- What Your Breast Biopsy Report Really Means
- Before Surgery Pathway: Do You Really Need Surgery
- After Surgery Pathway: Why Risk Can Still Remain
- Anatomy & Pathophysiology
- Symptoms
- Classification
- Diagnosis
- Risk Stratification
- Associated Disorders and Systemic Correlations
- Less Common but Clinically Significant Associations
- Rare but High-Significance Associations
- Clinical Significance
- What Should You Do After a Benign or Atypical Breast Diagnosis?
- Long-term prevention strategy
- Clinical Decision Pathway After a Benign or Atypical Breast Diagnosis
- Allopathy (Conventional Treatment)
- Limitations of Conventional Treatment
- What Patients Are Not Told After Diagnosis
- Integrative Safety: Where Ayurveda Fits and What Should Never Be Delayed
- Ayurvedic Perspective on Benign Breast Lump and Atypical Hyperplasia
- Ayurvedic Treatment Principles
- Integrative Ayurvedic Plan
- Ayurvedic Herbal Formulation for Breast Cancer
- Herbs and Scientific Support
- Global Medical Guidance
- Is There a Better Way to Approach Recovery
- What Should You Do Next
- Case Study (Real-World Patient Patterns)
- Frequently Asked Questions (FAQs)
Being told that your breast biopsy is “benign” should bring relief. Yet for many women, it creates a deeper layer of confusion and anxiety. Reports often include terms like atypical hyperplasia, proliferative changes, dense breast tissue, or high-risk lesions. At that moment, the question is no longer just “Is this cancer?” but rather “What does this really mean for my future?”
According to the World Health Organization (WHO), early understanding and timely decision-making play a critical role in long-term cancer outcomes.
However, in real clinical practice, many patients are left in a grey zone. They are told that the condition is not cancer, yet they are also advised to consider surgery, repeated imaging, or long-term surveillance. This gap between reassurance and uncertainty is where most patients begin searching for answers.
Some women are at the stage where surgery has not yet been performed and are trying to understand whether it is truly necessary. Others have already undergone biopsy or excision and are surprised to learn that follow-up, screening, and risk monitoring still continue. Clinical pathways guided by organizations such as the American Society of Clinical Oncology (ASCO) emphasize evidence-based treatment and surveillance, but they often focus primarily on structural diagnosis and intervention rather than the broader biological environment that influences long-term outcomes.
This is where many patients begin to feel that something is missing. They are monitored, but not fully guided. They are treated, but not always supported in long-term recovery or prevention.
Guidelines from the National Institute for Health and Care Excellence (NICE) also highlight the importance of recognizing when symptoms require urgent evaluation, reinforcing that timely medical care should never be delayed.
At the same time, there is growing global interest in understanding how supportive, integrative approaches may contribute to overall health when used responsibly.
The National Center for Complementary and Integrative Health (NCCIH) clearly states that complementary approaches should not replace necessary medical treatment, but when used appropriately, they may support well-being, symptom management, and recovery alongside standard care.
This article is designed to bridge that gap.
It provides clear, clinically grounded answers for both:
- patients who are deciding whether surgery is necessary
- patients who have already undergone treatment but want to understand what comes next
At the same time, it introduces an integrative perspective that does not oppose modern medicine, but rather complements it by focusing on long-term balance, recovery, and resilience.
Because the goal is not just to label a condition.
The goal is to understand it, manage it wisely, and move forward with clarity and confidence.
What Your Breast Biopsy Report Really Means

Receiving a breast biopsy report is often the most confusing stage of the entire journey. Many patients expect a clear answer, either cancer or not cancer, but instead receive terms that feel technical and uncertain. Words like atypical hyperplasia, proliferative changes, flat epithelial atypia, papilloma, or radial scar can create more questions than answers. The most important point to understand is that a benign or atypical result does not mean cancer is present, but it also does not always mean that the risk is completely absent. This is why your report must be interpreted carefully in the context of imaging, clinical findings, and long-term risk, rather than being read in isolation.
Why Biopsy Reports Often Feel Confusing
A breast biopsy examines a small sample of tissue under a microscope. While this provides important diagnostic information, it does not always represent the entire lesion or surrounding tissue. This is why doctors do not rely only on the pathology report. They compare imaging results such as mammography, ultrasound, or MRI with biopsy findings and clinical examination. This process is known as radiologic–pathologic correlation. When imaging and biopsy findings match, the situation is considered concordant and may not require further intervention. When they do not match, further evaluation or surgery may be recommended to rule out a more serious condition. Understanding this concept helps explain why different patients with similar biopsy terms may receive very different treatment recommendations.
Common Terms in Breast Biopsy Reports
Atypical ductal hyperplasia, often referred to as ADH, is one of the most commonly reported atypical findings. It indicates that the cells lining the breast ducts are growing in an abnormal pattern. ADH is not cancer, but it is considered a high-risk lesion because it is associated with an increased likelihood of developing breast cancer in the future. In some cases, doctors recommend surgical excision to ensure that no nearby area of more advanced disease was missed during the initial biopsy. However, not every case requires immediate surgery, and the decision depends on imaging findings, lesion characteristics, and overall risk profile.
Atypical lobular hyperplasia, or ALH, involves abnormal cell growth in the lobules of the breast. Like ADH, it is not cancer but signals an increased long-term risk. ALH is often managed more conservatively when imaging and biopsy findings are consistent, but follow-up remains essential because the associated risk is not limited to one area and may involve both breasts over time.
Flat epithelial atypia, known as FEA, represents an early abnormal change in the ductal lining and is often detected when microcalcifications appear on a mammogram. In many cases, FEA may not require surgery if the lesion is small and well-sampled, but if there is uncertainty or additional abnormalities, excision may still be advised.
An intraductal papilloma is a small growth within a breast duct. When it occurs without atypia, it is usually benign and may be managed with observation if it is asymptomatic. However, if there are symptoms such as nipple discharge or if atypical cells are present, surgical removal is more commonly recommended.
A radial scar, also known as a complex sclerosing lesion, is a structural change in breast tissue that can mimic cancer on imaging. When no atypia is present and imaging findings are consistent, monitoring may be sufficient. However, if atypical cells are found or imaging raises concern, excision is often advised to ensure safety.
The Most Important Question: Do You Need Surgery
This is the point where most patients feel uncertain. The decision to proceed with surgery is not based on a single term in the biopsy report. It depends on multiple factors including whether imaging and pathology findings are aligned, whether atypical cells are present, the size and location of the lesion, personal and family history, and the ability to maintain consistent follow-up. Some patients can safely undergo surveillance with regular imaging, while others are advised to proceed with surgery to ensure that no more advanced disease is present. This explains why two patients with similar biopsy reports may receive different clinical recommendations.
Why a Benign Result Does Not Always Mean No Risk
One of the most misunderstood aspects of breast biopsy results is the difference between diagnosis and risk. Certain benign and atypical conditions act as risk markers rather than immediate disease. They indicate that the breast tissue environment may already be showing changes associated with increased future risk. Even when a lesion is removed, the underlying biological tendency may still exist. This is why follow-up imaging, clinical exams, and risk-reduction strategies remain important. Diagnosis explains what is present today, while risk assessment helps predict what may develop over time.
Where Most Patients Feel Uncertain
At this stage, many patients experience a gap in understanding. They are told that the condition is not cancer, yet they are advised to consider surgery or ongoing surveillance. Follow-up imaging is scheduled, but a clear long-term plan is often not explained in detail. Patients are rarely guided on how to improve tissue health, address metabolic or hormonal factors, or reduce long-term risk beyond monitoring. This lack of clarity can lead to ongoing anxiety and confusion.
What This Means for You Right Now
If your biopsy report includes terms such as atypia or high-risk lesions, the next step is not panic but structured understanding. You need to know whether your condition requires surgery or can be safely observed, whether imaging and biopsy findings are consistent, what your long-term risk profile looks like, and what actions you can take beyond routine surveillance. This forms the foundation for making a confident and informed decision about your care.
Transition to the Next Step
Once you understand your biopsy report, the path forward depends on your current stage. If you have not had surgery yet, the focus shifts to deciding whether it is necessary and how to approach that decision safely. If you have already undergone surgery or excision, the focus changes to understanding why follow-up still matters and what can be done to reduce future risk and support long-term health.
Before Surgery Pathway: Do You Really Need Surgery

