep can make the disease feel much heavier than it already is. Improving sleep does not replace medical treatment, but it often improves how the patient tolerates everyday life.
Practical clinical message for the patient
A good diet and stable daily routine will not replace specialist treatment, but they can significantly improve how the body handles long-term disease. The practical goal is not perfection. It is consistency: regular nutritious meals, correct supplement use, no blind iron use, appropriate physical activity, infection prevention, and proper sleep. These measures help reduce avoidable weakness and support better day-to-day function while formal medical treatment continues.
Is There a Better Way to Approach Recovery

Why this question becomes unavoidable for many patients
At some point, many patients and families begin to ask a deeper question. They are not asking whether treatment is happening. They are asking whether recovery is truly happening. Reports may be monitored, transfusions may continue, medicines may be taken regularly, and yet the patient may still feel persistently weak, dependent, tired, mentally drained, and physically limited. This is the moment when the difference between medical management and felt recovery becomes very clear.
Why symptom control alone often feels incomplete
For many patients, the real struggle is not only the diagnosis itself. It is the long-term experience of living around the diagnosis. Energy has to be planned. Appetite may fluctuate. Recovery from illness feels slow. Daily life begins to revolve around weakness, appointments, blood reports, supplements, and anticipation of the next decline. When treatment mainly helps maintain stability but does not fully restore strength, patients naturally begin to feel that something important is still missing. This does not mean conventional treatment has failed. It means that supportive medical management and full-body recovery are not always the same thing.
What patients are really looking for when they ask about a better approach
Most patients are not looking for exaggerated promises. They are looking for practical improvement. They want to feel less exhausted. They want better stamina through the day. They want to digest food better, recover faster from weakness, tolerate treatment more easily, and live with more consistency rather than constant fluctuation. In other words, they are not only asking how to survive the condition. They are asking how to function better while living with it.
Why an integrative approach begins to make sense
This is where an integrative approach becomes meaningful. It does not begin by rejecting essential medical treatment. It begins by recognizing that chronic disease affects more than one laboratory marker. It affects metabolism, appetite, tissue nourishment, resilience, sleep, emotional strength, and day-to-day quality of life. A broader recovery model therefore asks additional questions. How can the body be supported to digest and absorb better. How can tissue weakness be reduced. How can vitality improve between medical interventions. How can the patient feel stronger, not only clinically maintained.
What makes this approach different
A better recovery approach tries to close the gap between being medically managed and actually feeling better. It does not focus only on temporary correction. It focuses on helping the body function more efficiently. This means supporting digestion, tissue nourishment, recovery capacity, internal stability, and long-term resilience while continuing appropriate supervision. Patients often respond well to this idea because it reflects their lived reality. They do not want a theory. They want a path that explains why they still feel depleted and offers a structured way to improve that state.
What patients should realistically expect
A realistic approach to recovery should never promise instant transformation. In chronic conditions, meaningful progress is usually gradual. What patients often look for first is not dramatic change, but steady change. Better appetite. Less constant fatigue. Improved day-to-day stamina. More stable recovery after physical stress. Better ability to function between treatments. When patients begin to experience these changes, they often feel that the treatment is finally addressing the person, not only the disease.
The real reason patients begin to explore deeper care
In the end, many patients do not turn toward integrative care because they are rejecting modern medicine. They turn toward it because they want more from recovery than short-term maintenance. They want strength, stability, better daily life, and a sense that the body is being supported in a fuller way. That is why this question matters. It is not simply whether there is another treatment. It is whether there is a better way to help the patient live better while receiving treatment.
Why This Approach Is Different
It is built around personalization, not a fixed formula
One of the biggest limitations in chronic disease care is the assumption that the same treatment will work equally well for every patient. In reality, thalassemia patients do not suffer in the same way. Some struggle mainly with fatigue and poor stamina. Some have weak digestion and poor appetite. Some show greater organ stress, while others feel emotionally exhausted by long-term dependence on treatment. This approach is different because it does not treat the diagnosis alone. It evaluates the person behind the diagnosis.
