- The Global Cancer Burden in the USA, UK, Australia, Singapore, and Canada
- Understanding Where You Are in the Breast Cancer Journey
- How Structured Support Can Fit Alongside Oncology Care
- Treatment Delays and Their Clinical Significance in Advanced Health Systems
- Bridging Institutional Excellence With Structured Parallel Support
- Global Healthcare Reality/Waiting Period
- The Biological Significance of Treatment Delays
- System Gaps and the Experience of Structured but Fragmented Care
- Why Structured Parallel Support Becomes Relevant
- Biological Rationale
- Key Signs That Require Immediate Breast Evaluation
- Metabolic Dysfunction and Insulin Signaling in Cancer Biology
- When Patients Seek Additional Guidance
- Active Oncology Support-Pre Surgical Optimization in Breast Cancer Care
- Active Oncology Support- Chemotherapy Phase Support in Breast Cancer Care
- Active Oncology Support-Radiation Phase Support in Breast Cancer Care
- Common Mistakes Patients Make After a Breast Cancer Diagnosis
- Post Treatment Recurrence Prevention in Breast Cancer Care
- High Risk Prevention in Breast Cancer
- Ayurvedic Integration in High Risk Prevention
- Bridging Modern Oncology and Rasayana Science
- High Risk Prevention Model
- Structured Care Model
- Safety and Red Flags
- Lifestyle and Metabolic Correction
- Stress Stabilization and Psychological Support
- Integrative Biological Stabilization Theory
- FAQ
- Related Clinical Pathways in Breast Oncology
- How Breast Cancer Care Works in Different Countries
- Reference
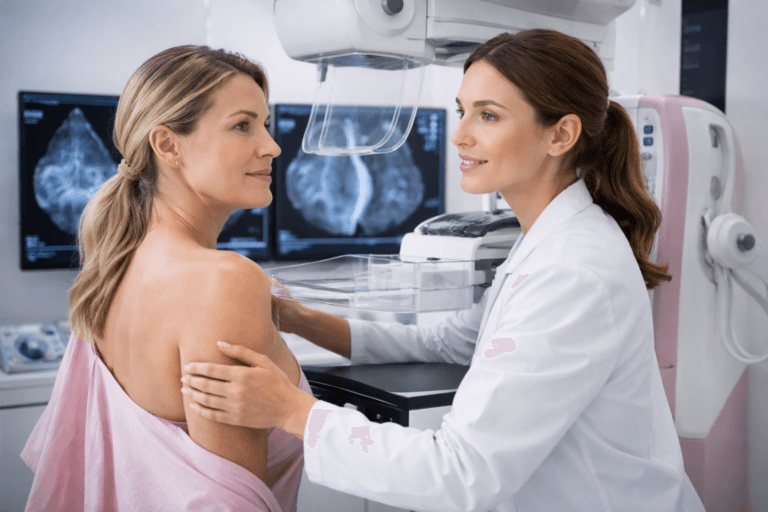
Breast cancer integrative support is designed to work alongside active treatment, including surgery, chemotherapy, and radiation, while focusing on long-term biological stabilization and recurrence risk reduction.
The Global Cancer Burden in the USA, UK, Australia, Singapore, and Canada
Cancer care in high income countries is advanced, yet the burden continues to grow. In the United States, data from national cancer surveillance systems show millions of new diagnoses each year. In the United Kingdom, the National Health Service manages an increasing oncology workload across screening, diagnosis, and treatment pathways. Australia, Singapore, and Canada report similar upward trends in cancer incidence through their national registries and public health agencies. These patterns are consistent with the global findings reported by Sung et al. [1], which highlight the scale of cancer incidence and mortality worldwide.
If you are a patient in the United States, you may interact with institutions such as the National Cancer Institute, major academic hospitals, or regional cancer centers. In the United Kingdom, your care may be coordinated through the NHS and regional cancer alliances. In Australia, Cancer Australia and state based health systems guide oncology services. In Singapore, the National Cancer Centre Singapore plays a central role. In Canada, provincial cancer agencies such as Cancer Care Ontario or BC Cancer oversee structured pathways.
These systems are highly developed. However, as incidence rises, demand also rises. When you enter the system, you may encounter waiting periods for imaging, biopsy confirmation, multidisciplinary review, or surgery scheduling. From a public health perspective, these systems are managing large populations. From your perspective, it feels personal and immediate.