Reaching the point where surgery is mentioned is often the moment when fear becomes real. Many women go into a biopsy hoping for a simple answer. Instead, they are told the lesion is benign or atypical, but that surgery may still be discussed. This creates immediate confusion. The patient hears that it is not cancer, yet the language around excision, risk, and follow-up makes it feel more serious than she expected. Caregivers often react in the same way. They ask a practical question that sounds very simple but is emotionally loaded: if this is not cancer, why are doctors talking about surgery at all? That tension is one of the most important realities to acknowledge, because it is exactly where many patients begin searching for something more complete than a short clinic explanation. Mayo Clinic notes that not everyone with atypical hyperplasia needs surgery, but some patients do, depending on imaging findings, biopsy results, and other risk factors.
If You Have Not Had Surgery Yet
If you have not had surgery yet, your first need is not speed. It is clarity. Many patients feel pressured by the word “atypia” without fully understanding what it means. Some leave the appointment more frightened by the recommendation than by the diagnosis itself. Others go home, read the report again, and become alarmed by terms such as high-risk lesion, microcalcifications, proliferative change, discordance, or excision advised. This is where emotional distress increases. The patient may fear that waiting is dangerous, while at the same time fearing that surgery may be unnecessary. The caregiver may begin seeking multiple opinions, reading online forums, and worrying whether too much or too little is being done. This is a real part of the pre-surgical journey, and it should be addressed openly.
Clinically, the decision is based on more than one word in the pathology report. Doctors look at whether the imaging findings and biopsy findings truly match, whether atypical cells are limited or more extensive, whether the area was sampled adequately, and whether the lesion has features associated with a higher chance of hidden malignancy. Mayo explains that management depends on the findings seen on mammogram and the biopsy result, which is why two women with apparently similar reports may still receive different advice.
When Observation May Be a Safe Option
For some patients, observation with structured follow-up is a safe and reasonable choice. This is usually considered when the lesion is small, adequately sampled, and clearly matches the imaging findings. In such cases, the medical team may recommend repeat imaging and clinical review instead of immediate surgery. From the outside, this sounds simple, but for the patient it rarely feels simple. Many women struggle with the emotional burden of being told to “watch and wait.” They may ask themselves whether surveillance is true safety or just delayed intervention. Some become anxious before every mammogram or ultrasound, worried that a missed change may appear later. This is why observation should never be described casually. It is not doing nothing. It is an active plan that depends on disciplined follow-up and a clear understanding of risk.
This is also where Ayurveda can be discussed in a more meaningful way. When a patient is under observation, she often feels that modern care is monitoring the lesion but not addressing the rest of her body. She may want guidance on inflammation, digestion, hormonal balance, stress, sleep, and long-term tissue health. These are not minor concerns. They are part of why so many patients begin looking for integrative support. The safest and most credible way to present Ayurveda here is as a structured supportive approach during surveillance, not as a reason to ignore imaging or biopsy recommendations. NCCIH advises that complementary approaches should not replace necessary treatment or delay medical care, but may support well-being when used appropriately alongside standard care.
When Surgery Is Strongly Recommended
There are also situations where surgery becomes the safer option. This usually happens when imaging and pathology do not align, when atypical changes are more extensive, when the biopsy may not have sampled the whole area adequately, or when the lesion has features associated with higher upgrade risk. In these cases, surgery is not being recommended merely to “remove something suspicious.” It is being used to answer a more important question: was the biopsy enough to tell the full truth about the lesion? That distinction matters because patients often interpret surgery as an aggressive step, while the medical team may see it as the most reliable way to avoid missing a more advanced problem.
This is where many families become emotionally divided. One person wants the lesion removed immediately for peace of mind. Another worries about overtreatment, scarring, cost, recovery time, or the possibility that surgery is being done mainly out of caution. These are real concerns, and they should not be dismissed. The best articles are the ones that acknowledge both fears: the fear of doing too little and the fear of doing too much. Mayo makes clear that surgery may be recommended when mammogram images show a more concerning pattern or when other factors raise concern.
What Most Patients and Caregivers Do Not Realize
One of the biggest misunderstandings at this stage is the belief that the decision is only about removing a lump. In reality, the decision is about confidence in the diagnosis and future risk. Atypical and proliferative lesions can act as warning signals that the breast tissue environment is already showing biological changes linked with higher long-term risk. This means that even before surgery is chosen or declined, the deeper question has already appeared: why did this change happen, and what can be done to reduce the chance of further progression over time.
Caregivers often understand this only later. At first, they focus on the immediate issue, whether surgery is needed. Then they begin to realize the longer journey may involve repeated imaging, risk discussions, and ongoing uncertainty. That is the moment when many families feel that standard care has identified the problem, but not fully answered how to support the patient beyond the next appointment. This feeling is one of the strongest reasons people remain engaged with integrative care discussions.
Where Ayurveda Fits Before Surgery
Before surgery, Ayurveda should not be positioned as a substitute for proper breast evaluation. That approach would weaken trust and can be unsafe. Instead, it should be framed in a way patients and caregivers find believable: as the part of care that supports the person while the lesion is being evaluated and decisions are being made. At this stage, patients are often looking for help with anxiety, sleep disturbance, digestive stress, inflammatory burden, hormonal imbalance, and the feeling that their whole system has become unstable after the biopsy result. A responsible Ayurvedic perspective can address those needs while respecting the importance of imaging, pathology, and surgical consultation when required. NCCIH’s guidance supports this type of careful complementary framing rather than replacement logic.
This is also where the emotional shift begins. A patient who feels trapped between fear and uncertainty is more likely to continue reading when she sees that the article understands her real situation. You as patient or care taker don’t just want to know whether surgery may be needed. You want to know whether there is anything meaningful you can do right now to support your/her body, reduce future risk, and move forward with more confidence. That is where Ayurveda becomes relevant, not as a slogan, but as a practical long-term layer of support.
What You Should Do Right Now
If you have not had surgery yet, the most important next step is to understand your individual pathway. You need to know whether your imaging and biopsy findings are concordant, whether your lesion falls into a lower-risk or higher-risk category, and whether surveillance or excision is the safer route in your case. But you should also begin thinking beyond the immediate decision. Whether you proceed with surgery or remain under observation, the presence of atypical or proliferative changes is a signal that your long-term health deserves attention at more than one level.
This is where the article should guide the reader gently but clearly. Medical decisions should be made with proper diagnostic safety. At the same time, many patients also want a deeper plan for tissue health, metabolic balance, inflammation control, emotional steadiness, and long-term resilience. That combination, not panic and not denial, is what helps patients move forward wisely.
Transition to the Next Step
Many patients assume that once the surgery decision is made, the hardest part is over. In reality, that is not always true. Some women who undergo biopsy or excision later discover that follow-up still continues, that future risk may still need attention, and that the feeling of uncertainty does not always disappear with removal alone. Understanding that next phase is essential, because it is often the moment when patients become most open to a broader, more supportive healing strategy.
References for this section
Mayo Clinic. (2024). Atypical hyperplasia of the breast: Diagnosis and treatment. Mayo Clinic. https://www.mayoclinic.org/diseases-conditions/atypical-hyperplasia/diagnosis-treatment/drc-20369778
Brief: Explains what atypical hyperplasia means, when surgery may or may not be needed, and why long-term follow-up remains important.
National Center for Complementary and Integrative Health. (2023). Cancer and complementary health approaches: What you need to know. U.S. Department of Health and Human Services. https://www.nccih.nih.gov/health/cancer-and-complementary-health-approaches-what-you-need-to-know
Brief: Clarifies that complementary approaches should not delay necessary medical treatment, but may support well-being when used appropriately alongside standard care.
After Surgery Pathway: Why Risk Can Still Remain
Many women believe the hardest part is over once a biopsy or excision has been completed. The abnormal area has been sampled or removed, the wound begins to heal, and the family expects closure. But for many patients, this is the stage where a second layer of confusion begins. They are told the lesion has been removed, yet follow-up appointments continue. Mammograms are still discussed. In some cases, MRI is mentioned. Sometimes risk-reducing medicines are brought into the conversation. That feels contradictory to patients and caregivers. A very common question is, “If the lesion is gone, why am I still being treated like someone at risk?” This confusion is real, and it deserves a clear explanation. Mayo Clinic notes that atypical hyperplasia increases future breast-cancer risk and that management after diagnosis may include more frequent screening, clinical breast exams, and, in selected higher-risk patients, additional imaging such as breast MRI.
If You Already Had Biopsy or Excision
After biopsy or surgical excision, many patients expect a final answer. Instead, they may find themselves entering a new category, high-risk follow-up. This can be emotionally difficult because the procedure may have removed the visible lesion, but it does not always remove the reason that lesion developed in the first place. Families often feel unprepared for this. They expected a procedure, a recovery period, and then reassurance. What they were not prepared for was the possibility that surveillance might still continue even after surgery. Johns Hopkins advises that patients diagnosed with atypical ductal hyperplasia should be followed in a high-risk breast clinic, where screening, lifestyle measures, and risk-reducing strategies are discussed. MD Anderson similarly explains that if excision confirms atypical ductal hyperplasia without cancer, patients are then moved into high-risk screening discussions that can include annual mammograms, breast MRI, and conversations about risk-reducing medication.
Why Removal Does Not Always End the Risk
This is one of the most misunderstood parts of the post-surgical journey. Some breast lesions are not just isolated findings. They are also markers that the breast tissue has already shown a pattern associated with increased future risk. The American Society of Breast Surgeons states that certain high-risk lesions, including atypical ductal hyperplasia and lobular neoplasia, are linked to elevated lifetime breast-cancer risk that is not mitigated by surgical excision. In simple terms, surgery may remove the abnormal area that was found, but it does not necessarily erase the broader risk pattern associated with that type of tissue change. That is why a woman can be told, truthfully, that the lesion has been removed and, at the same time, that her future risk still deserves close attention.
For patients, this can feel unfair. Many feel they did everything they were told to do, attended imaging, underwent biopsy, agreed to excision, recovered from the procedure, and yet still do not feel fully released from worry. Caregivers often struggle with this too. They may wonder whether something was missed, whether the surgery was incomplete, or whether the doctors are simply being overcautious. In most cases, the issue is neither missed surgery nor panic. The issue is that high-risk breast care is not only about what was removed today. It is also about what that tissue pattern may mean for the years ahead. Mayo Clinic’s guidance reflects this long-term approach by recommending ongoing screening and risk-reduction planning after atypical hyperplasia is diagnosed.
Why Follow-Up Continues After Surgery
Ongoing follow-up is usually not a sign that surgery failed. More often, it means the patient’s risk profile still matters. Follow-up may include regular clinical breast exams, annual mammography, and, in selected patients, supplemental imaging depending on breast density, family history, genetic risk, and the pattern of findings. Breast MRI can be useful in some higher-risk settings, but it can also create another kind of burden. It may lead to false-positive findings, extra testing, and renewed anxiety in women who already feel exhausted by the process. Mayo Clinic notes both the role of MRI in selected higher-risk patients and the fact that additional imaging can sometimes lead to unnecessary worry and more testing.
This creates a real burden that patients and caregivers often feel but do not always know how to describe. Every new scan can reopen fear. Every follow-up visit can feel like waiting for another surprise. Some women become more vigilant but less peaceful. Others begin avoiding appointments because they feel emotionally drained by repeated uncertainty. This is exactly the point where an article should go beyond medical definitions. Patients do not only need to know that surveillance continues. They need to understand why it continues and what they can do to feel less passive inside that process.
What Most Patients Feel but Rarely Say
After surgery, many women quietly expect reassurance and closure. What they often feel instead is an in-between state. They are not being treated as cancer patients, but they no longer feel fully low-risk either. They are told to move forward, but they still carry the memory of the report, the procedure, and the follow-up plan. Caregivers notice this as well. Some women become more worried about minor sensations in the breast. Others become anxious before every mammogram. Some keep asking the same question in different ways: “If the tissue has already been removed, what are we still trying to prevent?”
This is where conventional care can feel incomplete from the patient’s perspective. The lesion has been addressed structurally, but the patient may still feel there is no full explanation for why the abnormal change developed, no practical plan for improving the internal environment of the body, and no deeper roadmap for long-term resilience beyond screening and medication discussions. That feeling of an unfinished story is one of the strongest reasons patients keep searching for more comprehensive answers.
Where Ayurveda Fits After Surgery
This is the stage where Ayurveda can be presented most convincingly and responsibly. After surgery, the role of Ayurveda should not be framed as replacing follow-up or dismissing high-risk screening. It should be framed as addressing the part of healing that many patients feel was left unaddressed. Once the lesion has been removed and urgent structural decisions have been made, patients often become more open to questions about digestion, metabolic balance, inflammatory load, hormonal stability, stress recovery, sleep, tissue nourishment, and long-term resilience. These are the areas where an integrative approach feels relevant to real life, not just theory.
This positioning is also more credible medically. NCCIH states that complementary approaches should not replace necessary medical treatment, but may support well-being and symptom management when used appropriately alongside standard care. That is exactly how this section should guide the reader. Ayurveda here is not the alternative to proper follow-up. It is the supportive layer that can help patients feel they are doing something meaningful for their whole system while medical surveillance continues.
What You Should Do After Surgery
If you have already had biopsy or excision, the next step is not to assume the story is over. It is to understand your position clearly. You need to know whether your pathology places you in a higher-risk category, what screening is recommended going forward, whether dense breast tissue or family history changes that plan, and what long-term measures may help you feel less passive and more supported. This is where the conversation should move beyond fear alone. It should include tissue health, risk reduction, emotional steadiness, and a plan that feels sustainable for years, not just weeks.
The most helpful message for patients and caregivers at this stage is simple. Surgery may have addressed the lesion. It may not have addressed the entire risk environment. That is why follow-up continues, and that is also why many women begin to value an integrative approach more deeply after surgery than before it.
Transition to the Next Step
Once patients understand that risk can remain even after excision, the next question becomes more practical. What can actually be done to reduce that risk and support long-term tissue health in a way that feels active rather than passive? That is where the discussion moves naturally into integrative safety, long-term risk reduction, and the role of a structured Ayurvedic plan alongside medical follow-up.
References
[1] Mayo Clinic. (2024). Atypical hyperplasia of the breast: Diagnosis and treatment. Mayo Clinic.
link: https://www.mayoclinic.org/diseases-conditions/atypical-hyperplasia/diagnosis-treatment/drc-20369778
Brief: Explains that atypical hyperplasia may be managed with surgery, more-frequent screening, and risk-reducing medication, and notes that additional imaging such as breast MRI may be used in selected higher-risk patients.
[2] Johns Hopkins Medicine. (2024). Atypical ductal hyperplasia (ADH). Johns Hopkins Medicine.
link: https://www.hopkinsmedicine.org/health/conditions-and-diseases/breast-cancer/atypical-ductal-hyperplasia
Brief: Describes ADH as a high-risk breast lesion and recommends follow-up in a high-risk breast clinic with discussion of screening, lifestyle, and risk-reduction.
[3] American Society of Breast Surgeons. (2024). Resource guide: Surgical management of benign or high-risk lesions. American Society of Breast Surgeons.
link: https://www.breastsurgeons.org/docs/statements/asbrs-high-risk-lesions.pdf
Brief: States that certain high-risk lesions, including ADH and lobular neoplasia, carry elevated lifetime breast-cancer risk that is not removed by surgical excision alone.
[4] MD Anderson Cancer Center. (2024). Atypical ductal hyperplasia: What it is and how it’s treated. MD Anderson Cancer Center.
link: https://www.mdanderson.org/cancerwise/atypical-ductal-hyperplasia–what-it-is-and-how-to-treat-it.h00-159695967.html
Brief: Explains the pathway from biopsy to excision and notes that patients with ADH are often moved into high-risk screening and prevention discussions if cancer is not found.
[5] National Center for Complementary and Integrative Health. (2023). Cancer and complementary health approaches: What you need to know. U.S. Department of Health and Human Services.
link: https://www.nccih.nih.gov/health/cancer-and-complementary-health-approaches-what-you-need-to-know
Brief: Clarifies that complementary approaches should not replace necessary medical care, but may support well-being and symptom management when used responsibly alongside standard treatment.
Anatomy & Pathophysiology
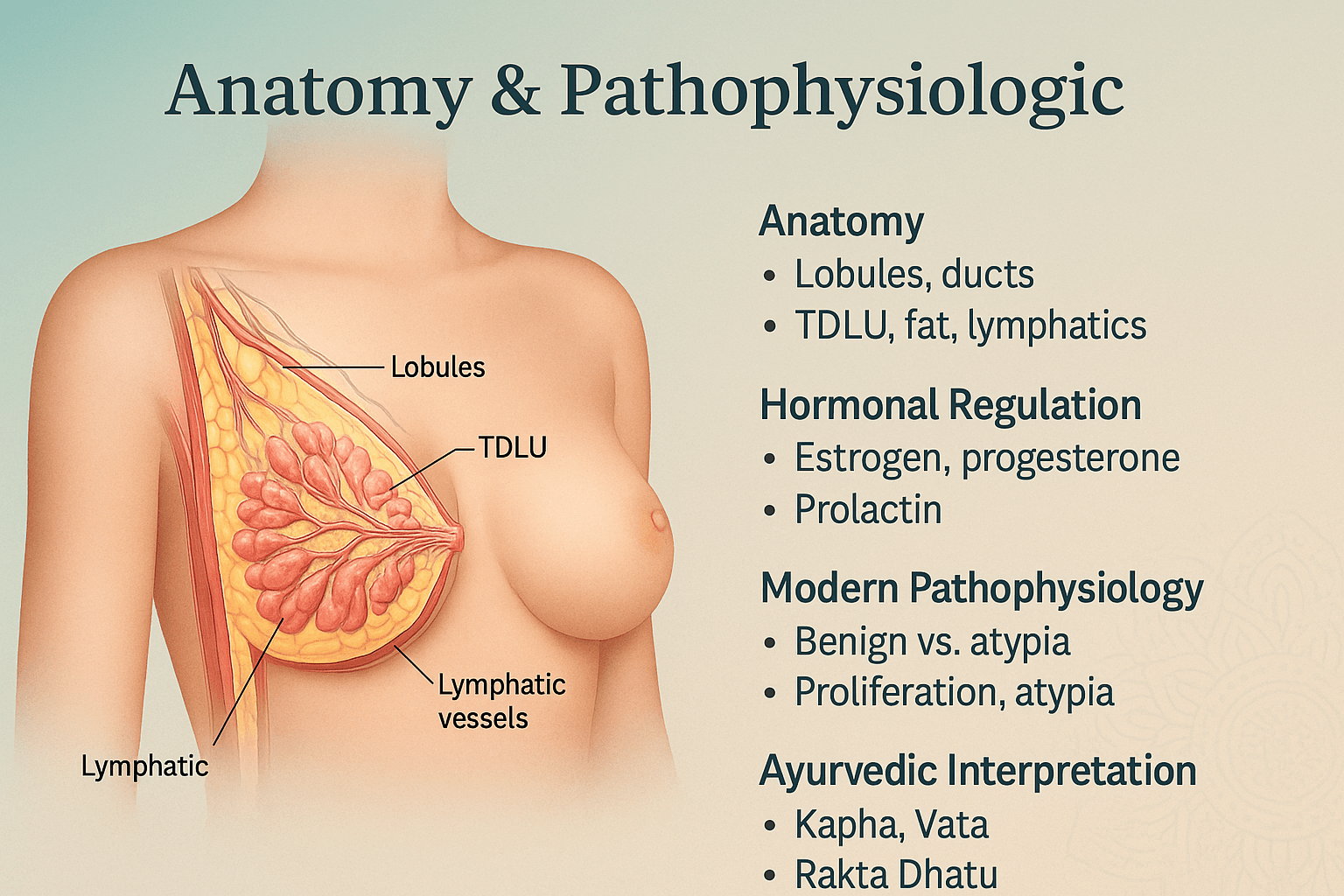
The female breast is a specialized exocrine gland whose primary biological function is to produce and secrete milk for infant nourishment. Structurally, it is composed of glandular tissue, fibrous connective tissue, and adipose tissue, all enveloped within skin and underpinned by the pectoral fascia overlying the pectoralis major muscle [11]. Each breast contains 15–20 lobes, and within each lobe are multiple lobules that house clusters of acini or alveolar glands. These alveoli are lined by secretory epithelial cells and surrounded by contractile myoepithelial cells, which aid in milk expulsion [12].
At the core of breast functionality lies the terminal ductal-lobular unit (TDLU) , a small but vital structure comprising the lobule and its draining terminal duct. This is the origin site for the majority of both benign and malignant breast lesions, making it a key focus of histopathological and radiological evaluation [13]. The TDLU is embedded within a supportive stroma containing collagen, elastic fibers, fibroblasts, immune cells, and a rich microvasculature. Lymphatic drainage from the breast flows predominantly toward the axillary lymph nodes, but also to the internal mammary and supraclavicular nodes, establishing pathways for both immune surveillance and potential disease spread [14].
Hormonal Regulation
Breast tissue is highly hormone-sensitive, undergoing dynamic changes across a woman’s lifespan.
- Estrogen stimulates proliferation of the ductal epithelium and increases stromal vascularity.
- Progesterone induces lobuloalveolar maturation, preparing the gland for potential lactation.
- Prolactin, regulated by the anterior pituitary, drives milk synthesis during lactation.
- Other modulators, such as growth hormone, insulin-like growth factor-1 (IGF-1), and cortisol, influence both developmental and pathological processes [15].
During the menstrual cycle, rising estrogen and progesterone levels in the luteal phase increase stromal edema and lobular cellularity, producing cyclical breast tenderness. These hormonal surges can exaggerate underlying benign lesions, such as cysts or fibroadenomas, causing them to temporarily enlarge [16].
Modern Pathophysiology of Benign & Atypia
Benign breast conditions result from disruptions in the normal balance between cell proliferation, differentiation, and apoptosis.
- Non-proliferative lesions involve minimal cellular growth and include simple cysts, mild fibrous changes, and duct ectasia.
- Proliferative lesions without atypia show increased cell numbers but preserve normal architectural patterns.
- Atypical hyperplasia—whether ductal (ADH) or lobular (ALH)—is characterized by both increased cell numbers and abnormal cytologic features, such as altered nuclear size, shape, and chromatin pattern. These atypical changes indicate a genomic instability that places the patient at 4–5 times higher lifetime risk of developing breast carcinoma compared to women without such findings [17].
At the cellular level, atypical hyperplasia often exhibits:
- Clonal proliferation of epithelial cells with partial resemblance to ductal carcinoma in situ (DCIS)
- Altered cell polarity and irregular luminal architecture
- Upregulated expression of ER/PR receptors and proliferative markers such as Ki-67
- Variable loss of E-cadherin in lobular lesions, indicating changes in cell adhesion [18]
Ayurvedic Interpretation
In Ayurveda, the breast is referred to as Stana and is considered a tissue structure nourished primarily by Rakta Dhatu (blood tissue) and Meda Dhatu (adipose/fat tissue). According to Charaka Samhita and Sushruta Samhita, breast disorders often originate from imbalances in the Kapha Dosha, which governs tissue stability and growth, and Pitta Dosha, which governs metabolic and hormonal processes [19].
Benign proliferative conditions correspond to Kapha Vriddhi (excess tissue accumulation) often coupled with Vata Avarana (obstruction of microchannels, or Srotas). This blockage disrupts the natural circulation of nutrient essence (Rasa Dhatu) and blood (Rakta Dhatu), leading to localized swellings or nodules. Atypical hyperplasia, in Ayurvedic terms, may be conceptualized as Rakta Dhatu Dushti (vitiated blood tissue) , a precursor to more severe Arbuda (tumorous growth) states if left unchecked.
Integrative Relevance
Understanding both the microanatomy and Ayurvedic pathophysiology of the breast helps clinicians make informed decisions about diagnosis, monitoring, and therapy. In modern oncology, this knowledge guides radiologists and pathologists to focus on high-yield anatomical sites such as the TDLU. In Ayurveda, it underpins therapeutic strategies that aim to not only resolve the local lesion but also restore systemic balance, preventing recurrence or progression.
Symptoms
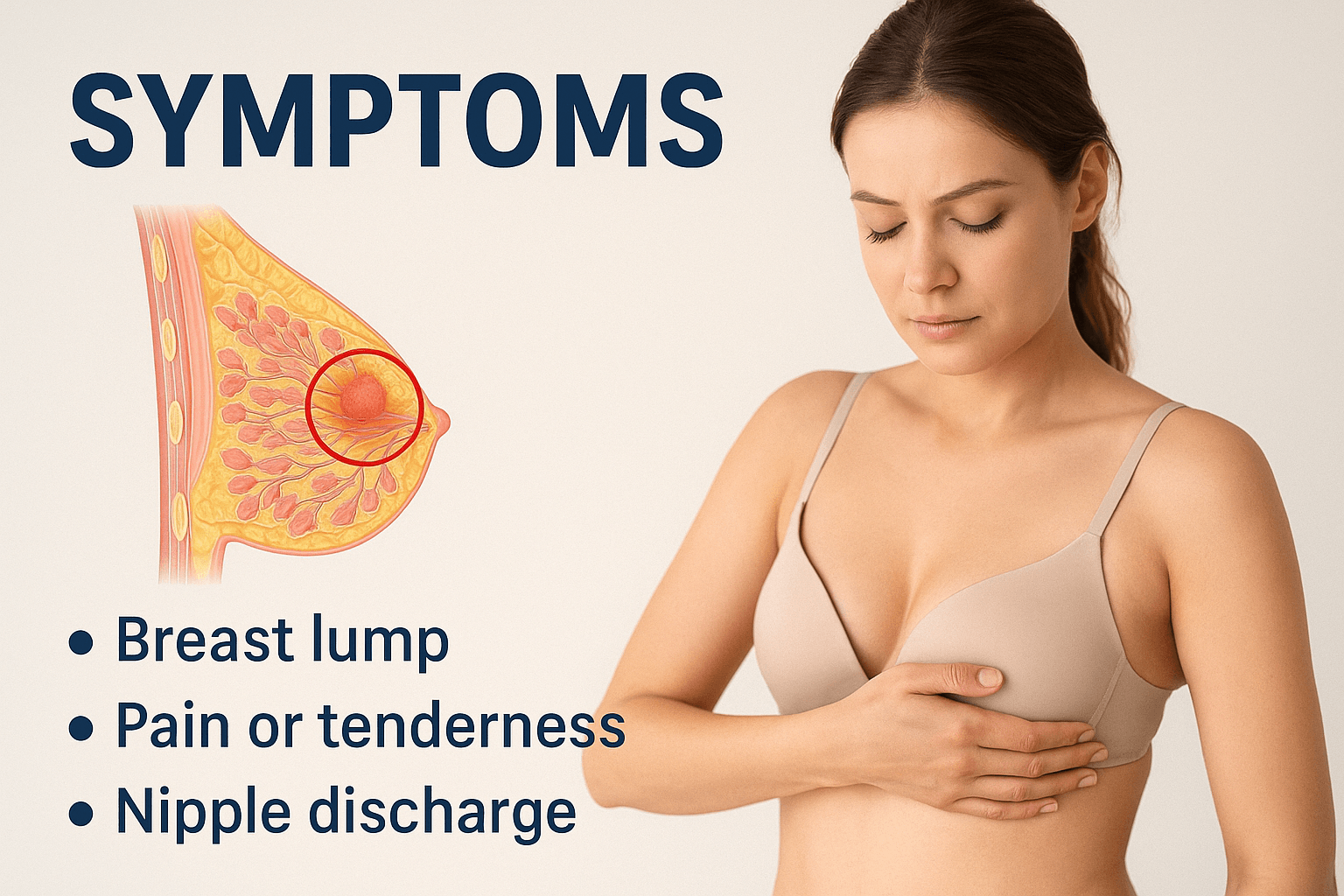
The majority of benign and atypical breast lesions are asymptomatic and detected only on imaging, which is why structured screening programs dramatically reduce late cancer diagnosis. When symptoms do appear, they are usually related to mechanical tissue distortion, cystic fluid pressure, or localized proliferative activity rather than invasive disease. Recognizing the difference between physiological cyclic changes and persistent structural alterations is the first step in accurate clinical triage.
Benign breast conditions and atypical hyperplasia manifest with a spectrum of clinical signs that range from completely silent to overtly symptomatic. While a significant proportion of patients present without any discomfort—particularly in cases detected through routine mammography—others develop symptoms that may mimic those of malignant disease, causing understandable anxiety [20].
Early recognition and accurate interpretation of these symptoms are crucial not only for differential diagnosis but also for determining the urgency of intervention and the choice between surgical versus conservative management [21].
A. Common Symptoms
1. Localized Breast Lump
One of the most frequent presentations is a palpable lump, often described as smooth, well-circumscribed, and mobile under the skin.
- In fibroadenomas, lumps are firm yet elastic, typically ranging from 1–3 cm, and more common in women aged 15–35.
- Cysts may feel fluctuant and can change in size with the menstrual cycle.
- In atypical hyperplasia, the lump may not be easily palpable and is frequently an incidental finding on imaging [22].
2. Breast Pain or Tenderness (Mastalgia)
Pain may be cyclical (linked to the luteal phase of the menstrual cycle) or non-cyclical.
- Cyclical mastalgia is often bilateral and diffuse, whereas non-cyclical pain is localized and may suggest ductal or lobular pathology.
- Pain alone is rarely a sign of malignancy but significantly affects quality of life [23].
3. Nipple Discharge
A hallmark symptom in certain ductal lesions.
- Benign presentations: clear, greenish, or milky discharge (galactorrhea).
- Atypia concern: spontaneous, unilateral, and bloody discharge should prompt immediate ductal evaluation [24].
4. Generalized Lumpiness
A rope-like or granular feel to the breast tissue, particularly in women with fibrocystic changes, which often fluctuates with hormonal cycles [25].
B. Less Common / Often Missed Symptoms
1. Sudden Change in Lump Size
Rapid growth after menstruation, pregnancy, or hormone therapy should be evaluated for proliferative pathology such as phyllodes tumor or atypia [26].
2. Persistent Itching or Skin Texture Changes Without Redness
This can be an early marker of dermal lymphatic congestion or low-grade inflammatory changes [27].
3. Burning or Tingling Sensation Without Visible Lump
May arise from nerve irritation due to ductal distension or stromal edema, particularly in the luteal phase [28].
4. Subtle Nipple Changes
Mild inversion, retraction, or deviation of the nipple axis, even without mass, may signal underlying fibrosis or ductal scarring [29].
5. Localized Warmth Without Infection
Occasional benign lesions can cause focal hyperemia due to hormonal vascular responses [30].
C. Ayurvedic Symptom Correlation
Ayurveda interprets these clinical signs in the context of Dosha imbalances and Dhatu vitiation:
- Kapha-Dominant:
- Heavy sensation in the breast (Gurutva),
- Painless, slow-growing lump (Kapha Vriddhi),
- Increased breast size during certain seasons like Hemanta Ritu (winter).
- Heavy sensation in the breast (Gurutva),
- Pitta-Dominant:
- Local heat (Ushna),
- Redness, mild tenderness, occasional burning,
- Associated menstrual aggravation due to Rakta Dushti (vitiated blood tissue).
- Local heat (Ushna),
- Vata-Dominant:
- Irregular lump margins,
- Variable consistency,
- Pain aggravated at night or in cold weather (Shita Kala),
- Sudden size fluctuation due to Vata Avarana (obstructed movement in channels).
- Irregular lump margins,
Classical Ayurvedic texts such as Sushruta Samhita, Nidana Sthana, and Charaka Samhita, Chikitsa Sthana, describe such conditions under Stana Granthi (benign glandular swellings) and Arbuda Moolavastha (pre-tumorous state), emphasizing early intervention to prevent chronicity [31].
D. Lesser-Known Patient-Reported Observations
Patients with benign or atypical breast lesions sometimes describe subtle sensations or changes that are rarely highlighted in standard oncology literature:
- “Heaviness that disappears after menstruation” — often linked to hormonal breast congestion.
- “A moving bubble sensation” — sometimes due to cystic fluid shifts.
- “Itchy breast skin without rash” — mild lymphatic irritation or hormonal sensitivity.
- “Pain with arm movement” — linked to lesions located near the axillary tail of Spence.
- “Feeling of fullness when stressed” — psychoneuroendocrine interaction influencing vascular tone in breast tissue.
Recognizing these subjective experiences can help in earlier detection and more accurate patient reassurance.
E. Clinical Significance
While many benign breast symptoms require only observation, a subset , particularly spontaneous unilateral nipple discharge, progressive enlargement, and nipple axis deviation, require detailed evaluation. This involves clinical breast examination, imaging (mammography, ultrasound, or MRI), and histopathology when indicated.
Ayurvedic assessment adds another layer by detecting Dosha imbalance through Nadi Pariksha, correlating systemic imbalances with local breast changes, and enabling preventive interventions before pathological progression [32].
A painless breast lump does not automatically indicate cancer. Most palpable breast masses, especially in premenopausal women, are benign. However, any new lump that persists beyond one menstrual cycle requires imaging evaluation to establish structural diagnosis and BI-RADS classification.
Classification
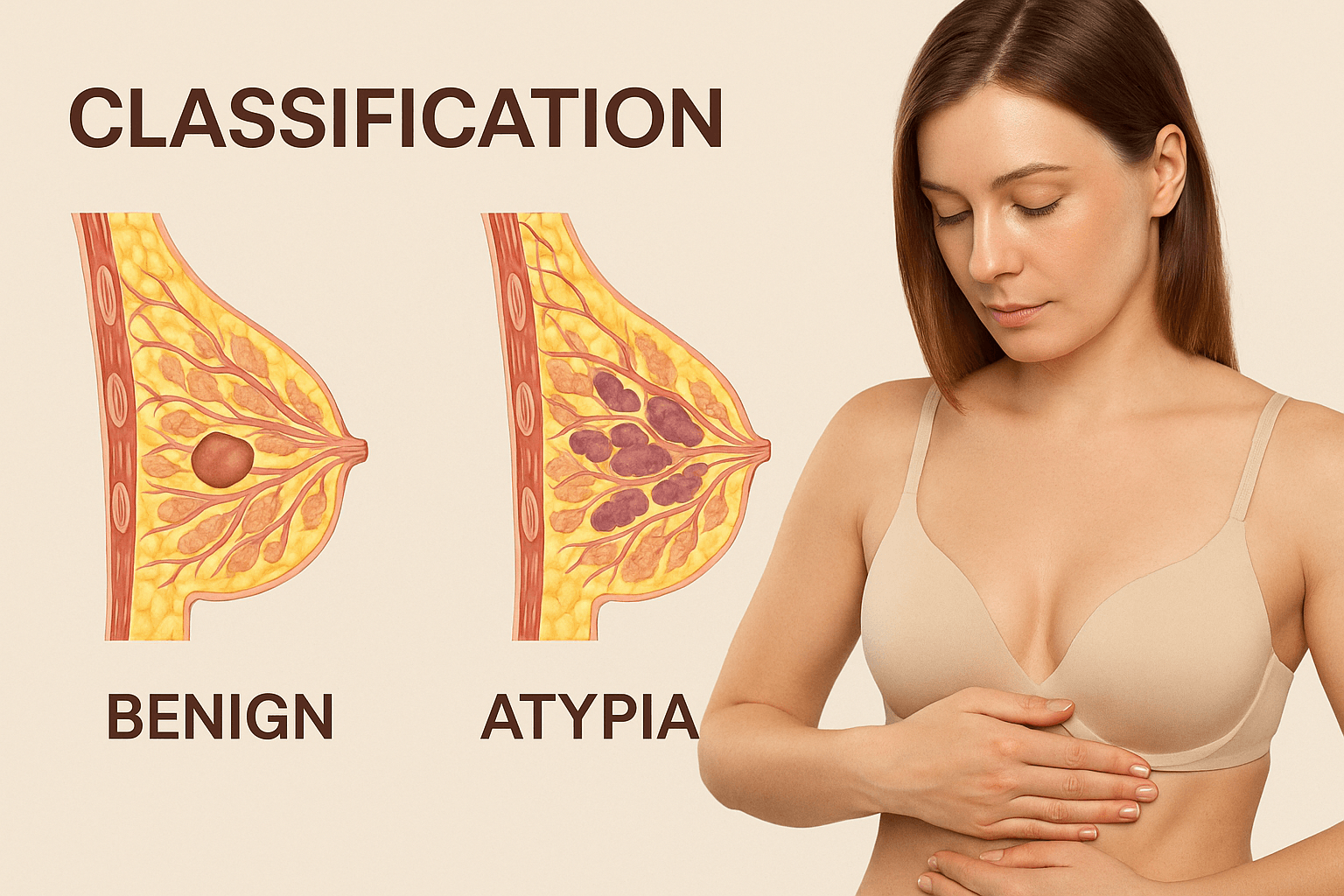
Benign breast conditions represent a wide histopathological spectrum, ranging from simple physiological changes to high-risk proliferative disorders that can serve as precursors to malignancy. Proper classification allows clinicians to assess cancer risk, tailor surveillance strategies, and select the most appropriate management plan for each patient. From a clinical and pathological standpoint, these lesions are typically divided into three major categories: non-proliferative lesions, proliferative lesions without atypia, and proliferative lesions with atypia [33].
Non-proliferative lesions
These are the most common benign breast abnormalities and are characterized by minimal or no increase in epithelial cell proliferation. Cellular architecture remains essentially normal, and there is generally no significant increase in the risk of developing breast cancer. They often arise from physiological hormonal responses and may fluctuate with the menstrual cycle.
Common examples include:
- Simple breast cysts – fluid-filled sacs that develop within lobules, usually presenting as smooth, well-circumscribed lumps, sometimes tender premenstrually.
- Fibrocystic changes – a combination of fibrosis and cyst formation, producing a lumpy or rope-like texture.
- Duct ectasia – dilatation and thickening of large ducts near the nipple, often causing green or brown sticky discharge.
- Mild ductal hyperplasia – an increase in ductal epithelial cells without architectural distortion, usually detected incidentally on biopsy [34][35].
Proliferative lesions without atypia
These conditions involve an increased number of epithelial cells lining ducts or lobules, yet the cells retain their normal size, shape, and nuclear characteristics. They are associated with a modest increase in lifetime breast cancer risk, approximately 1.5–2 times higher than baseline. Although they are not premalignant in themselves, their presence signals a breast tissue environment with increased mitotic activity and potentially heightened sensitivity to hormonal stimulation.
Representative examples:
- Usual ductal hyperplasia – proliferation of ductal epithelial cells producing irregular ductal spaces, often discovered on screening biopsies.
- Intraductal papilloma – a small, wart-like growth inside a duct, which may present with clear or blood-tinged nipple discharge.
- Sclerosing adenosis – lobular enlargement with increased numbers of acini and stromal fibrosis, which can mimic carcinoma on imaging due to architectural distortion [36].
Proliferative lesions with atypia
This category carries the highest relative risk among benign lesions, with a four- to fivefold increase in the likelihood of developing breast cancer compared to the general population. These lesions are histologically notable for both increased epithelial proliferation and cytological abnormalities such as nuclear enlargement, irregular chromatin, and loss of typical cellular organization. Many pathologists consider these lesions as precursors to carcinoma in situ if left untreated.
Key types include:
- Atypical ductal hyperplasia – features partially resembling ductal carcinoma in situ (DCIS) but lacking the full extent or distribution required for a carcinoma diagnosis.
- Atypical lobular hyperplasia – abnormal proliferation within the lobules, often found incidentally in biopsies performed for other reasons.
- Flat epithelial atypia – early neoplastic change in columnar epithelial cells, sometimes associated with calcifications on mammography [37][38].
Special situations
Certain benign but complex lesions do not fit neatly into the above categories yet require special attention due to their diagnostic challenges.
- Radial scar (complex sclerosing lesion) – a central fibroelastic core with radiating ducts and lobules, which can closely resemble invasive carcinoma on imaging and histology.
- Papillomatosis – multiple small papillomas within the ductal system, which may coexist with areas of atypia or carcinoma in situ.
These lesions often necessitate excisional biopsy to exclude malignancy [38].
Ayurvedic correlation
In Ayurvedic medicine, benign breast conditions are grouped under the broader category of stana granthi, or glandular swellings of the breast. Based on the dominant dosha and dhatu involvement, they can be described in three progressive stages:
- Kapha-predominant granthi – slow-growing, well-circumscribed, painless masses, reflecting excess structural tissue growth (kapha vriddhi).
- Rakta dushti–associated granthi – lumps that become tender, warmer, and occasionally change in size with the menstrual cycle, corresponding to blood tissue vitiation.
- Vata–kapha granthi – irregular, mobile lumps with intermittent or sharp pain, often fluctuating in size due to obstructed movement of vata within the breast channels (srotas).
The Ayurvedic understanding parallels modern pathology’s progression from non-proliferative to atypical proliferative lesions. Kapha predominance reflects stable benign lesions, while the involvement of rakta dushti and vata imbalance indicates increasing structural irregularity and instability in tissue architecture [39].
Diagnosis
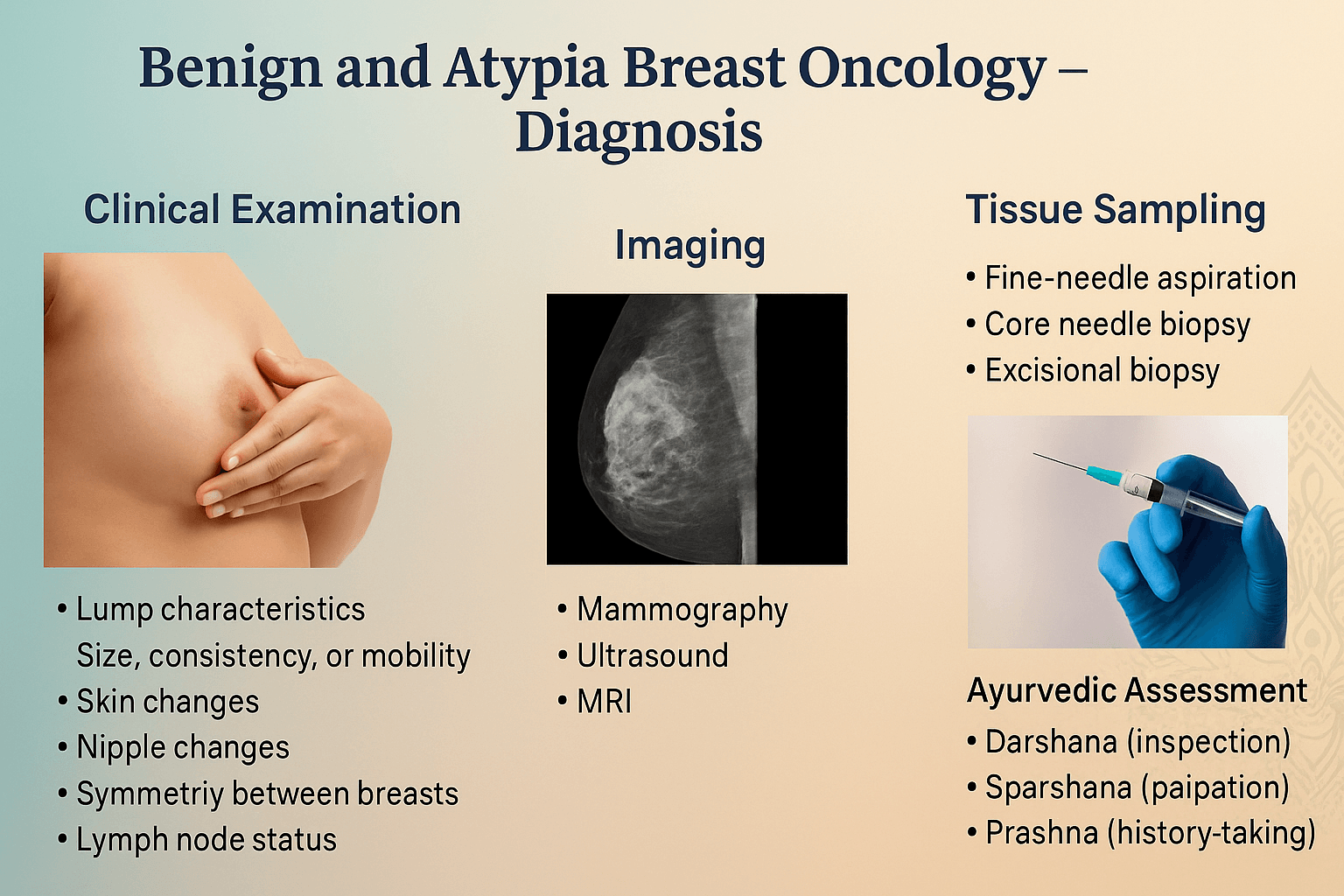
The modern diagnostic pathway for benign and atypical breast conditions follows a triple-assessment model consisting of clinical examination, imaging correlation, and histopathological confirmation. This structured approach ensures that lesions are not overtreated surgically while also preventing under-diagnosis of high-risk proliferative disease. Imaging assigns a BI-RADS score, which determines whether surveillance, short-interval follow-up, or biopsy is required.
The diagnosis of benign and atypical breast lesions requires a structured approach that integrates patient history, physical examination, imaging, and tissue sampling. The goal is to differentiate harmless conditions from high-risk or malignant disease, avoid unnecessary interventions, and ensure timely treatment when warranted [40].
Clinical history and examination
A thorough medical history should cover symptom onset, duration, menstrual correlation, history of trauma, lactation, prior breast biopsies, hormonal therapy, family history of breast cancer, and relevant comorbidities such as thyroid disorders or metabolic syndrome [41].
During breast examination, the clinician assesses:
- Lump location, size, consistency, and mobility.
- Skin changes including dimpling, erythema, or thickening.
- Nipple changes such as inversion, retraction, or discharge.
- Symmetry between breasts.
- Axillary and supraclavicular lymph node status [42].
For accurate assessment, examination is best performed in both the sitting and supine positions, and in premenopausal women, ideally within the first week after menstruation when hormonal swelling is minimal.
Imaging modalities
Mammography – The standard screening tool for women over 40, providing high-resolution images that can detect microcalcifications, architectural distortions, and subtle masses. Findings are reported using the BI-RADS (Breast Imaging-Reporting and Data System) classification, which guides next steps in management [43].
Ultrasound – Particularly valuable for younger women with dense breast tissue and for distinguishing cystic from solid lesions. It allows real-time guidance for fine-needle aspiration or core biopsy.
Magnetic resonance imaging (MRI) – Highly sensitive for detecting multifocal or multicentric disease and evaluating the extent of atypical lesions, especially in women at high genetic risk. Gadolinium-enhanced MRI can reveal abnormal vascular patterns suggestive of malignancy [44].
Digital breast tomosynthesis (3D mammography) – Improves lesion detection and reduces recall rates, especially in dense breasts.
Tissue diagnosis
Histopathological confirmation is crucial for any lesion with suspicious imaging or clinical features.
- Fine-needle aspiration cytology (FNAC) – Quick, minimally invasive, but less accurate in differentiating atypia from carcinoma.
- Core needle biopsy – Preferred for most solid lesions as it preserves tissue architecture, allowing accurate grading of atypia and assessment of margins.
- Vacuum-assisted biopsy – Useful for complete removal of small papillomas or for sampling microcalcifications.
- Excisional biopsy – Reserved for lesions where imaging and core biopsy results are inconclusive, or when high-risk pathology is found [45].
- After a biopsy confirms a benign or atypical lesion, management is no longer uniform but risk-based. The patient enters a personalized surveillance pathway that may include annual mammography, supplemental MRI for dense breasts or high lifetime risk, clinical breast examination at defined intervals, and discussion of preventive endocrine therapy in selected cases. This phase is preventive oncology rather than cancer treatment.
Ancillary studies
Immunohistochemistry can differentiate atypical ductal hyperplasia from low-grade ductal carcinoma in situ, using markers like E-cadherin, CK5/6, and ER/PR receptors. Hormone receptor testing also aids in risk assessment and therapeutic planning [46].
Ayurvedic diagnostic approach
In Ayurveda, diagnosis involves both local and systemic assessment.
- Darshana (inspection) – Observing breast shape, skin texture, and any visible swelling or color change.
- Sparshana (palpation) – Evaluating the size, consistency, tenderness, and mobility of the lump to identify dosha predominance.
- Prashna (history-taking) – Determining dietary habits, menstrual regularity, stress levels, and prior illnesses that may have contributed to dosha imbalance.
- Nadi Pariksha (pulse examination) – Detects vitiation of Kapha, Pitta, and Vata, as well as subtle srotas (channel) obstruction that may underlie benign or atypical changes [47]
Integrated diagnostic relevance
While modern imaging and pathology provide definitive structural and cellular diagnosis, Ayurvedic examination offers insight into constitutional tendencies and systemic imbalances that predispose to these lesions. Combining both approaches supports not only accurate classification but also personalized preventive and therapeutic strategies [48].
In Western clinical practice, most benign and high-risk breast lesions are detected through structured screening pathways such as routine mammography, GP referral to breast clinics, or high-risk surveillance programs. Women with dense breasts, family history, or prior atypical biopsy are often placed in annual imaging protocols that may include digital mammography, tomosynthesis, ultrasound, or breast MRI depending on calculated lifetime risk. This risk-based model is central to modern prevention and allows intervention years before malignant transformation occurs.
Risk Stratification
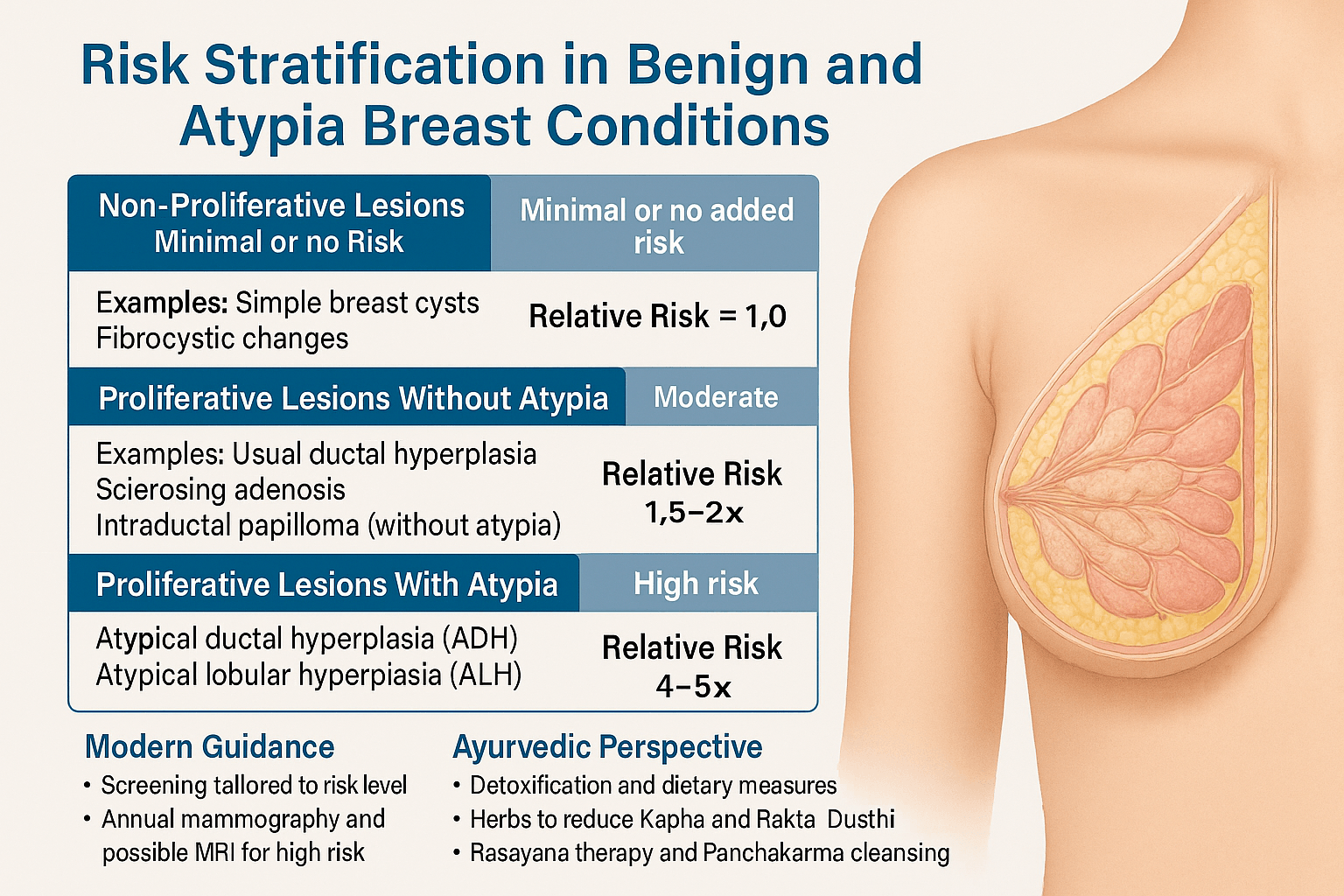
Not all benign breast diseases carry the same future risk. Non-proliferative lesions have no significant increase in cancer incidence, usual ductal hyperplasia carries a mild elevation, and atypical ductal or lobular hyperplasia increases lifetime breast cancer risk by approximately four to five times. Risk models such as the Gail model, Tyrer–Cuzick score, and BOADICEA calculator are used in high-risk clinics to determine surveillance intensity and preventive strategies.
Risk stratification in benign and atypical breast conditions is the process of estimating how likely a woman is to develop breast cancer in the future, based on the type of lesion present, personal medical history, family history, and genetic background. This classification is essential because it directly influences how closely a patient should be monitored, what preventive strategies may be appropriate, and how her care plan should be personalized [49].
Non-proliferative lesions – Minimal or no added risk
Non-proliferative lesions include simple breast cysts, fibrocystic changes, and mild ductal hyperplasia. Histologically, these conditions show either normal cell turnover or only a slight increase in epithelial proliferation without any abnormal nuclear features. Large epidemiologic studies, such as the Mayo Benign Breast Disease Cohort, have consistently found that these lesions carry a relative risk close to 1.0, meaning the lifetime breast cancer risk is similar to that of the general female population [50].
For these patients, standard population-based screening is appropriate. Routine mammography according to national guidelines (often every 1–2 years for women over 40) is sufficient, along with monthly breast self-awareness. Lifestyle measures that promote hormonal balance, such as maintaining a healthy weight, managing stress, and avoiding unnecessary hormone therapy, are encouraged.
From an Ayurvedic perspective, these cases often correspond to a state of Kapha stability, where tissue growth and metabolic activity remain in balance. The focus here is on preventive maintenance, seasonal detoxification (Shodhana), light oil massage (Abhyanga) to maintain Srotas (microchannel) patency, and dietary patterns that prevent Kapha aggravation [55].
Proliferative lesions without atypia – Moderate increase in risk
This group includes usual ductal hyperplasia (UDH), sclerosing adenosis, and intraductal papillomas without atypia. These lesions are characterized by an increase in the number of epithelial cells lining the ducts or lobules, yet the cells retain normal nuclear and architectural features. Several longitudinal studies have shown these lesions to be associated with a 1.5–2-fold increase in breast cancer risk compared to the general population [51].
Although these are not considered precancerous, their presence signals an active breast tissue environment with higher cell turnover, possibly influenced by hormonal sensitivity. In clinical practice, this group benefits from closer surveillance, annual rather than biennial mammography is often advised, and breast ultrasound is valuable for women with dense breast tissue. Lifestyle modification plays a significant role: limiting alcohol intake, avoiding long-term hormone replacement therapy unless essential, and supporting metabolic health.
In Ayurveda, such lesions often reflect early Kapha Vriddhi (increase) along with mild Rakta Dushti (blood tissue vitiation). The recommended approach includes herbal formulations like Kanchnar Guggulu for reducing abnormal tissue proliferation, Triphala Guggulu for detoxification, and dietary measures that pacify Kapha while purifying Rakta. Regular exercise and seasonal detox therapies help maintain dosha balance [55].
Proliferative lesions with atypia – High-risk category
Atypical ductal hyperplasia (ADH) and atypical lobular hyperplasia (ALH) represent the most significant risk among benign breast lesions. Histologically, these lesions show both increased cellular proliferation and abnormal nuclear or architectural patterns that partially resemble carcinoma in situ, though they lack invasive properties. Multiple studies indicate that these conditions confer a 4–5 times higher lifetime risk of developing invasive breast cancer compared to women without such findings [52].
This elevated risk is bilateral, meaning cancer can develop in either breast, not just the one with the lesion. Importantly, the risk persists for decades after diagnosis. Management typically involves annual mammography, consideration of supplemental MRI in women with a calculated lifetime risk over 20%, discussion of chemoprevention (e.g., tamoxifen or raloxifene), and selective genetic counseling for those with a strong family history. Surgical excision of the atypical area is often advised to rule out adjacent carcinoma.
In Ayurveda, atypia corresponds to a pronounced Kapha–Pitta aggravation combined with significant Rakta Dushti. The treatment approach here is more intensive, structured Panchakarma cleansing to reset systemic balance, Rasayana therapies such as Abhrak Bhasma and Swarna Bhasma (under specialist supervision) for cellular repair, and a strict pathya–apathya (dietary and lifestyle regimen) to prevent recurrence or progression. Psychological counseling, meditation, and stress control are also emphasized to address the mind–body link in cancer susceptibility [55].
Family history and genetics – Risk amplification
A strong family history, particularly a first-degree relative with breast cancer, increases the baseline risk in all three categories. In women with atypia combined with such a family history, the lifetime risk can reach 25–30% [53]. While BRCA1 and BRCA2 mutations are rare in women with purely benign disease, genetic counseling and testing are warranted when family history suggests hereditary breast and ovarian cancer syndromes.
Risk prediction models such as the Gail Model, Tyrer–Cuzick Model, and BOADICEA incorporate age, reproductive history, histologic findings, and family history to calculate individual 5-year and lifetime risk estimates. Women with a calculated lifetime risk above 20% are generally considered for supplemental MRI in addition to mammography [54].
Atypical hyperplasia is not cancer, but it is one of the strongest predictors of future breast cancer. With proper surveillance and preventive care, progression can often be avoided.
Associated Disorders and Systemic Correlations