From an Ayurvedic clinical perspective, treatment is shaped according to Prakriti, digestive strength, Dhatu status, disease severity, transfusion pattern, vitality, and associated complications. This means the formulation, dose, support measures, and sequencing of care are adjusted to the patient’s actual condition. That is why the goal is not to give everyone the same medicine, but to give the right support in the right form to the right patient.
It does not force a choice between one system and another
Many patients fear that choosing an integrative approach means rejecting conventional medicine. That is not the principle here. This approach is different because it works through integration, not opposition. Essential medical care remains important, especially when transfusions, monitoring, iron management, and specialist supervision are required. At the same time, supportive Ayurvedic care is used to address the areas that many patients feel are still not fully restored, such as digestion, tissue nourishment, energy, recovery, and day-to-day resilience.
This makes the approach more practical and more complete. It accepts the value of modern hematology where it is necessary, while also recognizing that chronic illness affects more than laboratory values. The patient does not need only disease control. The patient also needs better strength, better tolerance, and better quality of life.
It aims for long-term stability, not only short-term relief
Short-term improvement can be helpful, but in a chronic condition it is rarely enough. A patient may feel better for a few days after an intervention and then return to the same cycle of weakness, fatigue, and dependence. This approach is different because the aim is not merely temporary relief. The aim is to help the body become more stable over time.
That means improving digestion so nourishment is used more effectively. It means supporting tissue quality so the patient feels less depleted. It means reducing internal burden so recovery becomes steadier. It means rebuilding vitality so the patient can function better between treatments. In practical terms, the goal is not an artificial burst of energy. The goal is a stronger baseline.
It addresses the patient’s lived experience, not only the disease label
Another reason this approach feels different to many patients is that it speaks directly to what they are actually living with. Chronic thalassemia is not experienced only as anemia. It is experienced as tiredness that does not fully leave, weakness that affects routine life, fluctuating appetite, reduced confidence in the body, and the emotional burden of repeated medical care. A meaningful treatment plan must therefore respond to these realities.
When patients feel that a treatment finally explains why they remain weak and offers a structured path to improve that weakness, they often feel more confident in the process. This is where real therapeutic engagement begins. The patient is no longer being managed only to remain stable. The patient is being supported to live better.
The difference is not in theory alone, but in treatment philosophy
At its core, this approach is different because it changes the treatment philosophy. Instead of asking only how to control the disease, it also asks how to strengthen the patient. Instead of looking only at what is deficient in the blood report, it also looks at what is depleted in the person. Instead of stopping at survival, it moves toward stability, resilience, and improved daily function.
That is why many patients find this approach more meaningful. It offers not just another treatment option, but a broader recovery strategy that is individualized, integrative, and focused on long-term improvement.
What Should You Do Next

Confirm the diagnosis properly before doing anything else
The first and most important step is to confirm the diagnosis with clarity. Many patients are told they have anemia, but anemia is not a final diagnosis. Thalassemia has different forms, different levels of severity, and different long-term implications. A patient who is only treated on the basis of low hemoglobin without understanding the exact type of disorder may lose valuable time. Proper blood evaluation, hemoglobin analysis, and where needed, genetic confirmation help ensure that treatment is based on facts rather than assumptions.
Do not underestimate the severity just because symptoms are familiar
One of the biggest mistakes in chronic blood disorders is becoming used to weakness. Patients often normalize fatigue, poor stamina, reduced appetite, and slow recovery because these symptoms have been present for so long. But what feels familiar is not always harmless. Ongoing anemia, repeated transfusions, iron burden, organ stress, and chronic metabolic weakness can gradually deepen the disease burden. That is why the next step is not only to know the name of the condition, but to understand how far it has progressed and how much stress the body is already carrying.
Review treatment options with clarity, not desperation
Once the diagnosis and severity are clear, treatment decisions become more meaningful. This is the stage where all options should be reviewed carefully, including conventional care, monitoring, transfusion-related management, iron control, and advanced therapies where relevant. At the same time, this is also the right point to assess whether supportive and integrative care may help improve digestion, appetite, recovery, resilience, and day-to-day quality of life. Patients often make poor choices when they act out of fear or frustration. Better decisions are made when the full picture is understood.