The increasing cancer burden documented globally [1] reinforces one important point. Structured oncology support models must scale alongside national healthcare systems. Patients in advanced economies still experience uncertainty, time gaps, and fragmented explanations despite strong institutional frameworks.
Understanding Where You Are in the Breast Cancer Journey
When a woman receives an abnormal mammogram or biopsy result, the next steps often feel unclear. Some patients are told they are high risk. Others receive benign findings or atypical cells. Some are diagnosed with DCIS or invasive breast cancer. Each stage requires a different medical pathway.
To help you understand your situation clearly, the following clinical guides explain the three most common diagnostic pathways in breast oncology.
High Risk and Genetic Breast Cancer
Women with strong family history, BRCA mutations, or hereditary cancer syndromes require structured monitoring and prevention strategies. These patients may not have cancer yet but carry elevated lifetime risk.
Read the full guide here:
https://panaceayur.com/disease-cure/oncology/breast-cancer/high-risk-and-genetics/
Benign Breast Conditions and Atypia
Many biopsy reports identify benign tumors, fibroadenoma, or atypical hyperplasia. These findings are not invasive cancer but can indicate increased risk depending on pathology and family history.
Learn more here:
https://panaceayur.com/disease-cure/oncology/breast-cancer/benign-and-atypia/
DCIS and Invasive Breast Cancer
Ductal carcinoma in situ and invasive breast cancer require structured oncology treatment under hospital supervision. Surgery, radiation, chemotherapy, or hormonal therapy may be recommended depending on stage and biology.
Read the clinical explanation here:
https://panaceayur.com/disease-cure/oncology/breast-cancer/dcis-and-invasive-cancer/
Why Understanding Your Category Matters
The most common mistake patients make is assuming every breast abnormality represents the same disease. In reality, breast pathology exists on a spectrum ranging from benign conditions to high risk lesions and invasive cancer.
Correct classification determines surveillance strategy, treatment intensity, and long term prevention planning.
When Patients Begin Looking for Additional Support
Many patients begin searching for lifestyle, metabolic, or integrative support during specific stages of their journey.
Some are waiting for surgery or chemotherapy scheduling. Others are in the survivorship phase after treatment. Some are identified as high risk and wish to focus on prevention strategies.
Understanding your diagnostic category first helps ensure that any supportive approach is coordinated safely alongside oncology care.
How Structured Support Can Fit Alongside Oncology Care
Modern oncology guidelines emphasize that any supportive program must work in parallel with hospital treatment rather than replace it. Surgery, chemotherapy, radiation, endocrine therapy, and imaging remain the central pillars of cancer management.
Supportive programs may focus on metabolic stability, inflammation control, lifestyle correction, and psychological resilience during the cancer journey.
This approach aligns with international oncology guidance that integrative care should complement standard treatment while maintaining physician supervision.
Treatment Delays and Their Clinical Significance in Advanced Health Systems
Patients in countries such as the United States, United Kingdom, Australia, Singapore, and Canada often assume that modern healthcare infrastructure eliminates risk from delays. However, evidence suggests that timing still matters. Hanna et al. [2] demonstrated that delays in surgery or systemic cancer treatment are associated with increased mortality risk across multiple cancer types.
This does not imply that health systems are negligent. It highlights that oncology is biologically time sensitive. Even within structured systems like the NHS in the UK or leading cancer centers in the US and Canada, scheduling logistics, diagnostic pathways, and multidisciplinary reviews can introduce unavoidable intervals.
If you are waiting for breast cancer surgery in the US, navigating referral timelines in the UK NHS, scheduling through an Australian public hospital, or coordinating care through a Canadian provincial program, you may experience weeks between diagnosis and definitive treatment. During this time, tumor biology continues to evolve. Inflammation, hormonal signaling, and immune modulation do not pause simply because a surgical date is pending.
The findings of Hanna et al. [2] reinforce the clinical reality that time is not neutral in oncology. This supports the concept of structured parallel stabilization during waiting phases, not as a substitute for hospital care, but as an organized supportive framework.
Bridging Institutional Excellence With Structured Parallel Support
Healthcare systems in the USA, UK, Australia, Singapore, and Canada are among the most advanced globally. Institutions such as the National Cancer Institute, NHS cancer pathways, Cancer Australia initiatives, National Cancer Centre Singapore, and Canadian provincial cancer agencies operate within evidence based frameworks.