Benign and atypical breast lesions are rarely isolated local findings. In both preventive oncology and the Ayurvedic understanding of Stana Roga, these changes often reflect deeper endocrine, metabolic, hepatic, lymphatic, and immune dysregulation. Identifying these associations is essential because they influence recurrence risk, progression potential, mammographic density, inflammatory signaling, and response to long-term preventive strategies.
Hormonal imbalance and estrogen dominance
Altered estrogen–progesterone balance is one of the most consistent associations. Women with anovulatory cycles, polycystic ovarian syndrome, luteal phase defects, early menarche, late menopause, or prolonged exposure to exogenous hormones frequently develop proliferative breast changes. Continuous estrogenic stimulation promotes ductal epithelial growth and stromal fibrosis, which clinically manifest as fibrocystic disease, usual ductal hyperplasia, and atypical hyperplasia. From an Ayurvedic perspective this reflects Kapha–Pitta vitiation with Meda and Rakta Dhatu dushti.
Thyroid dysfunction
Hypothyroidism is commonly observed in patients with cyclical mastalgia, nodular breast tissue, and fibrocystic disease. Reduced thyroid function alters estrogen metabolism, increases prolactin sensitivity, and promotes interstitial fluid retention, leading to breast density and persistent tenderness. Subclinical hypothyroidism is particularly significant because it may remain undiagnosed while continuously driving breast tissue proliferation.
Polycystic ovarian syndrome
Polycystic ovarian syndrome creates a metabolic and endocrine environment characterized by chronic anovulation, hyperinsulinemia, and reduced sex hormone–binding globulin. This leads to increased bioavailable estrogen and proliferative breast changes. In Ayurvedic correlation this represents Kapha-Meda aggravation with Artava Dushti and impaired Dhatu Agni.
Obesity and metabolic syndrome
In postmenopausal women, adipose tissue becomes the primary site for estrogen production through aromatization. This results in continuous low-grade estrogen exposure and chronic inflammatory signaling, both of which increase the likelihood of atypical hyperplasia. Insulin resistance further activates IGF-1 pathways that stimulate epithelial proliferation and stromal fibrosis, making metabolic correction central to recurrence prevention.
Chronic liver dysfunction
The liver is responsible for estrogen metabolism and detoxification. Non-alcoholic fatty liver disease, drug-induced hepatic stress, and impaired metabolic clearance lead to circulating estrogen excess and persistent fibrocystic changes. In Ayurvedic interpretation this corresponds to Yakrit Dushti and Ranjaka Pitta imbalance affecting Rakta Dhatu quality.
Hyperprolactinemia
Elevated prolactin levels are associated with mastalgia, duct ectasia, cyst formation, and nipple discharge. Even mild prolactin elevation can create a proliferative glandular environment and long-standing breast congestion.
Less Common but Clinically Significant Associations

Autoimmune disorders
Autoimmune thyroid disease, rheumatoid arthritis, systemic lupus erythematosus, and Sjögren syndrome are associated with higher breast density and chronic inflammatory mastalgia. Persistent immune activation alters stromal signaling and epithelial turnover, increasing the likelihood of proliferative lesions.
Diabetes mellitus and insulin resistance
Chronic hyperinsulinemia promotes cellular proliferation through insulin-like growth factor pathways and is associated with increased breast density and higher atypia risk. Advanced glycation end products further contribute to stromal fibrosis and microvascular compromise.
Chronic stress and HPA-axis dysregulation
Long-standing cortisol imbalance alters gonadotropin release, ovarian function, prolactin response, and inflammatory cytokine activity. This produces cyclical mastalgia, breast congestion, and nodularity without a dominant structural lesion and is a major factor in recurrent benign breast symptoms.
Dense breast tissue
Extremely dense breast tissue is an independent risk factor for proliferative disease and interval cancers. It also increases the frequency of benign biopsies due to reduced imaging sensitivity and represents a hormonally active stromal environment.
Iodine deficiency
Low iodine status increases breast tissue sensitivity to estrogen and is associated with fibrocystic changes, nodularity, and persistent breast discomfort. This becomes clinically relevant in patients with coexisting thyroid dysfunction.
Rare but High-Significance Associations
Genetic cancer predisposition syndromes
In patients with strong family history, atypical hyperplasia may be a phenotypic marker of underlying mutations such as BRCA1, BRCA2, PALB2, CHEK2, or ATM. In such cases the lesion represents genomic instability rather than purely hormonal proliferation and requires high-risk surveillance protocols.
Chronic lymphatic congestion
Impaired lymphatic drainage due to prior surgery, radiation exposure, or prolonged sedentary lifestyle produces persistent nodularity, localized inflammatory changes, and breast heaviness, reflecting structural fluid stasis rather than true tumor formation.
Clinical Significance
Recognizing these systemic associations transforms management from local lump observation to comprehensive risk correction. When endocrine, metabolic, hepatic, immune, and lymphatic factors are addressed alongside structured imaging surveillance, the biological drivers of proliferative breast disease can be significantly reduced and long-term tissue stability improved.
What Should You Do After a Benign or Atypical Breast Diagnosis?