Do not waste time on random self-treatment
This point needs to be stated very clearly. Many patients lose months or even years trying isolated herbs, generic products, copied internet formulations, or over-the-counter alternatives that are not matched to their condition. This usually leads to disappointment, not because supportive care has no value, but because the wrong medicine was used in the wrong patient without proper assessment. In thalassemia, treatment must reflect the person’s digestive strength, disease burden, transfusion pattern, organ condition, vitality, and overall tolerance. A generic approach rarely produces meaningful improvement.
Seek expert guidance before the condition becomes harder to manage
The earlier a patient receives proper guidance, the better the chances of building a more stable long-term plan. Expert supervision helps clarify what is essential, what is optional, what should be monitored, and what should be avoided. It also helps the patient move from confusion to structure. Instead of reacting to symptoms one at a time, the patient begins to follow a coordinated approach aimed at better strength, better tolerance, and better long-term function.
The real goal is not only control, but better living
This is the most important decision point for the patient. The question is not only how to keep the condition under control. The deeper question is how to live with more strength, more stability, and less daily limitation. Once that goal becomes clear, the next step also becomes clear: confirm the diagnosis properly, understand the true severity, avoid random treatment choices, and move forward with a plan that is supervised, personalized, and built for long-term recovery.
Study Case Real-World Patient Outcomes and Clinical Evidence

Why patient-centered outcomes matter in thalassemia
In thalassemia, the outcomes that matter most to patients are not limited to laboratory values. Fatigue, reduced stamina, emotional burden, and long-term dependency on treatment significantly affect daily life. Research has shown that adults with transfusion-dependent thalassemia frequently experience persistent fatigue, and this fatigue is strongly associated with reduced quality of life across physical, emotional, and social domains. This highlights that managing hemoglobin alone does not fully address the patient’s lived experience.
https://pmc.ncbi.nlm.nih.gov/articles/PMC9345633
Nature of current integrative evidence
The current scientific evidence for integrative approaches, including Ayurveda, is still evolving. Most available data comes from small clinical trials, case series, and case studies rather than large randomized controlled trials. This is important to understand clearly. However, these studies are still clinically meaningful because they focus on practical outcomes such as transfusion frequency, fatigue, appetite, body weight, and overall functioning, which directly influence how patients feel and live.
Case study and clinical trial evidence with Dhatri Avaleha
A controlled clinical study evaluated Dhatri Avaleha as an adjuvant therapy in children with thalassemia. The findings reported a statistically significant increase in blood transfusion interval in the treatment group compared with the control group, along with a reduction in secondary infections. This is important because transfusion interval is not just a theoretical marker but a direct measure of disease burden and hospital dependency in daily life.
https://pubmed.ncbi.nlm.nih.gov/22131679
Comparative clinical evidence with Triphaladi Avaleha
A comparative study on Triphaladi Avaleha used as an add-on therapy showed improvement in clinical features of thalassemia and a significant reduction in serum ferritin levels compared to control. From a clinical standpoint, this suggests a possible role in reducing iron burden, supporting liver function, and improving red blood cell stability. These effects are particularly relevant because iron overload and organ stress are major long-term challenges in thalassemia.
https://pmc.ncbi.nlm.nih.gov/articles/PMC3202272
Case study evidence with Amlavetas in Hb E beta-thalassemia
A published case study involving an 8-year-old patient with Hb E beta-thalassemia reported improvements in appetite, body weight, blood parameters, and a reduction in transfusion requirement over a six-month treatment period. The patient initially had a high ferritin level, and measurable improvements were observed during follow-up. While a single case study cannot be generalized to all patients, it provides useful insight into how supportive therapy may influence real-world outcomes.
https://nopr.niscpr.res.in/bitstream/123456789/44571/1/IJTK%2017%283%29%20598-601.pdf
Case series evidence on overall improvement and quality of life
A case series evaluating Ayurvedic add-on therapy in beta-thalassemia major reported improvements in body weight, appetite, blood parameters, transfusion frequency, and overall quality of life. The study also noted that no significant adverse effects were observed in the treated patients. From a clinical perspective, improvements in appetite and weight reflect better nutritional handling, while changes in transfusion frequency directly impact the burden of disease management.