Yet even within these systems, patients frequently report:
You may feel that consultation time is limited.
You may struggle to fully understand your pathology report.
You may feel uncertain about what can be done between diagnosis and treatment.
From the institutional side, rising case volumes and resource allocation challenges create pressure. From your side, uncertainty creates anxiety.
The global data from Sung et al. [1] demonstrate the magnitude of cancer incidence. The mortality association with treatment delay reported by Hanna et al. [2] underscores the importance of time sensitive care. Together, these data support a rational conclusion. Structured oncology support should evolve in parallel with established hospital systems.
You do not replace your oncologist. You do not interrupt evidence based treatment. Instead, you create an organized biological stabilization and prevention strategy that operates alongside national healthcare pathways.
In high income countries with strong public and private oncology networks, the future of care is not competition between systems. It is integration of structured supportive models that respect existing institutions while addressing the biological and psychological needs that patients experience throughout their journey.
Global Healthcare Reality/Waiting Period

Some biopsy findings such as atypical hyperplasia or other benign abnormalities may not represent invasive cancer but can still carry elevated risk. Understanding these conditions is important before making long term prevention decisions. You can read more in Benign Breast Conditions and Atypia.
Link https://panaceayur.com/disease-cure/oncology/breast-cancer/benign-and-atypia/
Cancer care systems in the United States, United Kingdom, Australia, Singapore, and Canada are among the most structured and evidence based in the world. Institutions follow established pathways such as the National Comprehensive Cancer Network guidelines in the United States [13], NHS cancer referral standards in the United Kingdom, Cancer Australia frameworks, National Cancer Centre Singapore protocols, and provincial cancer agency pathways in Canada.
These systems are designed to ensure that once a diagnosis is confirmed, patients move through clearly defined stages of imaging, biopsy confirmation, multidisciplinary review, surgical planning, and systemic therapy when indicated. From a policy perspective, the pathways are organized and evidence aligned. From your perspective as a patient, however, the lived experience can feel very different.
You may receive a diagnosis and then be told that surgery is scheduled several weeks later. You may wait for a multidisciplinary tumor board review. You may be placed on a waiting list for reconstruction planning. Even in advanced systems, coordination across radiology, surgery, oncology, and pathology can introduce unavoidable intervals.
This is not a failure of care. It is the reality of high demand within highly structured systems.
The Biological Significance of Treatment Delays
While healthcare systems focus on logistical sequencing and resource allocation, tumor biology continues to evolve. The question is not whether guidelines exist. The question is whether time matters biologically.
A large systematic review and meta analysis by Hanna et al. [2] demonstrated that delays in surgery, systemic therapy, and radiotherapy across several cancer types were associated with increased mortality risk. The analysis showed that even incremental postponements in definitive treatment could influence long term survival outcomes.
For you as a patient, this information can be unsettling. It does not mean that every delay leads to progression. It means that oncology is time sensitive. Tumor microenvironment dynamics, inflammatory signaling, immune interaction, and hormonal influence do not pause while administrative processes unfold.
In the United States, where insurance authorization and scheduling coordination can add time, this interval may feel bureaucratic. In the United Kingdom, NHS referral pathways and surgical backlogs may extend waiting periods. In Australia, differences between public and private systems can affect scheduling timelines. In Singapore and Canada, centralized oncology services must manage high patient volumes within structured national frameworks.
Across all these countries, the clinical reality remains the same. Time in oncology carries biological weight [2].
System Gaps and the Experience of Structured but Fragmented Care
NCCN guidelines [13] outline clear standards for staging, surgical intervention, systemic therapy, and follow up. These pathways are essential and evidence based. However, guidelines focus primarily on treatment sequencing. They do not fully address what happens biologically and psychologically during waiting periods.
You may receive excellent technical care while still feeling:
Uncertain about your internal biological state
Concerned about inflammation or immune function
Unsure how to optimize nutrition and metabolic health
Anxious about recurrence even before treatment begins
From a systems perspective, oncology care is protocol driven. From your perspective, there may be a gap between diagnosis and active biological stabilization.
This gap is not a criticism of institutions such as the National Cancer Institute aligned centers in the United States, the NHS in the United Kingdom, Cancer Australia, National Cancer Centre Singapore, or Canadian provincial cancer agencies. It is an acknowledgment that structured treatment pathways do not always provide structured metabolic or integrative stabilization during the waiting phase.