A diagnosis of a benign or atypical breast lesion is not the end of the evaluation. It is the beginning of a structured prevention and surveillance phase. The next step depends on the biopsy result, individual cancer risk, age, breast density, family history, and hormonal status. The goal is to prevent progression while avoiding unnecessary surgery.
If the lesion is non-proliferative
Non-proliferative findings such as simple cysts, mild fibrocystic change, or apocrine metaplasia usually require no surgical treatment. These patients enter a routine screening program with annual mammography after the age recommended for their country and periodic clinical breast examination. Symptom-directed follow-up is sufficient if the imaging and pathology are concordant.
If usual ductal hyperplasia or papilloma without atypia is reported
These lesions carry a mild increase in long-term cancer risk. Imaging follow-up is typically advised at regular intervals, and maintaining breast awareness becomes important. In patients with dense breasts, supplemental ultrasound may be recommended. The focus at this stage is risk reduction through metabolic, hormonal, and lifestyle correction.
If atypical ductal or lobular hyperplasia is diagnosed
Atypical hyperplasia places the patient in a high-risk surveillance category. Annual mammography is required, and in women with a calculated lifetime risk above the accepted threshold, breast MRI may be added. A detailed risk assessment using validated models helps determine whether preventive endocrine therapy should be discussed. Surgical excision may be advised if the core biopsy and imaging findings are not fully concordant.
If the biopsy result and imaging do not match
When radiology and pathology are discordant, excisional biopsy is recommended to ensure that a more advanced lesion has not been missed. This step is critical for patient safety and should not be delayed.
If there is a strong family history of breast or ovarian cancer
Patients with multiple affected relatives, early onset cancers in the family, or bilateral disease should undergo formal genetic risk evaluation. Identification of hereditary cancer syndromes changes both screening frequency and preventive strategy and may include MRI-based surveillance at an earlier age.
If the breasts are extremely dense
Dense breast tissue reduces mammographic sensitivity and independently increases cancer risk. These patients benefit from personalized screening protocols that may include tomosynthesis, ultrasound, or MRI depending on overall risk profile.
If the patient is premenopausal with cyclical breast symptoms
Cyclical pain and nodularity often reflect hormonal fluctuation rather than structural progression. These patients require symptom management, endocrine balance, and periodic imaging rather than repeated invasive procedures.
If the patient is postmenopausal
Any new palpable mass in a postmenopausal woman requires prompt imaging evaluation even if prior reports were benign, because background hormonal protection has declined.
Long-term prevention strategy
Once the immediate diagnosis is clarified, the patient moves into a long-term prevention plan. This includes maintaining optimal body composition, improving insulin sensitivity, regulating hormonal metabolism, supporting hepatic estrogen clearance, ensuring adequate micronutrient status, and following a structured imaging schedule. This phase is where progression risk can be significantly reduced.
Clinical Decision Pathway After a Benign or Atypical Breast Diagnosis
| Role of Integrative Risk Reduc | Future Cancer Risk | Future Cancer Risk | Role of Integrative Risk Reduction |
|---|---|---|---|
| Non-proliferative lesion (simple cyst, mild fibrocystic change) | No significant increase | Routine age-appropriate mammography and clinical breast exam | Hormonal balance, metabolic optimization, liver support for estrogen clearance |
| Usual ductal hyperplasia (UDH) | Mild increase (1.5–2×) | Annual imaging; breast awareness; ultrasound if dense breast | Weight regulation, insulin sensitivity, anti-inflammatory diet, lymphatic circulation support |
| Papilloma without atypia | Slight increase | Imaging surveillance; excision only if symptomatic or discordant | Prolactin regulation, ductal congestion reduction, endocrine stabilization |
| Atypical ductal hyperplasia (ADH) | Moderate to high (4–5×) | Annual mammography ± MRI; risk assessment models; consider preventive endocrine therapy | Tissue microenvironment correction, Kapha–Pitta modulation, immune surveillance support |
| Atypical lobular hyperplasia (ALH) | Moderate to high (4–5×, bilateral risk) | High-risk screening protocol; long-term surveillance | Systemic metabolic correction and Rasayana-based tissue resilience |
| Radiology–pathology discordance | Uncertain until excision | Surgical excision recommended | Post-diagnostic recovery and recurrence prevention strategies |
| Strong family history / genetic predisposition | High lifetime risk | Genetic counseling; MRI-based screening; earlier surveillance | Long-term inflammation control and Dhatu-level nourishment |
| Extremely dense breast tissue | Independent risk factor | Tomosynthesis, ultrasound, or MRI based on risk score | Hormonal metabolism correction and Meda Dhatu regulation |
| Premenopausal cyclical nodularity with benign imaging | Low structural risk | Symptom-based follow-up; avoid repeated invasive procedures | Cycle regulation, Apana Vata balance, stress axis stabilization |
| New palpable lump in postmenopausal patient | Higher baseline risk | Immediate diagnostic imaging and biopsy if indicated | Supportive care after structural diagnosis |
Allopathy (Conventional Treatment)

Conventional treatment for a benign breast lump or atypical hyperplasia is very different from the treatment used for confirmed breast cancer. In most cases, the goal is not chemotherapy or radiation. The real aim is to answer four practical questions clearly: was the lesion sampled accurately, do the imaging and pathology findings match, does the area need surgical removal, and does the patient now need long-term high-risk surveillance or risk-reducing medication. If a later excision finds ductal carcinoma in situ or invasive cancer, then treatment shifts into true cancer management. Until that point, conventional care is mainly about diagnostic accuracy, safe decision-making, and future risk reduction.
Diagnosis Confirmation Comes First
For most patients, treatment really begins with confirming what the lesion is. Atypical hyperplasia is usually found after something suspicious appears on mammogram, ultrasound, or clinical examination and a biopsy is performed. The American Society of Breast Surgeons states that percutaneous core needle biopsy is the preferred initial minimally invasive diagnostic step, and that radiologic-pathologic concordance must be assessed afterward. In simple terms, the pathology result must make sense in light of what the imaging showed. If the biopsy result does not adequately explain the imaging abnormality, further tissue sampling or surgical excision is usually recommended. This is one of the most important realities for patients to understand, because many people think the biopsy label alone decides treatment. In practice, the imaging-biopsy match is just as important.
Observation Is a Conventional Option in Selected Cases
Not every benign or atypical lesion goes straight to surgery. Mayo Clinic states clearly that not everyone with atypical hyperplasia needs surgery, especially when the risk of finding cancer is considered low based on mammogram findings, biopsy features, medical history, and prior breast procedures. The American Society of Breast Surgeons also recommends selective rather than routine excision for many high-risk lesions once concordance has been established. For some concordant lesions under observation, diagnostic imaging at 6, 12, and 24 months may be used to establish stability. This is why some patients are told to monitor carefully instead of proceeding directly to the operating room. To the patient, that can feel uncertain. To the clinical team, it often reflects a lower-risk pathway rather than neglect.
Surgery Is Used When More Certainty Is Needed
Surgical excision is commonly advised when the imaging and biopsy findings do not match, when the biopsy sample may not have captured enough tissue, or when the lesion type carries a higher chance of being “upgraded” to ductal carcinoma in situ or invasive cancer after a larger sample is examined. Mayo Clinic notes that surgery may be recommended if mammogram images remain concerning, and MD Anderson explains that when atypical ductal hyperplasia is found on core biopsy, the first step is usually an excisional biopsy because a core biopsy removes only a small amount of tissue. The tissue removed during surgery is then checked for hidden cancer. Mayo also notes that most people who undergo surgery for atypical hyperplasia do not turn out to have breast cancer, but some do, which is exactly why excision is recommended in selected cases.
Conventional Treatment Is Different for Different Lesions
One reason patients feel confused is that “benign” does not mean all lesions are treated the same way. The American Society of Breast Surgeons recommends routine surgical excision for atypical ductal hyperplasia in most cases, although selected low-risk patients may avoid surgery. For some other concordant lesions, management is more selective. Flat epithelial atypia may sometimes be observed in carefully chosen patients, especially when most of the target calcifications were removed on biopsy. Papillary lesions with atypia are generally excised, while management of papillomas without atypia depends on symptoms and sampling. Radial scars or complex sclerosing lesions without atypia may be observed in some settings, but lesions with atypia are more often excised because the upgrade risk is higher. This is why two women can both hear the word “atypia” and still receive different recommendations.
High-Risk Follow-Up Is Part of Conventional Care
If surgery confirms atypical hyperplasia but not cancer, conventional treatment usually moves into surveillance and risk management rather than ending completely. Mayo Clinic says atypical hyperplasia increases future breast-cancer risk, so patients may be offered more-frequent screening and, in selected higher-risk cases, additional imaging such as breast MRI. Johns Hopkins advises that patients diagnosed with atypical ductal hyperplasia should be followed in a high-risk breast clinic, where high-risk screening, lifestyle guidance, and risk-reducing strategies are discussed. MD Anderson similarly states that patients with ADH and no cancer after excision are referred for high-risk breast screening, which can include annual mammograms, breast MRI, and counseling about risk-reducing medication. This is a very important ground reality for patients and caregivers, because many assume surgery ends the journey, when in fact conventional management often continues in a different form.
Risk-Reducing Medicines Are Also Part of Standard Care
Conventional treatment is not only about procedures. Mayo Clinic also lists medicine to lower future breast-cancer risk as an option for some patients with atypical hyperplasia. These medicines include tamoxifen, raloxifene, anastrozole, and exemestane. MD Anderson also notes that patients may be referred for counseling about “chemoprevention,” meaning medication used to reduce the chance of future breast cancer rather than to treat an existing cancer. For some women, this becomes an important part of the discussion, especially if family history, age, hormonal factors, or overall risk level suggest a higher future probability of breast cancer.
What Conventional Treatment Does Well
Conventional care does several things extremely well in this setting. It identifies suspicious findings through imaging, confirms pathology through biopsy, uses surgery when more certainty is needed, and creates structured follow-up for women whose future risk is above average. It is strong at answering urgent questions such as whether cancer may already be present, whether the lesion was adequately sampled, and whether immediate excision is the safer option. For many patients, this diagnostic safety is the most important benefit of standard care.
What Patients and Caregivers Often Feel Inside This Process
Even when conventional care is medically appropriate, the experience can still feel incomplete to patients and families. A woman may be told the lesion is not cancer, yet surgery is still discussed. She may undergo excision and then be told she still needs high-risk follow-up. A caregiver may feel relieved after pathology confirms no cancer, then become confused again when annual mammograms, MRI, or risk-reducing medication are mentioned. These are not unusual reactions. They are part of the real emotional burden of benign and atypical breast disease. This is why a strong article should explain not only what conventional treatment does, but also why it can still leave patients searching for a deeper long-term plan.
What This Means for the Reader
If your report shows a benign breast lump, atypical hyperplasia, or another high-risk but non-cancerous lesion, conventional treatment is usually built around diagnostic confirmation, decision-making about surveillance versus excision, and long-term risk reduction. It is not automatically aggressive, and it is not automatically finished once the lesion is removed. Understanding that reality is essential, because it prepares the reader for the next question many patients naturally ask: if conventional treatment manages the lesion and the risk pathway, what else can support the body, recovery, and long-term tissue balance beyond surveillance alone? That is where the discussion can move responsibly into the limitations of conventional care and the role of integrative support.
References
[1] Mayo Clinic. (2024). Atypical hyperplasia of the breast: Diagnosis and treatment. Mayo Clinic.
Working link: https://www.mayoclinic.org/diseases-conditions/atypical-hyperplasia/diagnosis-treatment/drc-20369778
Brief: Explains that treatment may include surgery, more-frequent breast screening, and risk-reducing medication, and clarifies that not every patient needs surgery.
[2] American Society of Breast Surgeons. (2024). Resource guide: Surgical management of benign or high-risk lesions. American Society of Breast Surgeons.
Working link: https://www.breastsurgeons.org/docs/statements/asbrs-high-risk-lesions.pdf
Brief: Provides practical guidance on core needle biopsy, radiologic-pathologic concordance, selective versus routine excision, and follow-up pathways for high-risk breast lesions such as ADH, FEA, papillomas, and radial scars.
[4] Johns Hopkins Medicine. (2024). Atypical ductal hyperplasia. Johns Hopkins Medicine.
Working link: https://www.hopkinsmedicine.org/health/conditions-and-diseases/breast-cancer/atypical-ductal-hyperplasia
Brief: Describes typical diagnosis and treatment after ADH, including excisional biopsy, high-risk clinic follow-up, and risk-reducing strategies.
[5] MD Anderson Cancer Center. (2024). Atypical ductal hyperplasia: What it is and how it’s treated. MD Anderson Cancer Center.
Working link: https://www.mdanderson.org/cancerwise/atypical-ductal-hyperplasia–what-it-is-and-how-to-treat-it.h00-159695967.html
Brief: Explains why excisional biopsy is often the first step after ADH is found on core biopsy and how patients may later move into high-risk screening and chemoprevention discussions.
Limitations of Conventional Treatment
Conventional breast care is essential for diagnosis, imaging, biopsy, pathology review, and timely surgical decision-making. It is often the reason a serious lesion is detected early enough to be managed safely. That strength should be acknowledged clearly. But from the patient’s point of view, the journey often becomes difficult not because doctors do nothing, but because what happens next can feel incomplete, uncertain, and emotionally exhausting. In conditions such as atypical hyperplasia and other high-risk breast lesions, treatment may identify the lesion accurately while still leaving the patient with ongoing risk, repeated surveillance, and no clear sense of long-term closure. Mayo Clinic notes that not everyone needs surgery, some patients need more-frequent screening, and others may be offered risk-reducing medication, which means management often continues even after the initial biopsy result.
It Often Clarifies the Lesion, but Not the Patient’s Peace of Mind
One of the most common realities patients face is that a technically successful diagnosis does not always bring emotional clarity. A woman may hear that the lesion is benign or atypical and expect reassurance, only to be told that surgery is still being considered or that high-risk follow-up may continue. This creates a difficult psychological space. She is not being told she has cancer, but she also does not feel fully safe. Caregivers often struggle with the same contradiction. They ask, “If this is not cancer, why are we still talking about surgery, MRI, or medication?” That tension is built into the way these lesions are managed. The American Society of Breast Surgeons explains that decisions depend on imaging-pathology concordance, upgrade risk, operative and cosmetic risks, and whether the patient can comply with follow-up. In other words, the treatment pathway is individualized, but to patients it can feel uncertain and hard to predict.
Surgery Can Remove Tissue, but Not Always Remove Future Risk
This is one of the most important limitations for patients to understand. Surgery may remove the abnormal area that was sampled or identified on imaging, but in certain high-risk lesions it does not necessarily remove the broader future risk associated with that tissue pattern. The American Society of Breast Surgeons states that lesions such as atypical ductal hyperplasia and lobular neoplasia are associated with elevated lifetime breast-cancer risk that is not mitigated by surgical excision alone. That is why some women feel shocked after a procedure when they are still told they need ongoing screening, risk assessment, or medication discussions. From the medical perspective, this is appropriate risk management. From the patient’s perspective, it can feel as though the problem was treated but not truly resolved.
Surveillance Can Become a Long-Term Emotional Burden
Monitoring is often necessary, but it is not emotionally neutral. Annual mammograms, clinical breast exams, and sometimes MRI are meant to improve safety, yet they can also create a repeating cycle of anxiety. Some patients become fearful before every scan. Others begin to interpret minor breast sensations as signs of recurrence or progression. Caregivers may also relive the original fear with each new appointment. Mayo Clinic notes that after atypical hyperplasia, screening plans may include annual mammography, clinical exams, and supplemental imaging such as breast MRI in selected patients based on risk factors, family history, genetic findings, or dense breasts. MD Anderson similarly describes referral to high-risk screening that can include annual mammograms and breast MRIs after excision confirms ADH without cancer. Clinically, this makes sense. Emotionally, it can leave patients feeling as if they never fully leave the shadow of the diagnosis.
Risk-Reducing Medicines May Help, but They Also Bring New Concerns
For some patients, conventional care includes discussion of endocrine risk-reduction medicines such as tamoxifen, raloxifene, anastrozole, or exemestane. These options can be important, but they are not emotionally or physically simple decisions. The idea of taking medication for years after being told “you do not have cancer” can feel confusing or overwhelming. Mayo Clinic notes that these medicines are typically taken daily for five years to reduce breast-cancer risk. It also outlines meaningful trade-offs: raloxifene may have lower risks of blood clots and uterine cancer than tamoxifen in some postmenopausal patients, while aromatase inhibitors can cause hot flashes, vaginal dryness, joint and muscle pain, fatigue, and may increase osteoporosis risk. For many women, the conversation then shifts from fear of the lesion to fear of medication side effects, which can create a different kind of treatment fatigue.
The System Is Strong at Detection, but Often Weak at Whole-Person Recovery
Another limitation patients feel very deeply is that conventional care is excellent at identifying structural problems, but often less satisfying when patients ask broader questions. Many women want to know why their body developed this tissue change, whether inflammation, hormonal instability, weight gain, stress, poor sleep, or metabolic issues are contributing, and what they can actively do beyond waiting for the next scan. Standard care may address these questions to some extent, but in real practice many patients still leave with the sense that the lesion has been evaluated while the person has not been fully supported. This gap is one of the main reasons patients continue searching after biopsy or excision. It is not always because they reject conventional medicine. It is often because they feel conventional medicine has answered the urgent question, but not the larger one. That is where interest in Ayurveda and other integrative approaches often begins.
Some Patients Feel Pressured, While Others Feel Left Waiting
Real-world frustration often comes from timing. Some patients feel rushed toward surgery because uncertainty itself feels dangerous. Others feel left in prolonged observation with little help managing the fear that comes with watchful follow-up. Both experiences can be distressing. The American Society of Breast Surgeons explicitly frames excision decisions around shared decision-making, concordance review, upgrade risk, operative and cosmetic risks, and the patient’s ability to comply with follow-up. That is good clinical practice, but it also means the burden of decision-making can feel heavy to patients who are already anxious. Families may end up seeking multiple opinions not because the doctors are wrong, but because the emotional cost of uncertainty is high.
This Does Not Mean Conventional Care Has Failed
It is important to say this directly. These limitations do not mean conventional treatment is wrong or unnecessary. They mean that conventional care has a particular strength: it is designed to detect, classify, biopsy, monitor, and intervene when needed. That is vital. But patients often need something more than structural safety alone. They want continuity, reassurance, guidance on long-term tissue health, and a way to feel they are actively participating in recovery rather than only waiting for the next report. This is exactly why a responsible integrative discussion becomes relevant. The National Center for Complementary and Integrative Health states that complementary approaches should not replace necessary medical treatment or delay care, but some may help with symptom burden, anxiety, fatigue, and quality of life when used appropriately alongside standard care. That safety-centered framing is important because it allows patients to see Ayurveda not as an act of rebellion against medical care, but as a supportive response to the limitations they genuinely feel.
What This Means for Patients and Caregivers
The most honest conclusion is this: conventional care is often excellent at identifying risk, but it does not always give patients a full sense of recovery. A biopsy can answer what the lesion is. Surgery can remove what was found. Screening can reduce the chance of missing future disease. Medication can lower risk in selected people. But none of these automatically remove fear, restore confidence in the body, or create a long-term feeling of stability. That is why many patients keep reading after the biopsy, after the excision, and even after reassuring pathology. They are not only looking for treatment. They are looking for a deeper plan.
References
[1] Mayo Clinic. (2024). Atypical hyperplasia of the breast: Diagnosis and treatment. Mayo Clinic.
Working link: https://www.mayoclinic.org/diseases-conditions/atypical-hyperplasia/diagnosis-treatment/drc-20369778
Brief: Explains that not all patients need surgery, but many need ongoing screening, possible MRI, and discussion of risk-reducing medication after atypical hyperplasia is diagnosed.
[2] American Society of Breast Surgeons. (2024). Resource guide: Surgical management of benign or high-risk lesions. American Society of Breast Surgeons.
Working link: https://www.breastsurgeons.org/docs/statements/asbrs-high-risk-lesions.pdf
Brief: States that excision decisions should be individualized and that some high-risk lesions, including ADH and lobular neoplasia, carry future breast-cancer risk that is not removed by surgery alone.
[3] MD Anderson Cancer Center. (2024). Atypical ductal hyperplasia: What it is and how it’s treated. MD Anderson Cancer Center.
Working link: https://www.mdanderson.org/cancerwise/atypical-ductal-hyperplasia–what-it-is-and-how-to-treat-it.h00-159695967.html
Brief: Describes the pathway from core biopsy to excision and then into high-risk breast screening, MRI discussions, and risk-reducing medication counseling.
[4] Mayo Clinic. (2025). Breast cancer chemoprevention: Drugs that reduce risk. Mayo Clinic.
Working link: https://www.mayoclinic.org/diseases-conditions/breast-cancer/in-depth/breast-cancer/art-20045353
Brief: Reviews risk-reducing medicines such as tamoxifen, raloxifene, and aromatase inhibitors, including how long they are used and the side effects and risks patients must weigh.
[5] National Center for Complementary and Integrative Health. (2023). Cancer and complementary health approaches: What you need to know. U.S. Department of Health and Human Services.
Working link: https://www.nccih.nih.gov/health/cancer-and-complementary-health-approaches-what-you-need-to-know
Brief: Clarifies that complementary approaches should not replace or delay necessary medical care, but may help with symptom burden and quality of life when used appropriately alongside standard treatment.
What Patients Are Not Told After Diagnosis