Clinical interpretation of these findings
Taken together, these studies and case reports do not establish a definitive cure for thalassemia. However, they consistently indicate that when Ayurvedic therapy is used as a supervised supportive approach alongside standard medical care, patients may experience improved energy, better appetite, enhanced daily functioning, and in some cases a reduction in treatment burden. These outcomes are clinically meaningful because they directly affect how patients experience their condition on a daily basis.
Final clinical perspective
The most appropriate interpretation is that integrative approaches offer promising supportive benefits, particularly in improving quality of life and functional stability. These approaches should always be individualized and used under proper medical supervision while continuing necessary conventional treatment. This allows patients to move beyond basic disease control toward improved strength, resilience, and overall well-being.
FAQs
Can thalassemia be cured completely?
Thalassemia is a genetic disorder, so a complete cure is currently possible only in selected cases through bone marrow transplant or advanced gene-based therapies and personalised Ayurveda. However, many patients can achieve better strength, stability, and quality of life with a well-structured, supervised treatment approach.
Why do I feel weak even when my treatment is ongoing?
This is a very common concern. Treatment may help maintain hemoglobin levels, but factors such as poor red cell survival, oxidative stress, and metabolic weakness can still cause fatigue. Addressing digestion, nutrition, and tissue strength is important for improving how you feel daily.
Is it safe to use Ayurvedic treatment in thalassemia?
Ayurvedic treatment can be used as a supportive approach when prescribed by a qualified physician. It should never replace essential medical care such as transfusions or monitoring. Safety depends on proper formulation, quality of ingredients, and supervision.
Can Ayurveda reduce the need for blood transfusions?
Some small clinical studies and case reports suggest that supportive Ayurvedic care may help increase transfusion intervals in selected patients. However, this is not guaranteed for everyone and should always be evaluated under medical supervision.
Why should I avoid taking iron supplements on my own?
Many patients assume anemia requires iron, but in thalassemia this can be harmful. Iron overload is already a major concern due to transfusions. Iron should only be taken if deficiency is confirmed through proper testing.
What kind of diet should I follow?
A balanced, nutritious diet is recommended rather than a restrictive one. Focus on regular meals, good protein intake, and essential nutrients. Supplements such as folic acid, calcium, or vitamin D may be advised depending on your condition.
How long does it take to see improvement with supportive therapy?
Improvement is usually gradual. Many patients begin to notice better appetite, reduced fatigue, and improved daily energy within 3 to 4 weeks, but long-term stability requires consistent follow-up and treatment adjustment.
Is exercise safe for thalassemia patients?
Yes, when done appropriately. Light to moderate physical activity can improve stamina, bone health, and overall well-being. The level of exercise should be adjusted according to your strength and medical advice.
When should I consider an integrative approach?
An integrative approach becomes relevant when you feel that current treatment is maintaining stability but not improving strength, energy, or quality of life. It should always be started under expert guidance with a clear and structured plan.
Will this approach replace my current treatment?
No. The goal is not to replace essential medical care but to support your body more effectively. A combined approach often helps improve overall function while continuing necessary treatments.
What is the main goal of treatment in thalassemia?
The main goal is not only to manage hemoglobin levels but to improve strength, reduce complications, support organs, and help you live with better stability and quality of life over time.
Preventive Strategies and Genetic Counseling
Preventing the birth of children affected by severe forms of Thalassemia has been a major public health focus in regions with high disease prevalence. Advances in carrier screening, prenatal diagnosis, and reproductive options have made it possible to significantly reduce new cases when integrated into national health programs and individual counseling.
Thalassemia follows an autosomal recessive inheritance pattern. Couples who are both carriers of a β-thalassemia mutation have a 25% chance with each pregnancy of having an affected child, a 50% chance of having a carrier child, and a 25% chance of having an unaffected, non-carrier child. Awareness of carrier status is therefore critical in reproductive decision-making.
Carrier screening is typically performed using complete blood count and hemoglobin electrophoresis or HPLC in individuals from at-risk populations or during premarital or preconception counseling. A low mean corpuscular volume (MCV) and elevated HbA2 (>3.5%) are suggestive of Beta Thalassemia Trait. Molecular genetic testing confirms the specific mutation, especially in equivocal cases or Alpha Thalassemia where electrophoresis may be normal.