A diagnosis does not only deliver information. It changes the emotional rhythm of a patient’s life almost immediately. The scan, the callback, the biopsy, the waiting, and then the report all create the expectation that once a name is given to the problem, clarity will follow. In reality, many women feel the opposite. They are told the lesion is benign or atypical, but they do not feel fully reassured. They are told it is not cancer, yet surgery may still be discussed. They are told the abnormal area can be removed, yet follow-up may continue for years. This is one of the most disorienting parts of the journey, and it is rarely explained in a way that feels complete to patients or caregivers. Mayo Clinic makes clear that atypical hyperplasia is not breast cancer, but it does raise future breast-cancer risk and may lead to surgery, more-frequent screening, and risk-reducing medication discussions. (Mayo Clinic)
They Are Usually Told What Was Found, but Not What the Diagnosis Will Feel Like
Patients are often given the technical meaning of the diagnosis, but not the lived meaning of it. A woman may hear words like atypical ductal hyperplasia, flat epithelial atypia, papilloma, or high-risk lesion and immediately begin imagining worst-case scenarios. At home, she may reread the pathology report repeatedly without understanding whether she is in danger now or only at higher risk later. Caregivers often struggle too, because they want simple answers and discover that the answer is not simple. The emotional result is a strange in-between state. The patient is not treated as though she has cancer, but she no longer feels like someone who can simply forget the issue and move on. That uncertainty is not a small side effect of diagnosis. For many families, it becomes the dominant reality of the next several months. (Mayo Clinic)
They Are Not Always Told That “Benign” Can Still Lead to Long-Term Follow-Up
One of the most common misunderstandings after diagnosis is the belief that benign means finished. In reality, some benign and atypical lesions are treated as risk markers rather than isolated events. That means the abnormality may not be cancer today, but it can still indicate a tissue pattern associated with higher future risk. The American Society of Breast Surgeons states that certain high-risk lesions, including atypical ductal hyperplasia and lobular neoplasia, are associated with elevated lifetime breast-cancer risk that is not removed by surgical excision alone. This is one of the hardest truths for patients to accept because it means that even after biopsy or excision, the story may not be over. (American Society of Breast Surgeons)
They Are Not Always Prepared for How Heavy the Decision-Making Can Feel
Patients are often told that management depends on imaging findings, biopsy results, family history, and overall risk. Clinically, that is appropriate. Emotionally, it can feel overwhelming. A woman may feel pushed toward surgery because uncertainty itself is frightening. Another may resist surgery because she fears overtreatment, scars, cost, disruption to family life, or the possibility that the lesion would never have become dangerous. Caregivers frequently seek multiple opinions not because they reject medical advice, but because the burden of choosing feels heavy. The American Society of Breast Surgeons specifically frames management around concordance assessment, upgrade risk, operative and cosmetic risks, and the patient’s ability to comply with follow-up. That is sound clinical practice, but patients are not always told in advance how exhausting it can feel to live inside that decision process. (American Society of Breast Surgeons)
They Are Not Always Told That Surgery May Solve One Problem but Not the Whole One
When surgery is recommended, many patients believe the main purpose is final removal and closure. In some cases that is largely true. In other cases, surgery is also a diagnostic safeguard to make sure a more advanced lesion was not missed in the sampled tissue. Even when the excision confirms no cancer, the patient may still remain in a higher-risk category afterward. That can be emotionally difficult because the patient often expects the operation to end the problem completely. Instead, she may later hear about annual mammograms, clinical breast exams, MRI in selected cases, or risk-reducing medication. This is where many patients begin to feel that the lesion was addressed, but the deeper question of long-term recovery was not. Mayo Clinic and high-risk lesion guidance both reflect this reality by linking atypical hyperplasia to ongoing surveillance and prevention planning after diagnosis. (Mayo Clinic)
They Are Not Always Warned About the Emotional Cost of Surveillance
From a medical standpoint, surveillance is protective. From a human standpoint, it can be draining. Every mammogram can reopen fear. Every callback can trigger the same panic felt during the first abnormal scan. Some women become more alert to normal breast sensations and interpret them as danger. Others begin postponing follow-up because the anticipation itself feels too stressful. Families may think that surveillance is a low-burden plan compared with surgery, but many patients experience it as a repeated cycle of worry. Breast MRI can be useful in selected higher-risk situations, but ASBrS MRI guidance also notes that MRI is associated with frequent false-positive findings, increased costs of care, and increased patient anxiety. Patients deserve to be told that surveillance is not only a technical pathway. It is an emotional pathway too. (Mayo Clinic)
They Are Not Always Told That Medicines for Risk Reduction Can Create a New Kind of Distress
Some women are offered endocrine risk-reducing medicines after diagnosis. These options can be important, but the conversation is not always emotionally easy. Being told to take daily medication for years after hearing “you do not have cancer” can feel confusing, especially when the medicines bring their own side-effect concerns. Mayo Clinic notes that risk-reducing medicines such as tamoxifen, raloxifene, anastrozole, and exemestane may be used in some patients, while also outlining risks and side effects that can affect quality of life. Many patients quietly begin weighing one fear against another, fear of future cancer versus fear of medication burden. This is one more way diagnosis can feel like the beginning of a long journey rather than the end of a short one. (Mayo Clinic)
They Are Not Always Given a Whole-Person Recovery Plan
This may be the most important gap of all. Conventional care is often excellent at identifying structural abnormalities, clarifying pathology, guiding biopsy, deciding on surgery, and building a follow-up plan. But many women leave the diagnostic process still wondering what they can actually do for their body beyond watching, scanning, or taking medication. They want to know whether sleep, digestion, weight, metabolic health, stress, inflammation, hormonal rhythm, and tissue nourishment matter. They want to know whether the body can be supported, not only monitored. When those questions are left unanswered, patients often continue searching, not because they reject modern medicine, but because they feel it has answered the urgent question without answering the deeper one. This is the exact place where interest in Ayurveda and integrative care begins to feel relevant. NCCIH states that no complementary approach has been shown to cure cancer, and that such approaches should not replace or delay medical care, but selected complementary approaches may help manage symptoms, anxiety, fatigue, and quality of life alongside standard care. That guidance supports a responsible integrative bridge rather than an oppositional one. (NCCIH)
They Are Not Always Told That Caregivers Need Guidance Too
Caregivers are often expected to remain practical, but they carry their own version of the burden. They help interpret medical language, remember appointments, search online for answers, and hold the emotional weight when the patient becomes exhausted. Yet they are rarely given direct language for what the diagnosis really means over time. Many caregivers are not asking only whether the lesion is serious. They are asking whether life will now revolve around imaging, whether surgery truly ends anything, whether the next scan will reopen the same fear, and whether there is a meaningful way to support the patient beyond crisis management. An article that speaks to caregivers as well as patients immediately feels more honest because it reflects what actually happens in families after diagnosis. (Mayo Clinic)
What Patients Usually Need to Hear, but Often Do Not
Most patients do not need more alarming language. They need a more complete explanation. They need to hear that it is possible for a lesion to be non-cancerous and still important. They need to hear that surgery may be useful and still not erase all future risk. They need to hear that surveillance protects safety but can also create emotional fatigue. They need to hear that wanting something more than repeated scans is understandable. Above all, they need to hear that there is a difference between ignoring medical care and looking for a deeper, more supportive long-term plan. That difference is where trust is built, and it is also where patients become ready to consider Ayurveda in a serious, responsible way. (Mayo Clinic)
Transition to the Next Section
By this stage, many patients begin looking for a deeper plan that supports recovery, reduces fear, and addresses long-term tissue health. But before discussing where Ayurveda may help, one question must be answered clearly: where does integrative care fit safely, and what should never be delayed? (NCCIH)
References
[1] Mayo Clinic. (2024). Atypical hyperplasia of the breast: Diagnosis and treatment.
Working link: https://www.mayoclinic.org/diseases-conditions/atypical-hyperplasia/diagnosis-treatment/drc-20369778
Brief: Explains that atypical hyperplasia is not cancer, but may still lead to surgery, more-frequent screening, supplemental imaging in selected patients, and risk-reducing medication discussions. (Mayo Clinic)
[2] American Society of Breast Surgeons. (2024). Resource guide: Surgical management of benign or high-risk lesions.
Working link: https://www.breastsurgeons.org/docs/statements/asbrs-high-risk-lesions.pdf
Brief: States that certain high-risk lesions, including ADH and lobular neoplasia, carry elevated lifetime breast-cancer risk that is not removed by surgical excision alone, and emphasizes concordance assessment and individualized management. (American Society of Breast Surgeons)
[3] American Society of Breast Surgeons. (2018). Consensus guideline on diagnostic and screening magnetic resonance imaging of the breast.
Working link: https://www.breastsurgeons.org/docs/statements/Consensus-Guideline-on-Diagnostic-and-Screening-Magnetic-Resonance-Imaging-of-the-Breast.pdf
Brief: Notes that MRI can be useful in selected settings, but is also associated with false-positive findings, increased costs, and increased patient anxiety. (American Society of Breast Surgeons)
[4] National Center for Complementary and Integrative Health. (2023). Cancer and complementary health approaches: What you need to know.
Working link: https://www.nccih.nih.gov/health/cancer-and-complementary-health-approaches-what-you-need-to-know
Brief: Explains that complementary approaches should not replace or delay medical treatment for cancer, but selected approaches may help with symptoms, anxiety, fatigue, and quality of life when used appropriately alongside standard care. (NCCIH)
Integrative Safety: Where Ayurveda Fits and What Should Never Be Delayed

One of the biggest reasons patients hesitate to explore Ayurveda after a breast biopsy or excision is the fear of making the wrong decision at the wrong time. Some worry that using an integrative approach may delay necessary treatment. Others fear the opposite, that if they follow only standard surveillance, nothing meaningful is being done to improve the deeper biological environment that may have contributed to the lesion in the first place. This is where the article must be especially clear. Ayurveda can be presented responsibly and convincingly only when it is placed in the correct role. It should not be described as a substitute for urgent imaging, biopsy, surgical review, or oncology referral when those are clinically indicated. It should be described as a supportive and individualized approach that may help strengthen the patient during observation, recovery, and long-term risk-reduction phases while modern medical follow-up continues. The National Center for Complementary and Integrative Health states clearly that unproven products or practices should not be used to replace or delay medical treatment for cancer, and that some complementary approaches may interfere with treatment if used inappropriately. At the same time, NCCIH also notes that some complementary approaches may help with symptom burden, anxiety, fatigue, and quality of life when used alongside standard care.
Why This Section Matters So Much
Patients and caregivers often read integrative content when they are already emotionally overloaded. They may be carrying the memory of a frightening mammogram, a biopsy recommendation, a surgery discussion, or a pathology report they do not fully understand. In that state, they do not need exaggerated promises. They need guidance they can trust. If the content sounds anti-doctor, anti-surgery, or dismissive of necessary evaluation, many serious readers will immediately stop trusting it. On the other hand, if the article only repeats conventional follow-up advice and never addresses deeper recovery, emotional burden, inflammation, digestion, hormonal balance, or long-term tissue support, then Ayurveda will feel like an afterthought rather than a meaningful option. The strongest strategy is to show that modern medicine is essential for diagnosis and safety, while Ayurveda may offer support in the areas patients often feel are left incomplete.
What Should Never Be Delayed
There are moments when integrative care should clearly remain secondary to urgent medical evaluation. A new breast lump, suspicious calcifications on imaging, bloody nipple discharge, rapidly changing breast shape, skin dimpling, persistent unilateral nipple inversion, or discordance between biopsy findings and imaging should never be handled casually. Guidance from NICE on suspected cancer emphasizes the importance of timely recognition and referral when concerning findings are present. That matters because some patients delay action not out of denial, but out of confusion. They hope the finding is minor, assume a benign report means there is no urgency, or become overwhelmed by conflicting opinions. This is exactly why the article must say, in plain language, that Ayurveda should not delay urgent breast imaging, repeat biopsy when needed, surgical consultation, or specialist referral.
Where Ayurveda Fits Before Surgery
Before surgery, Ayurveda fits best as a supportive framework, not as a reason to avoid proper diagnosis. This is the stage where many patients feel mentally and physically unsettled. They may have disturbed sleep, digestive upset, emotional fear, inflammatory stress, and a constant sense of uncertainty while waiting for decisions. Some are under imaging surveillance. Others are deciding whether excision is necessary. At this point, Ayurveda can be introduced as an approach that may help support digestion, reduce internal stress burden, improve daily routine, strengthen resilience, and provide a more active sense of participation in health while standard medical assessment continues. This is especially important for patients who feel that observation alone leaves them passive and anxious.
The message should be simple and believable. Ayurveda before surgery is not about “replacing” the decision-making process. It is about supporting the body and mind while that process unfolds. That positioning is more credible to Western readers and more aligned with safety guidance from NCCIH, which advises patients to consult their health care providers before adding complementary products or practices to cancer-related care.
Where Ayurveda Fits After Surgery
After surgery or excision, Ayurveda becomes even more understandable to many patients. By this stage, the urgent structural decision has already been made. The lesion has been sampled or removed. Yet the patient may still feel far from restored. She may continue to worry about future risk, repeated scans, hormonal imbalance, stress, fatigue, poor sleep, or the feeling that the body is still carrying a deeper instability. This is where an integrative Ayurvedic approach can be presented as long-term support rather than emergency intervention. It may help the patient focus on routines, digestion, metabolic balance, inflammatory load, emotional steadiness, and tissue nourishment while standard surveillance continues.
This is also the stage where caregivers often become more open to Ayurveda. Earlier, they may have been focused almost entirely on whether surgery was needed. After surgery, they begin to see that the journey did not end with the procedure. Follow-up continues, anxiety continues, and the desire for something more restorative becomes stronger. A clear article should acknowledge this openly. Patients are not only trying to avoid disease. They are trying to rebuild a sense of safety in their own body.
The Importance of Coordinated Care
One of the most responsible ways to present Ayurveda in a cancer-related article is to emphasize coordination rather than separation. Patients should be encouraged to keep their breast specialist informed, continue recommended imaging, and discuss any herbs, supplements, or complementary practices they are considering. NCCIH warns that some herbs may interfere with cancer treatment or make chemotherapy drugs less effective, which is why any integrative plan must be individualized and medically aware. This point builds trust rather than weakening it, because serious readers want to know that safety is being taken seriously.
A coordinated model also helps resolve one of the biggest emotional conflicts patients face. They often feel they must choose between two worlds, conventional medicine or Ayurveda. But in real clinical life, many patients are not looking for an either-or answer. They want modern diagnostics for safety and they want deeper support for the parts of healing they feel are otherwise ignored. This is where your article becomes more persuasive than pages that argue too aggressively in one direction.
What Patients and Caregivers Need to Hear Clearly
Patients need to hear that it is reasonable to want more than surveillance alone. Caregivers need to hear that choosing an integrative path does not have to mean abandoning safety. Both need to hear that responsible Ayurveda is not built on avoidance. It is built on timing, personalization, and support. The strongest message in this section is not “Ayurveda cures everything.” The strongest message is that Ayurveda may become meaningful when it is used in the right phase, for the right purpose, and without delaying the medical steps that protect the patient from missed or advancing disease.
This kind of clarity is what keeps readers engaged. It reduces resistance, lowers fear, and helps patients feel that exploring Ayurveda can be done thoughtfully rather than impulsively.
What This Means for You Right Now
If you are in the middle of deciding what to do after a biopsy or after surgery, the safest way to think about Ayurveda is this. Use modern breast care to define the diagnosis, clarify urgency, and guide screening or intervention. Use Ayurveda, when appropriate, to support the body through stress, recovery, metabolic balance, routine, tissue nourishment, and long-term resilience. That is the most balanced and trustworthy way to move forward.
Once this safety foundation is clear, the next step becomes much more practical. If Ayurveda has a supportive role, then what exactly should the long-term plan focus on to reduce risk and strengthen tissue health over time? That is where the discussion naturally moves into risk reduction, diet, lifestyle, and a structured Ayurvedic strategy for long-term support.
References
National Center for Complementary and Integrative Health. (2023). Cancer and complementary health approaches: What you need to know. U.S. Department of Health and Human Services.
Working link: https://www.nccih.nih.gov/health/cancer-and-complementary-health-approaches-what-you-need-to-know
Brief: Explains that complementary approaches should not replace or delay medical treatment for cancer, warns that some herbs may interfere with treatment, and notes that selected complementary approaches may help with symptoms, anxiety, fatigue, and quality of life when used appropriately alongside standard care.
National Institute for Health and Care Excellence. (2023). Suspected cancer: recognition and referral (NG12). NICE.
Working link: https://www.nice.org.uk/guidance/ng12
Brief: Provides guidance on recognizing concerning symptoms and ensuring timely referral and evaluation when potentially serious findings are present.
Ayurvedic Perspective on Benign Breast Lump and Atypical Hyperplasia

Ayurvedic classics do not use the modern histopathology label “atypical hyperplasia.” For that reason, this section should be read as a classical interpretive bridge, not as a claim that an ancient text named the modern biopsy diagnosis word for word. In Ayurvedic reasoning, a benign breast lump or atypical proliferative change is better understood through the combined lens of Granthi (localized nodular swelling), Agni disturbance, Ama accumulation, Srotas dysfunction, and Dhatu involvement. Sushruta places Granthi within a dedicated nidana framework, while Charaka and Vagbhata explain how impaired transformation and disturbed channels allow tissue-level pathology to emerge.
For Gulf readers, the practical meaning can be stated simply. A breast lump is not always viewed only as an isolated structure. It may also reflect a deeper internal terrain problem.
Urdu: یہ صرف گلٹی کا مسئلہ نہیں، بلکہ جسم کے اندرونی توازن، سوزش، اور بافتی کمزوری کا اشارہ بھی ہو سکتا ہے۔
Arabic: ليست المسألة مجرد كتلة فقط، بل قد تعكس خللا داخليا في التوازن والالتهاب وقابلية النسيج.
Granthi: The Classical Lens for a Localized Lump
A very important classical anchor comes from Sushruta Samhita, Nidanasthana, Chapter 11, Verse 3:
Sanskrit
वातादयो मांसमसृक् च दुष्टाः सन्दूष्य मेदश्च कफानुविद्धम् ।
वृत्तोन्नतं विग्रथितं तु शोफं कुर्वन्त्यतो ग्रन्थिरिति प्रदिष्टः ॥३॥
Transliteration
Vatadayo mamsamasrk cha dushtah sandushya medash cha kaphanuviddham,
vrttonnatam vigrathitam tu shopham kurvanty ato granthir iti pradishtah.
Meaning
When the vitiated Doshas affect muscle, blood, and fat tissue, especially in association with Kapha, they produce a rounded, elevated, knot-like swelling called Granthi.
This verse is highly useful for a modern breast article because it gives a classical framework for understanding a palpable or imaging-detected nodular change without forcing a false one-to-one equation with cancer. The language of mamsa (muscle or soft tissue), asrk/rakta (blood), meda (fat tissue), and Kapha-associated swelling conceptually fits many patients who present with heaviness, density, round nodularity, cystic tendency, localized thickening, or recurrent benign breast changes. In this interpretation, the concern is not only the visible lump. The deeper issue is the tissue environment that allowed the lump-like change to arise.
Urdu: گرنتھی کا مطلب صرف گلٹی نہیں، بلکہ ایسی بافتی تبدیلی بھی ہے جو جسم کے اندر بگڑے ہوئے توازن سے پیدا ہوتی ہے۔
Arabic: مفهوم “غرنثي” لا يعني مجرد كتلة، بل يشير إلى تغير نسيجي ينشأ من اختلال داخلي في التوازن.
Agni and Ama: Why Ayurveda Looks Beyond the Lump
A second major classical key comes from Ashtanga Hridaya, Nidanasthana, Chapter 12, Verse 1:
Sanskrit
रोगाः सर्वेऽपि मन्देऽग्नौ सुतरामुदराणि तु ।
अजीर्णान्मलिनैश्चान्नैर्जायन्ते मलसञ्चयात् ॥१॥
Transliteration
Rogah sarve api mande agnau sutaram udarani tu,
ajirnan malinaish channair jayante malasanchayat.
Meaning
Disease arises when Agni becomes weak. Improper digestion, impure or unsuitable intake, and the accumulation of metabolic waste create the basis for pathology.
In a breast-health context, this verse is not used to say that every lump comes only from digestion. That would be too simplistic. Its value is deeper. It explains why Ayurveda begins by examining the metabolic and transformative state of the body. Many women who develop recurrent benign breast changes, painful nodularity, cyclical swelling, heaviness, weight gain, sluggishness, bloating, constipation, insulin resistance, inflammatory tendency, or hormonal irregularity often feel that the biopsy report is only the surface of a larger internal problem. The Agni-Ama model gives language to that experience. If transformation is weak and improperly processed material accumulates, tissues may become more congested, reactive, or disordered over time. That is why Ayurveda does not stop at the question, “What is the lump?” It also asks, “What terrain allowed this pattern to form?”
Urdu: جب ہاضمہ اور جسمانی تبدیلی کا نظام کمزور ہو جائے، تو صرف ایک عضو نہیں بلکہ پورا جسم اثر لے سکتا ہے۔
Arabic: عندما تضعف قوة الهضم والتحول الحيوي، فقد لا تتأثر منطقة واحدة فقط، بل قد يتأثر توازن الجسم كله.
Srotas: Why Channel Health Matters in Tissue Disorders
A third major classical anchor comes from Charaka Samhita, Vimanasthana, Chapter 5, Verse 3:
Sanskrit
यावन्तः पुरुषे मूर्तिमन्तो भावविशेषास्तावन्त एवास्मिन् स्रोतसां प्रकारविशेषाः ।
सर्वे हि भावा पुरुषे नान्तरेण स्रोतांस्यभिनिर्वर्तन्ते, क्षयं वाऽप्यभिगच्छन्ति ।
स्रोतांसि खलु परिणाममापद्यमानानां धातूनामभिवाहीनि भवन्त्ययनार्थेन ॥३॥
Transliteration
Yavantah purushe murtimanto bhavavisheshas tavanta evasmin srotasam prakaravisheshah.
Sarve hi bhava purushe nantaren srotamsy abhinirvartante, kshayam vapy abhigacchanti.
Srotamsi khalu parinamam apadyamananam dhatunam abhivahini bhavanti ayanarthena.
Meaning
The body contains as many kinds of channels as there are functional and structural elements. Without these channels, tissues cannot arise, be nourished, or decline properly. Srotas are the inner pathways that carry Dhatus as they undergo transformation.
This verse is extremely important when explaining why Ayurveda sees a breast lesion as more than an isolated event. The body is described as a network of channels, and disease develops when transport, nourishment, clearance, or transformation become disturbed. In practical clinical language, this helps explain why women with breast abnormalities may also report a wider pattern: breast fullness, cyclical congestion, edema, tenderness, irregular appetite, sluggish bowels, disturbed sleep, mental tension, or metabolic heaviness. Ayurveda interprets these as signs that the local lesion may be emerging within a broader background of Srotas dysfunction and Dhatu imbalance, not merely as a random local event.
Urdu: جسم راستوں اور نالیوں کا ایک مربوط نظام ہے۔ جب یہ راستے بند، سست، یا بگڑ جائیں تو بافتوں کی پرورش بھی متاثر ہوتی ہے۔
Arabic: الجسم شبكة مترابطة من القنوات. وعندما تضطرب هذه القنوات أو تنسد، يتأثر تغذي النسيج ووظيفته.
How This Applies Before Surgery
Before surgery, this Ayurvedic reading is especially valuable because many patients feel trapped in passive waiting. They are told to repeat imaging, decide on excision, or remain under surveillance, but they feel nothing is being done for the internal terrain. The classical model allows you to tell the reader something important and reassuring: even while diagnostic safety is being respected, there may already be meaningful work to do at the level of Agni, Ama, Kapha congestion, Pitta reactivity, Vata instability, and Srotas flow. This makes the patient feel less helpless. She is no longer only waiting for the next scan. She is beginning to understand why her body may have become vulnerable in the first place. That kind of framing often keeps the reader engaged because it matches what many patients actually feel after a confusing biopsy result.
How This Applies After Surgery
After surgery or excision, the Ayurvedic perspective often becomes even more compelling. At that point the urgent structural question may already have been addressed, but the patient still wants to know why follow-up continues and why she does not feel fully “finished” with the issue. The classical answer is that removal of a lesion does not automatically mean the terrain has been corrected. Kapha-related congestion, Pitta-related inflammatory tendency, Vata-related instability, weak Agni, Ama burden, or dysfunctional Srotas may still remain. That is exactly why many women become more open to Ayurveda after surgery than before it. They are no longer asking only, “What was removed?” They are asking, “What still needs to be corrected so that the same pattern does not keep returning?” This is where the Ayurvedic model feels most meaningful because it speaks directly to long-term tissue behavior, not only to the excised structure.
The Strongest Clinical Bridge for Patients and Caregivers
The most effective way to present this section is not to claim that Ayurveda and modern pathology are saying the same thing in different words. That would sound forced. The stronger and more honest message is this: modern breast care identifies the lesion, estimates urgency, and guides imaging, biopsy, or surgery. Ayurveda interprets the soil in which the lesion developed. One system defines the structure. The other studies the terrain. When readers understand that distinction, they are far more likely to continue reading and take Ayurveda seriously as part of a long-term plan.
For Gulf readers, this can be expressed simply and memorably:
Urdu: جدید طب گلٹی کی شناخت کرتی ہے، جبکہ آیوروید اس زمین کو سمجھتی ہے جس میں یہ گلٹی پیدا ہوئی۔
Arabic: الطب الحديث يحدد الكتلة، أما الأيورفيدا فتركز على البيئة الداخلية التي سمحت بظهورها.
What This Means for the Reader
If your report mentions a benign breast lump, atypical hyperplasia, papilloma, radial scar, or another high-risk but non-cancerous lesion, the Ayurvedic perspective does not ask you to ignore imaging or delay needed treatment. It asks a different set of questions. Is there Kapha-related congestion? Is there Pitta-driven reactivity? Is Vata creating instability, pain, and anxiety? Is Agni weak? Is Ama accumulating? Are Srotas delivering nourishment and clearance properly? These questions do not replace the biopsy report. They deepen the understanding of why the body may still need support even when the diagnosis is called benign or pre-malignant by modern standards. That is the foundation for the next section, which should move from interpretation into treatment principles.
Classical Anchors Used in This Section
Sushruta Samhita, Nidanasthana 11/3: Granthi is described as a rounded, elevated, knot-like swelling arising when Doshas disturb mamsa, rakta, and meda, especially with Kapha involvement.
Ashtanga Hridaya, Nidanasthana 12/1: Weak Agni, improper digestion, and accumulated waste are presented as major foundations of pathology.
Charaka Samhita, Vimanasthana 5/3: The body is explained as a network of channels through which Dhatus are carried and transformed, making Srotas central to both nourishment and disease.
Ayurvedic Treatment Principles

Once the Ayurvedic perspective is understood, the next practical question is how treatment is approached in a real patient. This is where many readers expect a list of herbs or formulations, but classical Ayurvedic treatment begins earlier than that. It starts by asking what kind of internal terrain allowed the tissue change to appear, why the body is reacting in this way now, and what must be corrected first so that deeper therapy can work properly. Modern reviews of Ayurvedic oncology repeatedly describe this logic as a whole-systems approach built around digestion, metabolism, inflammatory balance, tissue nourishment, and restoration of strength, rather than a purely lesion-centered method.
The first principle is terrain correction, not just lesion focus
One of the main differences patients notice is that Ayurveda does not stop at naming the lesion. It asks whether the body is currently dominated by Kapha-type congestion, Pitta-type inflammatory reactivity, Vata-type instability, or a mixed pattern that changes how the tissue behaves over time. This matters because two women may carry the same biopsy label and still have completely different underlying patterns. One may have heaviness, weight gain, sluggish digestion, and dense painful tissue. Another may have sharp cyclical pain, burning, irritability, stress, and inflammatory symptoms. A third may have fluctuating tenderness, anxiety, insomnia, irregular appetite, and post-procedural dryness or instability. A responsible Ayurvedic plan treats these as different internal states, not as one identical problem. Reviews of Ayurveda in cancer research describe this individualized, constitution- and imbalance-based model as central to how treatment is selected.
For patients, this principle is often deeply reassuring because it explains why generic advice has not always felt enough. Many women feel that standard care accurately identifies the lesion but does not fully explain why their body remains reactive, congested, anxious, hormonally unstable, or metabolically imbalanced even after the report is complete. Terrain correction gives language to that lived reality. It tells the reader that the goal is not only to “watch the breast” but to improve the biological environment in which breast tissue is functioning.
The second principle is to restore Agni before expecting durable change
In Ayurvedic reasoning, treatment is weak if Agni remains weak. Agni is not limited to stomach acid or digestion in the narrow modern sense. It refers to the body’s capacity to transform food, regulate metabolism, process internal inputs properly, and nourish tissues in an orderly way. Reviews connecting Ayurveda with oncology note that strengthening Agni and correcting the generation of Ama are major treatment goals because impaired transformation is believed to contribute to disturbed tissue behavior and chronic disease patterns.
This principle is highly relevant to real patients because many women with long-standing benign breast problems or atypical findings also report practical metabolic disturbances. They may live with bloating, sluggish bowel habits, emotional eating, weight gain, poor sleep, low morning appetite, afternoon crashes, sugar cravings, or a sense that the body no longer feels efficient or balanced. These concerns are often dismissed as background problems, yet they strongly influence how patients feel after diagnosis. Ayurveda treats them as clinically relevant, not peripheral. That is one reason patients often feel more understood once the discussion moves beyond the breast itself.
The third principle is to reduce Ama and clear obstructed pathways
If Agni is weak, Ayurveda expects Ama to accumulate. In practical terms, this means that treatment does not move directly to “strong medicine” without first considering whether the system is overloaded, congested, and poorly clearing what it should. Reviews on Ayurveda and cancer describe Ama as a key pathological concept and connect treatment with improving digestion, reducing stagnation, and supporting clearer tissue-level function.
Patients often recognize this principle from their daily experience even if they do not know the term Ama. They may say they feel heavy, puffy, inflamed, sluggish, mentally foggy, or internally “blocked” after years of stress, erratic routine, processed food, low activity, or poor sleep. Others feel that after biopsy or surgery their body has become even more reactive and less resilient. The Ayurvedic principle here is not mystical. It is a structured way of saying that before long-term tissue support can work well, the body often needs help becoming more receptive, better regulated, and less congested.
The fourth principle is to reopen and support Srotas
Ayurvedic treatment does not focus only on what has accumulated. It also focuses on where normal movement has been impaired. When Srotas, the functional channels of movement, nourishment, and clearance, are disturbed, tissues may not receive or release what they should in a balanced way. In a patient with breast-related concerns, this principle becomes clinically meaningful when there is recurring nodularity, cyclical swelling, heaviness, fluid retention, irregular tissue pain, or repeated “benign” findings over time. Ayurveda interprets these patterns as signs that circulation, transformation, nourishment, and tissue communication are not occurring smoothly. Reviews of Ayurvedic whole-systems thinking in oncology emphasize this systems-level logic rather than a single-target model.
This matters for readers because many feel frustrated when each new finding is treated as a separate event. A woman may have had dense breasts for years, then cystic changes, then biopsy, then atypia, then continued follow-up. From her perspective, these events often do not feel separate at all. They feel connected. Ayurveda’s Srotas-based reasoning helps explain why a history of repeated findings may reflect an ongoing terrain issue rather than isolated accidents.
The fifth principle is Dhatu support, not only symptom suppression
Ayurveda aims to improve the quality of the tissues themselves. In breast-related conditions, the emphasis often falls on Rasa, Rakta, Mamsa, and Meda Dhatu, because breast structure and reactivity are understood through the quality of nourishment, blood-related activity, muscular-fibrous integrity, and adipose-metabolic behavior. Whole-systems Ayurvedic reviews describe Rasayana-oriented care as preventive and restorative, with a focus on rebuilding strength, resilience, and immunity rather than only suppressing a short-term symptom picture.
This principle becomes very persuasive after surgery or prolonged surveillance. Many patients say that what they want most is not simply another test result. They want to feel that their tissues are becoming healthier, steadier, and less vulnerable over time. That desire is not irrational. It is one of the main reasons people continue searching after a supposedly reassuring diagnosis. Dhatu support gives treatment a long-term direction that patients can understand: not merely “remove what is wrong,” but improve the quality of what remains.
The sixth principle is to protect and rebuild Ojas
No long-term Ayurvedic plan is complete if the patient remains emotionally depleted, sleep-deprived, fearful, and physiologically exhausted. Ojas, in practical clinical writing, can be explained as the body’s reserve of resilience, steadiness, recovery capacity, and integrated vitality. After a worrying mammogram, biopsy, pathology report, surgery discussion, or years of repeated breast surveillance, many women are not only physically strained but also mentally worn down. Caregivers often notice this before the patient says it aloud. She may become more reactive, less rested, more fearful before appointments, and less trusting of her own body. Integrative Ayurvedic reviews repeatedly describe cancer-related Ayurvedic care as including restoration of strength, immunity, and quality of life, which closely aligns with the classical idea of protecting Ojas.
This is one reason why Ayurveda feels meaningful to many patients even before they discuss specific formulations. It acknowledges that healing is not only about the lesion or the scan. It is also about whether the person still feels internally strong enough to live without constant fear. That emotional reality should never be treated as secondary. It is part of why patients keep reading and part of why a responsible integrative plan can feel so valuable.
The seventh principle is sequencing, not rushing
A practical Ayurvedic treatment plan usually follows a sequence. First, assess urgency and do not delay necessary medical care. Second, correct diet, routine, sleep, stress load, digestion, and bowel pattern so the body becomes more stable. Third, choose herbs or formulations based on the dominant Dosha pattern, tissue behavior, strength of Agni, age, menstrual status, body composition, and whether the patient is in a pre-surgery, post-surgery, or surveillance phase. Fourth, consider deeper cleansing or Panchakarma-style strategies only when appropriate, and only as optional, individualized steps rather than routine advice for everyone. This sequencing is consistent with the way Ayurveda is described in modern integrative reviews, which emphasize personalized, multimodal care rather than one uniform prescription.
This principle protects patients from two extremes. One is doing nothing while waiting passively for the next scan. The other is jumping too quickly into aggressive herbs, detox ideas, or unstructured self-treatment without understanding the patient’s phase of care. The safest message is that Ayurveda should be introduced with timing, personalization, and awareness of medical context. NCCIH is very clear that complementary approaches should not replace or delay necessary treatment, and that some products may interfere with conventional therapy if used poorly.
What these principles mean in real life
For the reader, these treatment principles can be translated into one simple idea. Ayurveda is not merely asking, “What medicine should I take for this lesion?” It is asking, “What has become disturbed in my system that needs to be corrected if I want more durable balance?” That is a very different question, and it is why so many patients feel that Ayurvedic treatment principles sound closer to their lived reality than a purely report-based conversation. They are not only worried about the lesion. They are worried about why the body became vulnerable, why fear continues even after procedures, and how to build long-term steadiness rather than short-term reassurance alone.
This is the right foundation before moving into the next section. Once the principles are clear, the natural next question is not philosophical. It is practical. How are these principles applied step by step in a personalized plan before surgery, after surgery, and during long-term follow-up? That leads directly into Ayurvedic management.
References
Charaka Samhita, Sutrasthana 28.
Classical Ayurvedic reference used for the centrality of Agni in health and disease. A standard English translation is available in archived editions.
Sushruta Samhita, Nidanasthana, Granthi-related descriptions.
Classical Ayurvedic reference used to interpret localized nodular or glandular swellings through the lens of Granthi. A modern scholarly review discussing Granthi as a benign cystic node or nodule is available here.
Ashtanga Hridaya, Sutrasthana, foundational Dosha principles.
Classical Ayurvedic reference for systemic Dosha-based treatment logic. This section’s modern explanatory support comes from whole-systems Ayurvedic reviews in integrative oncology.
Sumantran, V. N., et al. (2012). Cancer, inflammation, and insights from Ayurveda.
A modern review explaining how Ayurveda links Agni, Ama, inflammation, and tissue behavior in chronic disease and cancer-related thinking.
Arnold, J. T. (2023). Integrating Ayurvedic medicine into cancer research programs, Part 2: Ayurvedic herbs and research opportunities. Journal of Ayurveda and Integrative Medicine.
A modern integrative review describing Ayurveda as an adjunct, not a substitute, and discussing Rasayana-oriented care, personalization, and restoration of strength and immunity.
Dhruva, A., et al. (2020). A 4-month whole-systems Ayurvedic medicine nutrition and lifestyle intervention for breast cancer survivors.
A feasibility study showing that a multimodal Ayurvedic nutrition and lifestyle intervention was feasible and acceptable in breast cancer survivors, relevant for whole-person recovery support.
National Center for Complementary and Integrative Health. (2023). Cancer and complementary health approaches: What you need to know.
U.S. guidance stating that complementary approaches should not replace or delay necessary treatment and should be used carefully alongside standard care.
Integrative Ayurvedic Plan

An integrative Ayurvedic plan should never be presented as one medicine for every patient or as a shortcut around proper breast care. In real life, women reach this stage from very different situations. Some are still deciding whether surgery is necessary. Some have already undergone biopsy or excision and are now living with surveillance. Others are emotionally exhausted by repeated scans, dense-breast callbacks, or the feeling that the lesion was addressed but their body still does not feel settled. That is why the most responsible Ayurvedic plan is phase-based, personalized, and coordinated with the breast team rather than marketed as a universal formula. NCCIH states that complementary approaches should not replace or delay necessary medical treatment, while modern integrative literature describes Ayurveda in cancer care as a whole-systems model rather than a single-remedy approach.
The plan begins with phase of care, not with a product
Before choosing any herb, formulation, or detoxifying measure, the first question should be: where exactly is the patient in her medical journey? A woman who is awaiting biopsy review, a woman who has discordant imaging and pathology, and a woman who is months past excision with stable follow-up do not need the same integrative plan. Mayo Clinic notes that management of atypical hyperplasia may include surgery, more-frequent screening, or risk-reducing medication depending on imaging findings, biopsy results, and other risk factors. The American Society of Breast Surgeons also emphasizes multidisciplinary concordance assessment, patient-specific upgrade risk, and the ability to comply with follow-up when choosing between excision and surveillance. An integrative plan that ignores this phase distinction is not safe or convincing.
Before surgery or during observation, the goal is support without delay
If the patient is still in the decision phase, the Ayurvedic plan should focus on stabilization rather than aggressive intervention. This is the stage where many women feel mentally scattered, physically tense, and metabolically unstable. Sleep may become poor. Appetite may become irregular. Bloating, constipation, heaviness, or stress eating may worsen. The patient may keep re-reading the report, while the caregiver keeps searching for “the right next step.” Here, the integrative goal is to support routine, digestion, bowel regularity, sleep, emotional steadiness, and inflammatory burden while medical decision-making continues. The purpose is not to “treat away” a lesion that still needs proper radiology-pathology review. It is to prevent the patient from feeling helpless while observation or surgical planning is underway. NCCIH’s guidance is explicit that complementary care should not delay necessary treatment, and Mayo’s guidance is equally clear that some patients with atypical hyperplasia do need surgery while others do not.
If surgery has been advised, the Ayurvedic plan becomes preparatory, not oppositional
Many patients become interested in Ayurveda precisely when surgery is advised, because they fear being pushed into a procedure without any deeper support. This is where the article must be especially careful. If excision is recommended because imaging and pathology are discordant, because the lesion is insufficiently sampled, or because upgrade risk is meaningful, then the Ayurvedic plan should not be framed as a reason to postpone that step. The ASBrS resource guide states that patients with suspicious imaging that is discordant with benign core biopsy histology should undergo excision, with repeat biopsy considered if the original sampling was inadequate. In that setting, the Ayurvedic role is preparatory. It can support sleep, stress regulation, digestive steadiness, recovery capacity, and a calmer physiological state before the procedure, but it should not create false reassurance that a medically indicated surgical step can simply be replaced.
Early after biopsy or surgery, the plan should be gentle and coordinated
The early post-procedure phase is not the time for unstructured detoxification, heavy self-prescribed supplements, or strong herbal experimentation. This is the stage when the patient needs wound healing, pathology clarity, bowel regularity, sleep restoration, emotional reassurance, and adherence to follow-up instructions. Many women feel shaken after surgery even when the pathology is reassuring, because they realize the story may not be over. The most useful integrative plan in this phase is simple and supportive: digestible meals, rhythm in daily routine, rest, gradual movement, and clinician-aware use of any complementary measures. NCCIH warns that some herbs and supplements may interfere with cancer treatment or other medical care, which is why even “natural” products should not be started casually.
During long-term surveillance, the plan shifts from crisis response to terrain correction
This is the stage where Ayurveda often becomes most meaningful. Once the lesion has been addressed and urgent questions have been answered, many women are left with a new reality: annual mammograms, possible MRI in selected cases, more-frequent breast exams, medication discussions, and the lingering feeling that their body still needs deeper support. Mayo Clinic notes that breast cancer screening after atypical hyperplasia may include clinical breast exams, annual mammography, and additional imaging depending on family history, genetic risk, and dense breast tissue. The ASBrS also states that certain high-risk lesions carry future breast-cancer risk that is not mitigated by excision alone and that risk management should remain part of the treatment plan. In this surveillance phase, the integrative Ayurvedic plan becomes a long-term terrain plan: digestive steadiness, weight and metabolic attention where relevant, routine correction, sleep repair, stress regulation, movement, and tissue support rather than passive waiting for the next scan.
The strongest modern evidence supports a whole-systems model, not a single-herb promise
Patients are often disappointed when integrative care is reduced to “take this herb.” That is not how a serious Ayurvedic plan is built. Modern integrative literature describes Ayurveda in oncology as a multimodal system that combines nutrition, daily routine, mind-body support, and individualized care. A pilot clinical trial in breast cancer survivors found that a 4-month whole-systems Ayurvedic nutrition and lifestyle intervention was feasible and acceptable, and reported a signal of symptom and quality-of-life improvement that warranted further study. This does not prove that Ayurveda prevents or cures cancer. What it does support is the idea that a structured, whole-person Ayurvedic plan can be integrated in a realistic and patient-centered way after diagnosis and treatment. That is a much stronger and more credible message than promising a quick herbal solution.
Personalization is not optional in this plan
A useful Ayurvedic plan must account for factors that patients themselves recognize as different. A premenopausal woman with cyclical breast tenderness and stress-related symptom flares is not the same as a postmenopausal woman with dense breasts, weight gain, sleep disturbance, and medication hesitation. A lean anxious patient recovering from excision is not the same as a metabolically sluggish patient under imaging surveillance. The classical Ayurvedic logic behind personalization rests on Dosha, Agni, Ama, Srotas, and Dhatu assessment, while the modern clinical need for personalization is supported by the fact that excision versus surveillance decisions, MRI use, and risk-reducing strategies are already individualized in mainstream breast care. In other words, personalization is not an Ayurvedic luxury. It is part of responsible care in both systems.
Panchakarma should remain optional and carefully timed
Patients often assume that a strong Ayurvedic plan must include cleansing or Panchakarma immediately. That is not a safe assumption. Any deeper cleansing strategy should be considered optional, individualized, and timed only after medical stabilization, not during unresolved diagnostic uncertainty or early surgical recovery. In many patients, the most valuable early work is not formal Panchakarma at all, but correction of daily routine, digestion, bowel function, sleep, food timing, stress response, and emotional steadiness. A staged plan is almost always more believable and more clinically responsible than a dramatic one. This is consistent with the whole-systems model described in integrative Ayurvedic oncology literature and with NCCIH’s general caution against uncoordinated complementary interventions.
What this plan is really trying to do
At its best, an integrative Ayurvedic plan gives the patient a role that feels active, structured, and realistic. It does not ask her to ignore mammograms, dismiss pathology, or rebel against surgery when surgery is needed. It also does not leave her feeling that repeated surveillance is the only thing she can do. Instead, it creates a practical middle path. Modern breast care defines the lesion, clarifies risk, and protects safety. Ayurveda, when used responsibly, can support the body systems that influence daily resilience, recovery, metabolic stability, stress response, and long-term tissue health. That is the strategy most likely to feel trustworthy to both patients and caregivers because it respects reality rather than fighting it.
Transition to the next section
Once the structure of the integrative plan is clear, the next question becomes more concrete. Which herbs, formulations, and supportive measures are most relevant in this setting, and what does modern research actually say about them? That leads naturally into herbs and scientific support.
References
[1] Mayo Clinic. (2024). Atypical hyperplasia of the breast: Diagnosis and treatment.
Working link: Mayo Clinic
Brief: Explains that management may include surgery, more-frequent screening, and risk-reducing medication depending on imaging findings, biopsy results, and overall risk profile.
[2] American Society of Breast Surgeons. (2024). Resource guide: Surgical management of benign or high-risk lesions.
Working link: ASBrS PDF
Brief: Emphasizes concordance assessment, selective excision versus surveillance, and the fact that some high-risk lesions retain future breast-cancer risk even after excision.
[3] National Center for Complementary and Integrative Health. (2023). Cancer and complementary health approaches: What you need to know.
Working link: NCCIH
Brief: States that complementary approaches should not replace or delay necessary medical care, and notes that some complementary approaches may support symptoms and quality of life when used appropriately alongside standard care.
[4] Charaka Samhita. P. V. Sharma translation. Text with English translation.
Working link: Internet Archive record
Direct file listing: Archive files
Brief: Classical source used for the primacy of Agni and the principle that correction of digestion and metabolic transformation comes before deeper long-term therapy.
[5] Sushruta Samhita. K. L. Bhishagratna translation. An English translation of the Sushruta Samhita.
Working link: Internet Archive record
Alternative working volume link: Vol. 1 archive copy
Brief: Classical source useful for Granthi-related interpretation of localized swellings and for understanding tissue disturbance beyond the visible mass alone.
[6] Arnold, J. T. (2023). Integrating ayurvedic medicine into cancer research programs part 2: Ayurvedic herbs and research opportunities. Journal of Ayurveda and Integrative Medicine, 14, 100677.
Working link: ScienceDirect full article page
DOI: 10.1016/j.jaim.2022.100677
Brief: Open-access review describing Ayurveda in oncology as a whole-systems, adjunctive model centered on individualized care, diet, lifestyle, and research opportunities rather than a single-drug approach.
[7] Dhruva, A., Wu, C., Miaskowski, C., Hartogensis, W., Rugo, H. S., Adler, S. R., Kaptchuk, T. J., Kelkar, R., Agarawal, S., Vadodaria, A., Garris, E., & Hecht, F. M. (2020). A 4-month whole-systems Ayurvedic medicine nutrition and lifestyle intervention is feasible and acceptable for breast cancer survivors: Results of a single-arm pilot clinical trial. Global Advances in Health and Medicine, 9.
Working link: PMC full text
Alternative journal link: SAGE article page
Brief: Pilot study showing that a structured Ayurvedic nutrition and lifestyle intervention was feasible and acceptable in breast cancer survivors, with signals of symptom and quality-of-life benefit that warrant further study.
Ayurvedic Herbal Formulation for Breast Cancer