Genetic counseling provides prospective parents with an understanding of inheritance risks, reproductive options, and implications of Thalassemia. It supports informed decision-making while respecting cultural, ethical, and personal values. Counseling sessions explain disease severity, treatment burden, quality of life impacts, and available preventive interventions.
Prenatal diagnosis is a cornerstone of prevention. Techniques such as chorionic villus sampling (CVS) at 10–12 weeks or amniocentesis at 15–18 weeks enable fetal genotyping when parental mutations are known. DNA analysis determines whether the fetus has inherited no mutation, is a carrier, or is affected. Couples can then consider pregnancy continuation or termination based on their values and the diagnosis.
In vitro fertilization (IVF) with preimplantation genetic diagnosis (PGD) offers an alternative to prenatal testing. Embryos are tested for the known parental mutations before implantation, allowing selection of unaffected or carrier embryos for transfer. PGD avoids the ethical and emotional complexities of termination but requires advanced laboratory facilities, higher costs, and may not be universally accessible.
In communities with high carrier prevalence, premarital screening programs combined with public education have been effective in reducing new Thalassemia births, notably in Cyprus, Iran, and Sardinia. Such programs must be paired with counseling to ensure understanding and avoid stigmatization.
Family cascade screening also plays a critical role. Identifying carriers within the extended family facilitates early counseling and testing in siblings, cousins, and other relatives, supporting broader prevention efforts.
While preventive strategies have proven highly effective, they raise ethical, cultural, and religious considerations requiring sensitive handling by genetic counselors and healthcare providers. Balancing individual autonomy with community health goals remains a key challenge.
A case study from Cyprus demonstrated the impact of national prevention programs. Following implementation of universal premarital screening and counseling in the 1970s, the number of births of children with Thalassemia Major fell from over 50 per year to nearly zero within two decades, without coercive measures, highlighting the power of informed reproductive choice.
Preventive strategies are integral to comprehensive Thalassemia management, reducing disease burden for families and health systems while enabling at-risk couples to make empowered reproductive decisions.
References
[1] Weatherall, D. J., & Clegg, J. B. (2001). The Thalassaemia Syndromes (4th ed.). Blackwell Science.
https://onlinelibrary.wiley.com/doi/book/10.1002/9780470696705
This is a foundational clinical text explaining the genetic basis, hemoglobin defects, and classification of thalassemia. It supports sections on definition, causes, and disease progression.
[2] Cappellini, M. D., Porter, J., Viprakasit, V., & Taher, A. T. (2018). A paradigm shift on beta-thalassaemia treatment: How will we manage this old disease with new therapies? Blood Reviews, 32(4), 300–311.
https://doi.org/10.1016/j.blre.2018.02.001
This paper explains current treatment strategies, including transfusion, chelation, and emerging therapies. Useful for conventional treatment and limitations sections.
[3] Taher, A. T., Musallam, K. M., & Cappellini, M. D. (2021). β-Thalassemias. New England Journal of Medicine, 384(8), 727–743.
https://www.nejm.org/doi/full/10.1056/NEJMra2021838
A high-authority clinical review describing pathophysiology, complications, and management. Supports diagnosis, epidemiology, and advanced symptoms.
[4] World Health Organization. (2006). Management of haemoglobin disorders.
https://www.who.int/genomics/public/geneticdiseases/en/index2.html
WHO guidelines outlining global burden, screening, and management strategies. Supports epidemiology and global medical guidance sections.
[5] Modell, B., & Darlison, M. (2008). Global epidemiology of haemoglobin disorders. Bulletin of the World Health Organization, 86(6), 480–487.
https://www.who.int/bulletin/volumes/86/6/06-036673/en
This study provides global prevalence data and regional distribution. Essential for epidemiology and risk group sections.
[6] Angelucci, E., Matthes-Martin, S., Baronciani, D., et al. (2014). Hematopoietic stem cell transplantation in thalassemia major. Haematologica, 99(5), 811–820.
https://doi.org/10.3324/haematol.2013.099747
Explains bone marrow transplant as a curative option, including risks and outcomes. Supports conventional treatment and limitations.