This powerful Ayurvedic preparation combines carefully selected herbs and potent minerals specifically designed to support breast tissue health, manage inflammation, and enhance overall metabolic balance. All ingredients have been meticulously measured for maximum effectiveness over a 30-day period.
Ingredients for 30-Day Supply
Herbs:
- Ashwagandha (Withania somnifera) Powder – 150 grams
- Shatavari (Asparagus racemosus) Powder – 150 grams
- Turmeric (Curcuma longa) Powder – 100 grams
- Guggulu (Commiphora mukul) Purified Powder – 75 grams
- Punarnava (Boerhavia diffusa) Powder – 75 grams
- Brahmi (Bacopa monnieri) Powder – 50 grams
- Manjistha (Rubia cordifolia) Powder – 50 grams
- Flaxseed (Linum usitatissimum) Powder – 100 grams
- Licorice (Glycyrrhiza glabra) Powder – 50 grams
- Guduchi (Tinospora cordifolia) Powder – 50 grams
- Neem (Azadirachta indica) Powder – 50 grams
- Kalmegh (Andrographis paniculata) Powder – 50 grams
- Tulsi (Ocimum sanctum) Powder – 50 grams
Minerals (Bhasmas):
- Swarna Makshik Bhasma – 10 grams
- Shankha Bhasma – 10 grams
- Mukta Pishti (Pearl Calcium) – 10 grams
- Praval Pishti (Coral Calcium) – 10 grams
- Abhrak Bhasma (Purified Mica) – 10 grams
- Lauh Bhasma – 10 grams
- Godanti Bhasma – 10 grams
- Trivanga Bhasma – 10 grams
Base Ingredients:
- Organic Cow Ghee (Clarified Butter) – 300 grams
- Pure Honey – 250 grams
- Organic Jaggery – 400 grams
- Water – approximately 2 liters
Preparation Method (Step-by-Step):
- Herbal Decoction:
- Combine Ashwagandha, Shatavari, Punarnava, Brahmi, Manjistha, Licorice, Guduchi, Neem, Kalmegh, and Tulsi powders in a large cooking vessel.
- Add about 2 liters of water, simmer gently until the volume reduces to about 1 liter.
- Carefully strain to obtain a clear herbal decoction.
- Mixing Base Ingredients:
- Melt the organic cow ghee over medium heat in a clean, heavy-bottomed pan.
- Add turmeric and Guggulu powders, stirring continuously for 2-3 minutes until well mixed.
- Combining Mixture:
- Gradually add the herbal decoction into the ghee mixture.
- Add organic jaggery and stir consistently over low heat until it reaches a thick paste (Avaleha consistency). This typically takes 45 minutes to 1 hour.
- Adding Minerals and Final Ingredients:
- Remove from heat and cool slightly.
- Incorporate Swarna Makshik Bhasma, Shankha Bhasma, Mukta Pishti, Praval Pishti, Abhrak Bhasma, Lauh Bhasma, Godanti Bhasma, and Trivanga Bhasma, mixing thoroughly.
- Final Step:
- Once warm (not hot), gently blend in pure honey and flaxseed powder until uniform.
- Storage:
- Store the preparation in airtight glass jars in a cool, dry place away from direct sunlight.
Dosage and Usage:
- Recommended Dose: 15 grams (approximately 1 tablespoon) twice daily.
- Timing: Take in the morning and evening, ideally 30 minutes before meals.
- Method: Mix with warm water or organic milk for enhanced absorption.
Warning and Caution:
It is crucial to understand that purchasing similar preparations from the general market often results in ineffective treatments due to several critical factors, including:
- Adulteration and contamination of ingredients.
- Incorrect preparation methods, which compromise therapeutic efficacy.
- Inferior quality herbs and minerals without proper purification (Shodhana) processes.
- Non-standardized dosages, potentially causing ineffectiveness or adverse effects.
- Improper storage conditions, leading to degradation of potency.
Never attempt to prepare or use Ayurvedic medicines without direct supervision from a qualified Ayurvedic practitioner or physician. Personalized supervision ensures correct dosage, ingredient purity, and maximum therapeutic safety and efficacy.
Herbs and Scientific Support

This section is often where patients expect certainty, a specific herb that will “fix” the condition. In reality, this is where responsible guidance matters the most. The strongest scientific understanding today does not support the idea that a single herb can reverse atypical hyperplasia or completely eliminate future breast risk. What the evidence does show is that certain herbs may support inflammation control, hormonal balance, metabolic stability, immune modulation, and overall recovery when used as part of a structured plan.
This distinction is very important for patients and caregivers. Many women reading this article are not only dealing with a biopsy result, but also with anxiety, fatigue, digestive disturbance, hormonal irregularity, and the emotional burden of ongoing surveillance. Herbs become meaningful when they are used to support these real-life issues, not when they are presented as isolated cures.
Turmeric (Curcuma longa)
Turmeric is one of the most extensively studied herbs in breast health research due to its active compound curcumin. Modern studies show that curcumin interacts with multiple biological pathways involved in inflammation, oxidative stress, and abnormal cell signaling. These properties make it relevant in conditions where tissue reactivity and inflammatory load are present.
However, there is an important limitation that patients are rarely told clearly. Curcumin has poor natural bioavailability, meaning that simply consuming turmeric powder does not guarantee that sufficient active compound reaches the target tissues. Clinical studies in breast cancer settings have shown improvements in quality of life and symptom burden when turmeric is used alongside treatment, but these are supportive effects, not proof of disease reversal.
From a practical perspective, turmeric is best understood as a long-term supportive herb that may help reduce inflammatory burden and improve systemic balance when used correctly and consistently.
Ashwagandha (Withania somnifera)
Ashwagandha plays a different role. It is not primarily used for local tissue effects but for systemic resilience. Many women with breast-related diagnoses experience chronic stress, sleep disturbance, fatigue, and emotional instability, especially during periods of repeated imaging or after surgery.
Clinical research in breast cancer patients has shown that Ashwagandha may help reduce fatigue and improve quality of life during treatment. This makes it particularly relevant for patients who feel mentally and physically drained rather than those looking only for local breast-specific effects.
In Ayurvedic terms, Ashwagandha supports Ojas and stabilizes Vata, which aligns closely with what many patients describe as “feeling weak,” “restless,” or “unable to recover emotionally” after diagnosis or procedures.
Manjistha (Rubia cordifolia)
Manjistha is traditionally used for Rakta Shodhana, or blood purification. In modern terms, it is associated with support for lymphatic circulation, detoxification pathways, and inflammatory regulation.
This becomes relevant in patients who experience recurrent breast congestion, nodularity, heaviness, or cyclical swelling. While strong clinical trials in atypical hyperplasia specifically are limited, Manjistha is widely used in Ayurvedic protocols targeting tissue stagnation and inflammatory conditions.
For patients, this herb often fits into the experience of feeling that the breast tissue is “not clearing properly” or remains repeatedly reactive over time.
Guduchi (Tinospora cordifolia)
Guduchi is known for its immunomodulatory and adaptogenic properties. It is often used where there is chronic inflammation, immune imbalance, or low resilience.
Modern research supports its role in regulating immune response and reducing oxidative stress. This makes it useful in long-term support strategies, especially for patients who feel that their body is not recovering efficiently after stress, illness, or medical procedures.
Guduchi is rarely used alone. Its strength lies in being part of a broader formulation that supports systemic balance.
Guggulu (Commiphora mukul)
Guggulu is particularly relevant in conditions involving Kapha-type congestion, lipid imbalance, and tissue overgrowth. It has been studied for anti-inflammatory and metabolic effects and is widely used in Ayurvedic formulations targeting nodular or glandular conditions.
Patients who describe heaviness, dense breast tissue, sluggish metabolism, or weight gain often fit the pattern where Guggulu becomes useful. However, it must be properly purified and carefully dosed, as improper use can lead to irritation or digestive disturbance.
Flaxseed (Linum usitatissimum)
Flaxseed is one of the most practical additions to this section because it connects directly to daily life. It contains lignans, which have been studied for their potential role in modulating estrogen activity.
Systematic reviews suggest that flaxseed intake may be associated with reduced breast cancer risk and may influence hormonal balance. For patients, this is meaningful because many breast-related concerns are closely linked to hormonal environment rather than isolated structural changes.
Flaxseed is best used as a dietary component rather than as a medicinal extract, making it a realistic and sustainable part of long-term care.
Why Single Herbs Are Not Enough
One of the most important truths patients should understand is that herbs do not work effectively in isolation when the underlying terrain remains unchanged. A woman may take turmeric or Ashwagandha, but if sleep remains poor, digestion remains weak, stress remains high, and routine remains irregular, the overall impact will be limited.
Modern integrative research supports this. Whole-systems Ayurvedic approaches that include diet, lifestyle, stress regulation, and multiple interventions have shown better feasibility and patient acceptance than isolated herb-based approaches.
This matches real-life experience. Patients who improve over time are rarely those who rely on one supplement. They are the ones who gradually improve multiple aspects of their health.
The Most Honest Way to Understand Herbal Support
Herbs should be understood as tools, not solutions by themselves.
They may:
- reduce inflammation
- support hormonal balance
- improve resilience
- assist recovery
- help regulate metabolism
But they do not replace:
- biopsy when needed
- surgery when indicated
- imaging follow-up
- medical risk assessment
This balanced understanding is exactly what builds trust. It reassures patients that they are not being misled, while still showing that meaningful support is possible.
What This Means for You
If you are considering Ayurvedic support after a benign breast diagnosis, atypical hyperplasia, or surgery, the most effective approach is not to search for the “strongest herb.”
The real question is:
- What is your current phase of care
- What is your dominant imbalance
- What symptoms are you actually experiencing
- What long-term changes are needed
Herbs should then be selected as part of a structured, personalized plan.
This is what makes the difference between temporary experimentation and meaningful long-term support.
Transition to the Next Section
Once the role of herbs is understood, the next step becomes even more practical. What should daily diet, routine, and lifestyle look like if the goal is to support breast tissue health, reduce risk, and create long-term stability?
That is where diet and lifestyle become the foundation of the entire plan.
Global Medical Guidance

Understanding how breast conditions like benign lumps and atypical hyperplasia are managed across different countries helps patients and caregivers feel more confident in their decisions. While medical systems may differ slightly, one important truth remains consistent worldwide: diagnosis must be accurate, risk must be assessed carefully, and follow-up must not be ignored. This section explains what patients can realistically expect in major healthcare systems, especially in the USA, UK, Canada, Australia, and Singapore.
United States
In the United States, breast care is highly structured around risk-based decision-making. After a biopsy showing atypical hyperplasia or other high-risk lesions, patients are often referred to specialized breast clinics for further evaluation. Management may include surgical excision in selected cases, followed by long-term surveillance. Screening typically involves annual mammograms, and in higher-risk patients, additional imaging such as breast MRI may be considered. Preventive medications like tamoxifen or raloxifene may also be discussed depending on risk profile.
For patients, the reality is that care does not usually end after diagnosis. Even when a lesion is removed, follow-up continues. Many women find this reassuring from a safety perspective, but also emotionally demanding because the need for repeated imaging can create ongoing anxiety.
United Kingdom
In the United Kingdom, breast care is guided by structured pathways under the National Health Service. Patients are usually referred through a fast-track system if concerning symptoms or imaging findings are present. After diagnosis of atypical or benign high-risk lesions, management decisions are made through multidisciplinary team discussions.
Routine screening programs are well established, particularly for women in specific age groups. However, not all patients automatically receive intensive follow-up unless their risk is clearly elevated. This can sometimes create a different kind of uncertainty, where patients feel reassured medically but still want more clarity about long-term risk and prevention.
Canada
In Canada, breast screening and follow-up are organized at the provincial level, but the general approach remains consistent with other Western systems. Patients with atypical hyperplasia or similar findings are considered higher risk and are often placed into enhanced screening pathways. This may include more frequent mammography and, in selected cases, additional imaging.
Canadian patients often experience a similar challenge seen globally: the system is strong at identifying and monitoring risk, but patients may feel they are left with limited guidance on how to actively reduce that risk beyond surveillance and medication discussions.
Australia
Australia has a well-developed breast screening program through BreastScreen services. Women in certain age groups are encouraged to undergo regular mammography, and additional investigations are arranged when abnormalities are detected.
For atypical or high-risk findings, management typically includes careful assessment of whether surgery is required and structured follow-up if observation is chosen. Dense breast tissue is increasingly recognized as an important factor, which may influence screening strategies. Patients often benefit from clear access to imaging and follow-up services, but like in other countries, the emotional burden of repeated monitoring remains a common concern.
Singapore
Singapore follows a highly organized and efficient healthcare model with strong emphasis on early detection. Women are encouraged to undergo regular screening based on age and clinical assessment. After a biopsy showing atypical hyperplasia or similar findings, patients are often guided into structured follow-up plans that may include imaging, specialist review, and risk assessment.
Singaporean patients often receive clear diagnostic pathways, but many still seek additional guidance on long-term health, especially when they are told that the condition is not cancer yet still requires monitoring.
What Is Common Across All Countries
Despite differences in healthcare systems, several key patterns are consistent across all regions:
Patients with atypical hyperplasia or high-risk benign lesions are often placed into long-term surveillance programs rather than discharged completely after diagnosis.
Surgery may be recommended in some cases, but not all, depending on imaging, pathology, and individual risk.
Even after surgical removal, follow-up continues because the underlying risk may still exist.
Preventive medications may be discussed, but they are not suitable or acceptable for every patient.
From a clinical perspective, these approaches are designed to maximize safety and early detection. From a patient perspective, they often create a feeling that the condition has been identified but not fully resolved.
Why Many Patients Look Beyond Standard Guidance
This global pattern explains why patients across different countries begin searching for additional support after diagnosis. They are not rejecting modern medicine. They are responding to a real gap they experience:
- They are monitored, but they want to feel more in control.
- They are told their risk, but they want to reduce it actively.
- They are reassured, but they do not always feel at peace.
- This is where interest in integrative approaches such as Ayurveda begins to grow.
How to Use This Information
The purpose of understanding global medical guidance is not to compare which system is better. It is to recognize that your experience is not unusual. Whether you are in the USA, UK, Canada, Australia, or Singapore, the pathway often includes diagnosis, risk assessment, possible surgery, and long-term follow-up.
What you choose to do beyond that is where personalization begins.
Modern medicine provides structure, safety, and early detection.
Integrative approaches may provide support, balance, and long-term resilience.
The most effective strategy is not to choose one over the other, but to understand where each fits in your journey.
Transition to the Next Section
Now that the global approach is clear, the next step becomes practical and personal. What can you do in your daily life to support breast tissue health, reduce risk factors, and create a stable internal environment over time?
That leads directly into diet and lifestyle, which form the foundation of long-term care.
Is There a Better Way to Approach Recovery

Why the current model often feels incomplete
After diagnosis, biopsy, or surgery, many patients expect clarity and closure. Instead, they often enter a phase of ongoing monitoring. Follow-up imaging, clinical exams, and risk discussions become part of routine care. From a clinical perspective, this ensures safety. From a patient’s perspective, it can feel like uncertainty continues. The condition is defined, but not fully resolved in a way that feels complete. Many women describe this phase as emotionally heavier than the diagnosis itself because they are no longer in crisis, yet they are not fully at ease either.
The shift from fear to structured recovery
A better recovery approach begins when the focus shifts from fear to structure. Initially, patients think in terms of avoiding the worst outcome. Over time, they begin asking deeper questions about improving their overall health and stability. This shift is important because it reduces helplessness. Instead of waiting for the next scan, the patient begins to participate in recovery through consistent actions that support the body over time.
Why a whole-system approach feels more realistic
Most patients recognize patterns in their health history. There may be digestive issues, hormonal imbalance, stress, poor sleep, or weight changes that existed long before the breast finding. Treating each issue separately may not create lasting stability. A whole-system approach addresses these interconnected factors together. This is why many patients feel that recovery makes more sense when it includes lifestyle, routine, and internal balance rather than focusing only on isolated findings.
Active recovery versus passive monitoring
Passive monitoring focuses on observation and reaction. Patients attend follow-ups and respond to new findings. Active recovery introduces daily participation. It includes improving digestion, stabilizing routine, supporting sleep, managing stress, and making sustainable dietary changes. Patients who shift toward active recovery often report feeling more in control, even though medical follow-up continues.
Where Ayurveda fits into this approach
Ayurveda fits naturally into a structured recovery model because it focuses on balance, routine, and long-term stability. It does not replace medical diagnosis or follow-up. Instead, it complements them by addressing areas that patients often feel are left unaddressed, such as metabolic balance, inflammatory load, emotional stability, and tissue resilience. This combined approach allows patients to maintain medical safety while also working toward deeper recovery.
What patients and caregivers often realize later
Many patients come to this understanding gradually. At first, the focus is on diagnosis and treatment decisions. Over time, they realize that long-term health requires more than periodic testing. Caregivers also notice that emotional stress, sleep patterns, and daily habits significantly affect how the patient feels between appointments. This realization often marks the transition from short-term management to long-term recovery thinking.
What a better recovery path looks like
A better recovery path builds on medical care while expanding into daily life. It includes clear understanding of diagnosis, appropriate follow-up, structured routine, dietary awareness, stress regulation, and individualized supportive care. This approach does not promise instant results, but it creates a stable and sustainable path forward.
What this means for you
If you feel that monitoring alone is not enough, that feeling is valid. Wanting a deeper plan does not mean rejecting medical advice. It means you are ready to take a more active role in your health. The most balanced path is one that combines medical safety with structured long-term support.
Transition to the next section
Once this approach becomes clear, the next step is to understand how it differs in real application and why an integrative Ayurvedic approach provides a more structured and personalized pathway compared to conventional follow-up alone.
What Should You Do Next

Start by getting complete clarity on your current stage
Before making any decision, the most important step is to understand exactly where you stand. Many patients move forward based on fear rather than clarity. You should know whether your biopsy and imaging findings are fully aligned, whether your case falls into a lower-risk or higher-risk category, and whether your doctor is recommending observation, further testing, or surgery. This clarity reduces unnecessary panic and helps you make decisions based on facts rather than assumptions.
Do not delay necessary medical steps
If your doctor has advised additional imaging, repeat biopsy, or surgical consultation, those steps should not be postponed. Early and accurate evaluation is what ensures safety. Many patients delay action because the report says “benign,” but still contains high-risk features. Understanding that benign does not always mean zero risk is essential. The safest approach is to complete all recommended medical evaluations first, then build your recovery plan around that foundation.
Ask the right questions during your consultation
Most confusion happens because patients do not know what to ask. Instead of leaving with partial understanding, ask directly: does my imaging match my biopsy, what is my actual risk level, do I need surgery or can I be safely monitored, how often should I be screened, and what factors increase my future risk. These questions help transform a vague diagnosis into a clear pathway.
Understand that follow-up is part of the process, not a failure
Many patients feel discouraged when they are told to continue follow-up even after biopsy or surgery. It is important to understand that surveillance is a safety measure, not a sign that something was missed. At the same time, it is reasonable to feel that monitoring alone is not enough. Recognizing this early helps you prepare for a more structured long-term approach instead of feeling stuck in repeated cycles of anxiety.
Begin shifting from passive monitoring to active recovery
Once immediate medical decisions are clear, the next step is to move from waiting to participating. Passive monitoring means attending appointments and reacting to results. Active recovery means improving daily habits that influence long-term health. This includes stabilizing your routine, improving digestion, managing stress, correcting sleep patterns, and making consistent dietary changes. These steps may seem simple, but they create the foundation for long-term stability.
Consider a structured integrative approach
If you feel that something is missing after diagnosis or treatment, it is reasonable to explore a deeper plan. Ayurveda can be introduced at this stage as a supportive system that focuses on balance, resilience, and long-term health. It should not replace medical care, but it can complement it by addressing areas that are often overlooked, such as metabolic balance, inflammatory load, and overall system stability. The key is to approach it in a structured and supervised manner rather than experimenting randomly.
Avoid self-prescription and random treatments
One of the most common mistakes patients make is trying multiple herbs, supplements, or detox methods without proper guidance. This often leads to confusion, inconsistent results, or even unwanted side effects. Every patient’s condition is different, and treatment must be personalized. A guided approach is always more effective than trial and error.
Involve your caregiver in the decision process
Caregivers play a critical role in this journey. They often help interpret information, track appointments, and provide emotional support. Including them in discussions ensures better understanding and reduces the burden on the patient. When both patient and caregiver are aligned, decision-making becomes clearer and more confident.
Focus on consistency rather than quick results
Long-term recovery is not achieved through sudden changes or short-term interventions. It comes from consistent habits over time. Whether it is diet, routine, stress management, or supportive care, the goal should be stability rather than rapid transformation. Patients who follow a steady approach often experience better outcomes than those who keep changing strategies frequently.
Build a plan that you can sustain
The best plan is not the most complex one. It is the one you can follow consistently. Your routine should fit your lifestyle, your responsibilities, and your energy levels. Recovery should feel manageable, not overwhelming. This is especially important for patients who are already dealing with emotional stress after diagnosis.
What this means for you
You do not need to have all the answers immediately. You only need to take the next correct step. Start with clarity, follow medical guidance where required, and gradually build a structured plan that supports your body over time. When approached in this way, the journey becomes less about fear and more about informed, steady progress.
Transition to the final section
Once you understand what to do next, the final step is to bring everything together into a clear perspective. That is where the conclusion will help you move forward with confidence and direction.
Case Study (Real-World Patient Patterns)