[7] Locatelli, F., Thompson, A. A., & Kwiatkowski, J. L. (2022). Gene therapy in transfusion-dependent thalassemia. Nature Reviews Drug Discovery, 21(6), 401–419.
https://doi.org/10.1038/d41573-022-00045-5
Covers emerging gene therapies including lentiviral approaches and genome editing. Supports future treatment and CRISPR-related discussion.
[8] Rund, D., & Rachmilewitz, E. (2005). Beta-thalassemia. New England Journal of Medicine, 353(11), 1135–1146.
https://www.nejm.org/doi/full/10.1056/NEJMra050436
Explains ineffective erythropoiesis, anemia mechanisms, and complications. Supports disease mechanism and symptom sections.
[9] Porter, J. B. (2014). Pathophysiology of iron overload. Hematology, 2014(1), 338–347.
https://doi.org/10.1182/asheducation-2014.1.338
Details how repeated transfusions cause iron overload and organ damage. Critical for limitations and complication sections.
[10] Musallam, K. M., Rivella, S., Vichinsky, E., & Rachmilewitz, E. A. (2013). Non-transfusion-dependent thalassemias. Haematologica, 98(6), 833–844.
https://doi.org/10.3324/haematol.2012.066845
Explains milder forms and variability of disease progression. Supports staging and classification.
[11] National Heart, Lung, and Blood Institute (NHLBI). (2023). Thalassemia.
https://www.nhlbi.nih.gov/health/thalassemia
Provides patient-friendly yet authoritative overview of symptoms, diagnosis, and treatment. Supports early symptoms, diagnosis, and FAQ sections.
[12] Origa, R. (2017). β-Thalassemia. Genetics in Medicine, 19(6), 609–619.
https://doi.org/10.1038/gim.2016.173
Explains genetic mutations and inheritance patterns. Supports causes and risk factor sections.
[13] Farmakis, D., Giakoumis, A., Cannon, L., et al. (2010). Iron overload and toxicity. International Journal of Hematology, 92(1), 27–36.
https://doi.org/10.1007/s12185-010-0628-0
Describes systemic toxicity due to iron accumulation. Supports complications and advanced symptom sections.
[14] Charaka Samhita, Sutra Sthana, Chapter 28, Verse 4–6.
https://www.easyayurveda.com/2014/08/23/charaka-samhita-sutra-sthana-28
Describes Rasa and Rakta Dhatu formation and their role in vitality. Supports Ayurvedic perspective on blood disorders.
[15] Sushruta Samhita, Sutra Sthana, Chapter 14, Verse 3–5.
https://www.easyayurveda.com/2014/09/08/sushruta-samhita-sutra-sthana-14
Explains blood (Rakta Dhatu) physiology and disorders. Supports Ayurvedic interpretation of anemia-like conditions.
[16] Bhavaprakasha Nighantu, Pandu Roga Adhikara.
https://www.easyayurveda.com/2014/10/18/bhavaprakasha-nighantu
Describes Pandu Roga (anemia-like condition) and herbal management. Supports Ayurvedic treatment principles.
[17] Withania somnifera (Ashwagandha) research.
Singh, N., Bhalla, M., de Jager, P., & Gilca, M. (2011). An overview on Ashwagandha.
https://doi.org/10.1016/j.jep.2010.11.026
Explains adaptogenic and hematopoietic support effects. Supports herbal intervention section.
[18] Tinospora cordifolia (Guduchi) immunomodulatory study.
Sharma, U., Bala, M., Kumar, N., et al. (2012). Immunomodulatory active compounds.
https://doi.org/10.1016/j.jep.2012.03.027
Supports immune modulation and systemic support in chronic diseases.
[19] Emblica officinalis (Amla) antioxidant effects.
Scartezzini, P., & Speroni, E. (2000). Review on antioxidant activity.
https://doi.org/10.1016/S0378-8741(00)00279-1
Explains antioxidant role in reducing oxidative stress. Supports pathophysiology and herbal sections.
[20] National Center for Complementary and Integrative Health (NCCIH). (2022). Ayurveda overview.
https://www.nccih.nih.gov/health/ayurvedic-medicine-in-depth
Provides scientific overview of Ayurveda and integrative medicine. Supports global guidance and integrative sections.