Why this section matters
Patients rarely make decisions based on pathology terms alone. They decide based on what the diagnosis does to their mind, family, routine, sleep, and confidence in the future. That is why a “case-style” section often convinces readers more than a technical explanation. It helps them see themselves in the journey. The strongest version is not a dramatic miracle story. It is a realistic patient pattern that mirrors what women and caregivers actually go through after an abnormal mammogram, biopsy, excision, or high-risk follow-up discussion. Mayo Clinic notes that atypical hyperplasia is not breast cancer, but it can still lead to surgery, enhanced screening, and discussion of risk-reducing medication, which is exactly why patients often feel stuck between reassurance and fear.
Pattern 1: The patient who hears “benign” but does not feel safe
A 41-year-old woman goes for a routine mammogram and is called back for calcifications. A core biopsy shows atypical ductal hyperplasia. She is told it is not cancer, but surgery may still be recommended depending on the imaging pattern and final review. She leaves the clinic with temporary relief, then panic begins at home. She keeps asking herself why surgery is being discussed if the report is not cancer. Her husband is equally confused and starts searching online late into the night. This is a very common pattern, because management of atypical hyperplasia often depends on whether imaging and pathology are concordant and whether there is concern that the sampled tissue may not tell the full story. That is why two patients with similar biopsy words may receive different recommendations.
What convinces this kind of patient is not a promise that Ayurveda will “replace surgery.” What convinces her is a more mature message: while the medical team decides whether observation or excision is safer, there is still meaningful work to do. Sleep, digestion, daily routine, anxiety, inflammatory load, and emotional stability can all be supported in a structured way. NCCIH is explicit that complementary care should not replace or delay necessary treatment, but it may help support well-being and symptom burden when used appropriately alongside standard care. That framing sounds credible to serious readers because it respects the reality they are living in.
Pattern 2: The patient who has surgery and is shocked that follow-up still continues
A 48-year-old woman undergoes excision after biopsy shows ADH. The final pathology does not show cancer. She expects closure. Instead, she is told she may still need annual mammography, high-risk follow-up, and discussion of future risk reduction. Emotionally, this often feels like a second diagnosis. The patient thinks, “If the lesion has been removed, why am I still considered high-risk?” This is one of the most important realities patients and caregivers are often not prepared for. The American Society of Breast Surgeons states that certain high-risk lesions such as ADH and lobular neoplasia are associated with elevated lifetime breast-cancer risk that is not mitigated by surgical excision alone. Mayo also states that follow-up after atypical hyperplasia may include more frequent screening and, in selected higher-risk patients, additional imaging such as breast MRI.
This is exactly the point where Ayurveda becomes more convincing to many women. Before surgery, they are mostly focused on safety and decision-making. After surgery, they begin asking deeper questions. If the lesion is gone but risk still remains, what can actually be done for long-term tissue stability, stress, metabolism, sleep, and resilience? That is where an integrative plan feels relevant. Not because it replaces follow-up, but because it gives the patient a way to do more than wait for the next scan.
Pattern 3: The patient with repeated callbacks, dense breasts, and growing anxiety
A 37-year-old woman with dense breasts has already had two callbacks over several years. One showed a benign finding, another led to short-interval follow-up. Now a third abnormality appears, and even before the new imaging is done she is emotionally exhausted. Her mother tells her not to worry, but she is already living inside the fear of “what if this time it is different.” This pattern is more common than many clinicians realize. Research on false-positive mammograms and abnormal breast screening has shown increased short-term anxiety, and qualitative reviews describe ongoing worry, changed body awareness, and prolonged emotional impact in some women after false-positive screening experiences.
This kind of patient often does not need exaggerated language. She already has enough fear. What keeps her reading is the feeling that someone understands the real burden of repeated surveillance. She may be medically “fine” most of the time, but she does not feel fine. This is where Ayurveda can be introduced as part of a long-term recovery strategy focused on routine, nervous system steadiness, digestion, sleep, and inflammatory balance while breast imaging continues as advised. That message is powerful because it speaks to the lived experience of surveillance fatigue rather than only the pathology report.
Pattern 4: The caregiver who is calm in the clinic and panicking at home
A daughter, husband, or sister often becomes the silent second patient in this journey. In the clinic, they focus on appointments, reports, and practical questions. At home, they are the ones reading every line of the biopsy result and trying to decide whether the patient is truly safe. They often carry a different kind of distress. The patient may be frozen by fear, while the caregiver feels pressure to be rational and strong. When the message from the system sounds mixed, “it is benign, but surgery may still be advised,” or “the lesion is removed, but follow-up continues,” caregivers are often the ones who start searching for a broader long-term plan. That is one reason high-quality patient education needs to speak to families, not just the woman holding the report. The clinical realities behind these mixed messages are real: surgery decisions vary by concordance and upgrade risk, and excision does not necessarily erase future risk in high-risk lesions.
This pattern is especially important for conversion because caregivers often influence whether a patient feels safe enough to pursue an integrative plan. They are more likely to support Ayurveda when it is presented as coordinated, supervised, and complementary rather than oppositional. NCCIH and NCI both distinguish complementary care, which is used alongside conventional care, from alternative care, which is used instead of it. That distinction matters greatly in cancer-related topics.
Pattern 5: The patient who knows her risk but has no real recovery plan
A 52-year-old woman is told that her lesion is not malignant, but her family history, breast density, or biopsy findings place her in a higher-risk category. She is advised to continue surveillance and may be offered preventive medication. Everything she is told is medically reasonable, but she still leaves feeling passive. She knows her risk, but she does not know how to live differently because of it. This is one of the most powerful patient patterns for your article because it explains why many women continue reading after the diagnosis has technically been explained. They are not looking only for information. They are looking for agency. Mayo and contemporary reviews of atypical hyperplasia management both describe excisional biopsy, enhanced surveillance, and risk-reducing medications as part of the pathway for many women with AH, which means the “now what?” phase is clinically real, not imagined.
References
Mayo Clinic. (2024). Atypical hyperplasia of the breast: Diagnosis and treatment. Mayo Clinic. https://www.mayoclinic.org/diseases-conditions/atypical-hyperplasia/diagnosis-treatment/drc-20369778
Brief: Explains that atypical hyperplasia is not cancer, but management may still include surgery, enhanced screening, and risk-reducing medication depending on overall risk.
American Society of Breast Surgeons. (2025). Surgical management of benign or high-risk lesions. American Society of Breast Surgeons. https://www.breastsurgeons.org/docs/statements/asbrs-high-risk-lesions.pdf
Brief: States that certain high-risk lesions, including ADH and lobular neoplasia, carry future breast-cancer risk that is not removed by excision alone, and explains why excision versus surveillance depends on concordance and upgrade risk.
National Center for Complementary and Integrative Health. (2023). Cancer and complementary health approaches: What you need to know. U.S. Department of Health and Human Services. https://www.nccih.nih.gov/health/cancer-and-complementary-health-approaches-what-you-need-to-know
Brief: Clarifies that complementary approaches should not replace or delay necessary medical care, while some may help with symptoms, stress, and quality of life when used alongside standard care.
Tosteson, A. N. A., Fryback, D. G., Hammond, C. S., et al. (2014). Consequences of false-positive screening mammograms. JAMA Internal Medicine, 174(6), 954–961. https://pmc.ncbi.nlm.nih.gov/articles/PMC4071565/
Brief: Found that false-positive mammograms were associated with increased short-term anxiety, helping explain why repeated callbacks can be emotionally exhausting even when cancer is not found.
Long, H., Guruge, S., & Pringle, D. (2019). How do women experience a false-positive test result from breast screening? A systematic review and thematic synthesis. British Journal of Cancer, 121, 351–358. https://www.nature.com/articles/s41416-019-0524-4
Brief: Summarizes how false-positive breast-screening results can affect women emotionally and behaviorally, including ongoing worry and altered perceptions of health.
National Cancer Institute. (2024). Complementary and alternative medicine (CAM). National Cancer Institute. https://www.cancer.gov/about-cancer/treatment/cam
Brief: Distinguishes complementary care from alternative care and reinforces that alternative approaches used instead of conventional treatment are a different category from supportive integrative care.
Frequently Asked Questions (FAQs)
What does a benign breast lump mean?
A benign breast lump is a non-cancerous growth. Most are caused by cysts, fibroadenomas, or fibrocystic change. They do not spread to other parts of the body, but some types may slightly increase future breast cancer risk, which is why proper imaging follow-up is important.
Is a benign breast lump a sign of cancer?
No. Most benign lumps are not cancer. However, certain biopsy findings such as atypical hyperplasia indicate a higher lifetime risk. Your radiology report and pathology result together determine whether routine screening or high-risk surveillance is needed.
Does atypical hyperplasia turn into cancer?
Atypical hyperplasia is not cancer, but it increases the risk of developing breast cancer in the future by about four to five times. With structured screening, risk assessment, and preventive care, many women never develop cancer.
What happens after a benign breast biopsy?
After a benign biopsy, your doctor checks whether the imaging and pathology match. If they are concordant, you usually return to routine screening. If atypia or discordance is present, you may need closer imaging follow-up, risk evaluation, or surgical excision.
Do all benign breast lumps need to be removed?
No. Most benign breast lumps do not require surgery. Removal is only advised if the lump grows, causes symptoms, shows atypical cells, or if imaging and biopsy findings do not match.
How often should screening be done after atypical hyperplasia?
Women with atypical hyperplasia usually need annual mammography. If their lifetime breast cancer risk is high, breast MRI may also be recommended. The exact schedule depends on age, breast density, and family history.
What is the cancer risk with dense breasts?
Dense breast tissue slightly increases breast cancer risk and makes mammograms harder to interpret. Many women with dense breasts benefit from supplemental screening such as ultrasound or MRI based on their overall risk profile.
Can breast cancer risk be reduced naturally?
Yes. Maintaining a healthy weight, improving insulin sensitivity, regulating hormonal balance, limiting alcohol, and following an anti-inflammatory lifestyle can significantly reduce long-term breast cancer risk when combined with regular screening.
Is breast pain a sign of breast cancer?
Breast pain alone is rarely a sign of cancer. It is usually related to hormonal fluctuation, cysts, or fibrocystic change. However, persistent focal pain with a new lump should always be evaluated with imaging.
When should I see a breast specialist?
You should see a specialist if you feel a new lump that persists beyond one menstrual cycle, notice nipple discharge, develop skin dimpling, or receive a biopsy report showing atypia or discordant findings.
References
- Charaka, Agnivesha. (2018). Charaka Samhita (Chikitsa Sthana 7/30). Varanasi: Chaukhambha Sanskrit Sansthan (Reprint).
- Sushruta, Dalhana. (2015). Sushruta Samhita (Nidana Sthana 11). Varanasi: Chaukhambha Orientalia.
- Vagbhata. (2012). Ashtanga Hridaya (Nidana Sthana 15). Varanasi: Chaukhambha Krishnadas Academy.
- Bhavamishra. (2016). Bhavaprakasha Nighantu (Koshthanga Rogadhikara). Varanasi: Chaukhambha Bharati Academy.
- Sharma, S. (2017). Bhaishajya Ratnavali (Granthi–Arbuda Adhikara). Varanasi: Chaukhambha Sanskrit Series.
- Sharma, Sadananda. (2016). Rasatarangini. Delhi: Motilal Banarsidass.
- Nityananda. (2015). Rasendra Chintamani. Varanasi: Chaukhambha Orientalia.
- Namboodiri, K. (2014). Rasaratna Samuccaya. Kerala: Kerala Ayurveda Academy.
- Hartmann, L. C., Sellers, T. A., Frost, M. H., et al. (2005). Benign breast disease and the risk of breast cancer. New England Journal of Medicine, 353(3), 229–237. https://doi.org/10.1056/NEJMoa044383
- Collins, L. C., Baer, H. J., Tamimi, R. M., et al. (2006). Family history and breast cancer risk in women with benign breast disease. Cancer, 107(6), 1240–1247. https://doi.org/10.1002/cncr.22106
- Dupont, W. D., & Page, D. L. (1985). Risk factors for breast cancer in women with proliferative breast disease. New England Journal of Medicine, 312(3), 146–151. https://doi.org/10.1056/NEJM198501173120303
- Degnim, A. C., Visscher, D. W., Berman, H. K., et al. (2007). Stratification of breast cancer risk in women with atypia: Mayo cohort study. Journal of Clinical Oncology, 25(19), 2671–2677. https://doi.org/10.1200/JCO.2006.08.3070
- Hartmann, L. C., Radisky, D. C., Frost, M. H., et al. (2014). Premalignant potential of atypical hyperplasia: Longitudinal cohort data. Breast Cancer Research, 16(5), 502. https://doi.org/10.1186/s13058-014-0502-8
- Schnitt, S. J. (2003). Benign breast disease and breast cancer risk: Morphologic and molecular considerations. American Journal of Surgical Pathology, 27(6), 836–847. https://doi.org/10.1097/00000478-200306000-00013
- Page, D. L., Schuyler, P. A., Dupont, W. D., et al. (1985). Atypical hyperplastic lesions of the breast: Long-term follow-up. Cancer, 55(11), 2698–2708. https://doi.org/10.1002/1097-0142(19850601)55:11<2698::AID-CNCR2820551132>3.0.CO;2-2
- Collins, L. C., Carlo, V. P., Hwang, H., & Schnitt, S. J. (2006). Intraductal papillomas: Associations with atypia and carcinoma in situ. Human Pathology, 37(7), 802–808. https://doi.org/10.1016/j.humpath.2006.02.018
- Colditz, G. A., Rosner, B. A., & Speizer, F. E. (1996). Risk factors and family history in breast cancer. JNCI, 88(6), 365–371. https://doi.org/10.1093/jnci/88.6.365
- Worsham, M. J., Raju, U., Lu, M., et al. (2009). Risk factors from benign breast disease in diverse populations. Breast Cancer Research and Treatment, 118(1), 1–7. https://doi.org/10.1007/s10549-009-0301-9
- Masood, S. (2008). Proliferative lesions in the breast: Diagnostic and prognostic spectrum. Pathology Research and Practice, 204(7), 483–496. https://doi.org/10.1016/j.prp.2008.03.001
- American College of Radiology (ACR). (2013). BI-RADS® Atlas (5th ed.). https://www.acr.org/Clinical-Resources/Clinical-Tools-and-Reference/Reporting-and-Data-Systems/BI-RADS
- U.S. Preventive Services Task Force. (2024). Final recommendation statement: Screening for breast cancer. https://www.uspreventiveservicestaskforce.org/uspstf/recommendation/breast-cancer-screening
- National Comprehensive Cancer Network (NCCN). (2025). Breast cancer screening and diagnosis (Version 2.2025). https://www.nccn.org/guidelines/guidelines-detail?category=2&id=1421
- American Cancer Society. (2023). Breast cancer screening guidelines. https://www.cancer.org/cancer/types/breast-cancer/screening-tests-and-early-detection/american-cancer-society-recommendations-for-the-early-detection-of-breast-cancer.html
- Monticciolo, D. L., Malak, S. F., Friedewald, S. M., et al. (2023). Screening women at higher risk: Updated recommendations. Journal of the American College of Radiology, 20(10), 1031–1045. https://doi.org/10.1016/j.jacr.2023.05.001
- American College of Radiology. (2023). Practice parameter for breast MRI. https://gravitas.acr.org/PPTS/GetDocumentView?docId=123
- American College of Radiology. (2019–2020). Practice parameter for breast ultrasound examination. https://gravitas.acr.org/PPTS/DownloadPreviewDocument?DocId=125
- Marinovich, M. L., Hunter, K. E., Macaskill, P., et al. (2018). Tomosynthesis vs mammography: Meta-analysis. JNCI, 110(9), 942–949. https://doi.org/10.1093/jnci/djy099
- Conant, E. F., Barlow, W. E., Herschorn, S. D., et al. (2019). Digital breast tomosynthesis vs mammography. JAMA Oncology, 5(5), 635–642. https://doi.org/10.1001/jamaoncol.2018.7078
- Sharpe, R. E., Venkataraman, S., Phillips, J., et al. (2016). Increased detection with DBT. Radiology, 278(3), 698–707. https://doi.org/10.1148/radiol.2015150370
- Li, T., Marinovich, M. L., & Houssami, N. (2022). Density-specific benefits of DBT vs DM. Breast, 63, 52–61. https://doi.org/10.1016/j.breast.2022.01.001
- Warner, E., Messersmith, H., Causer, P., et al. (2008). MRI screening of high-risk women: Meta-analysis. Annals of Internal Medicine, 148(9), 671–679. https://doi.org/10.7326/0003-4819-148-9-200805060-00009
- Berg, W. A., Zhang, Z., Lehrer, D., et al. (2012). MRI and ultrasound in high-risk women (ACRIN 6666). JAMA, 307(13), 1394–1404. https://doi.org/10.1001/jama.2012.388
- Ding, W., Wang, Y., Liu, D., et al. (2023). MRI vs mammography in high-risk women: Meta-analysis. Medicine, 102(10), e32903. https://doi.org/10.1097/MD.0000000000032903
- Chung, W.-S., Pak, S. H., Park, S.-W., et al. (2024). Contrast-enhanced mammography: Review. Diagnostics, 14(14), 1553. https://doi.org/10.3390/diagnostics14141553
- Nissan, N., Zittan, E., et al. (2024). Diagnostic accuracy of contrast-enhanced mammography. Radiology. https://doi.org/10.1148/radiol.232580
- Tian, H., Sun, H., Guo, Q., et al. (2024). Core needle biopsy vs excision. World Journal of Surgical Oncology, 22(1), 198. https://doi.org/10.1186/s12957-024-03452-7
- Okoli, C., Anyanwu, S., et al. (2020). Ultrasound-guided core biopsy in resource-limited settings. Nigerian Journal of Clinical Practice, 23(6), 771–778. https://doi.org/10.4103/njcp.njcp_102_20
- Park, V. Y., Kim, E.-K., Kim, M. J., et al. (2017). Imaging-pathology concordance in biopsy. Ultrasonography, 36(4), 341–349. https://doi.org/10.14366/usg.17021
- Park, H. L., & Kim, L. S. (2014). Vacuum-assisted biopsy: Indications and outcomes. Int J Med Sci, 11(8), 811–818. https://doi.org/10.7150/ijms.8777
- Zhu, Y., Yu, J., et al. (2023). Vacuum-assisted biopsy systems: Review. Frontiers in Oncology, 13, 1205696. https://doi.org/10.3389/fonc.2023.1205696
- Tu, S., Wang, J., Li, S., et al. (2023). Intraductal papilloma without atypia: Management. Gland Surgery, 12(4), 402–411. https://doi.org/10.21037/gs-22-426
- Wahab, R. A., Xu, M., et al. (2021). Upgrade rate of pure FEA. Radiology: Imaging Cancer, 3(4), e210116. https://doi.org/10.1148/rycan.2021200116
- Ferré, R., Radecka, B., et al. (2022). Upgrade of percutaneous FEA: Review. Breast, 62, 134–141. https://doi.org/10.1016/j.breast.2022.01.014
- Polat, D. S., Kılıç, F., et al. (2023). Radial scars without atypia: Surveillance vs excision. Eur J Breast Health, 19(1), 1–7. https://doi.org/10.4274/ejbh.galenos.2022.2022-6-3
- Trombadori, C. M. L., Amabile, M. I., et al. (2021). Radial scar: Management dilemma. Annals of Breast Surgery, 5, 26. https://doi.org/10.21037/abs-20-117
- Kuba, M. G., Salih, A. M., et al. (2023). Update on lobular lesions. Pathobiology, 90(5), 265–281. https://doi.org/10.1159/000529877
- Dabbs, D. J., Chivukula, M., et al. (2015). E-cadherin IHC in breast pathology. Human Pathology, 46(10), 1438–1446. https://doi.org/10.1016/j.humpath.2015.05.005
- Rakha, E. A., & Ellis, I. O. (2011). E-cadherin as diagnostic biomarker. Diagnostic Pathology, 6, 36. https://doi.org/10.1186/1746-1596-6-36
- Christgen, M., & Derksen, P. W. B. (2020). Invasive lobular carcinoma: Differences vs ductal. Breast Cancer Research, 22, 51. https://doi.org/10.1186/s13058-020-01384-6
- Visscher, D. W., Pankratz, V. S., & Hartmann, L. C. (2010). Ki-67 as biomarker in atypia. Breast Cancer Res Treat, 121(2), 431–437. https://doi.org/10.1007/s10549-009-0584-x
- Gail, M. H., Brinton, L. A., Byar, D. P., et al. (1989). Individualized breast cancer risk projection. JNCI, 81(24), 1879–1886. https://doi.org/10.1093/jnci/81.24.1879
- Tyrer, J., Cuzick, J., & Sasieni, P. (2004). Breast cancer prediction model. Statistics in Medicine, 23(7), 1111–1130. https://doi.org/10.1002/sim.1668
- Lee, A., Mavaddat, N., Wilcox, A. N., et al. (2019). BOADICEA risk model. Genetics in Medicine, 21(8), 1708–1718. https://doi.org/10.1038/s41436-018-0406-9
- U.S. Preventive Services Task Force. (2019). Breast cancer: Medications for risk reduction. JAMA, 322(9), 857–867. https://doi.org/10.1001/jama.2019.11885
- Visvanathan, K., Fabian, C. J., Bantug, E., et al. (2019). Endocrine therapy for risk reduction: ASCO guideline. JCO, 37(33), 3152–3165. https://doi.org/10.1200/JCO.19.01472
- Tiwari, S., Dwivedi, S., & Sharma, A. (2011). Concept of granthi and arbuda in Ayurveda: Correlation with breast disease. AYU, 32(2), 242–246. https://doi.org/10.4103/0974-8520.92578
- Choudhary, A., Singh, R., & Sharma, V. (2015). Ayurvedic perspective of stana roga (breast disorders): A review. Journal of Ayurveda and Integrative Medicine, 6(2), 75–82. https://doi.org/10.4103/0975-9476.146547
- Aggarwal, B. B., & Harikumar, K. B. (2009). Curcumin’s therapeutic potential against breast cancer. Breast Cancer, 16(4), 273–280. https://doi.org/10.1007/s12282-009-0112-9
- Widodo, N., Takagi, Y., Shrestha, B. G., et al. (2010). Selective killing of cancer cells by Withania somnifera leaf extract. PLoS ONE, 5(10), e13536. https://doi.org/10.1371/journal.pone.0013536
- Singh, S., & Patel, D. K. (2020). Tinospora cordifolia (Guduchi): A reservoir for therapeutic applications. Indian Journal of Traditional Knowledge, 19(4), 812–824. http://nopr.niscair.res.in/handle/123456789/55611
- Jain, A., Chauhan, P., Meena, H., & Sharma, R. (2015). Abhrak Bhasma and its nanoformulations: Pharmacological profile. Ancient Science of Life, 34(3), 153–159. https://doi.org/10.4103/0257-7941.157149
- Singh, R., & Pandey, B. L. (2021). Swarna Bhasma in integrative cancer care: Evidence and directions. Journal of Ethnopharmacology, 277, 114265. https://doi.org/10.1016/j.jep.2021.114265