Why Structured Parallel Support Becomes Relevant
When global cancer incidence continues to rise and healthcare systems operate at high capacity, waiting intervals become an inevitable part of modern oncology. The data from Hanna et al. [2] underscore that timing matters. The existence of formal oncology pathways such as NCCN [13] confirms that treatment must remain evidence based and guideline driven.
Within this context, structured integrative support is not positioned as an alternative to hospital care. It is positioned as a parallel stabilization strategy during biologically meaningful intervals.
You continue your scheduled surgery, chemotherapy, or radiation exactly as advised by your oncology team. At the same time, structured support can focus on inflammation regulation, metabolic optimization, immune resilience, and stress stabilization. This approach respects national guidelines while acknowledging the biological significance of time.
In advanced healthcare systems across the United States, United Kingdom, Australia, Singapore, and Canada, the goal is not to replace established oncology pathways. The goal is to complement them responsibly, particularly during waiting phases where biological processes continue even when definitive treatment has not yet begun.
Biological Rationale

Chronic Inflammation and the Tumor Microenvironment
When you receive a cancer diagnosis, most discussions focus on the tumor itself. Surgery targets it. Chemotherapy targets rapidly dividing cells. Radiation targets localized tissue. However, modern oncology increasingly recognizes that cancer does not exist in isolation. It develops and progresses within a broader biological environment.
One of the most important components of that environment is chronic inflammation.
Inflammation as a Prognostic Factor
Systemic inflammation is not simply a symptom of disease. It can influence disease behavior. Roxburgh and McMillan [3] demonstrated that markers of systemic inflammatory response are associated with prognosis across multiple cancer types. Elevated inflammatory indicators correlate with poorer survival outcomes, suggesting that the internal inflammatory state of the body plays a meaningful role in cancer progression.
If you are undergoing treatment in the United States, the United Kingdom, Australia, Singapore, or Canada, your oncologist may monitor laboratory markers related to inflammation as part of broader assessment. While standard oncology pathways focus on tumor staging and histopathology, the systemic inflammatory response reflects how the entire body is interacting with the disease process.
From a biological standpoint, chronic low grade inflammation can promote:
Increased cellular proliferation
Angiogenesis or new blood vessel formation
Genomic instability
Altered immune response
These processes contribute to a microenvironment that may support tumor survival and growth.
Inflammation as a Hallmark of Cancer Biology
The foundational oncology framework described by Hanahan and Weinberg [8] identified tumor promoting inflammation as one of the hallmarks of cancer. This landmark work reshaped how clinicians and researchers understand cancer. Instead of viewing tumors as isolated masses, the field now recognizes the importance of the surrounding microenvironment, including immune cells, inflammatory mediators, and stromal components.
For you as a patient, this means that what happens in the broader biological environment matters. Inflammation is not just a background process. It is intertwined with tumor behavior, immune surveillance, and tissue response.
Across advanced healthcare systems, whether you are treated in an NCI affiliated center in the United States, within the NHS in the United Kingdom, under Cancer Australia frameworks, through the National Cancer Centre Singapore, or within Canadian provincial cancer programs, standard treatment protocols remain essential and evidence based. At the same time, the scientific understanding reflected in the hallmarks of cancer model [8] highlights why systemic factors such as inflammation deserve attention.
Why This Matters During Active Treatment and Waiting Phases
Inflammatory pathways do not pause while you are waiting for surgery or between chemotherapy cycles. They continue to influence the tumor microenvironment and overall physiological resilience. The association between systemic inflammation and prognosis [3] reinforces the importance of supporting a balanced internal environment during all phases of the oncology journey.
This does not replace surgery, chemotherapy, or radiation. Those interventions directly target malignant cells according to established guidelines. Instead, the recognition of inflammation as a core biological driver provides a rational basis for parallel strategies aimed at metabolic stabilization, immune modulation, and systemic support.
When we speak about structured integrative oncology support in high income countries, we are not challenging standard care pathways. We are acknowledging what modern oncology already recognizes. Cancer is influenced by the surrounding biological terrain, and chronic inflammation is a measurable and clinically relevant component of that terrain [3], [8].
Understanding this framework allows you to view treatment not only as tumor removal or suppression, but also as an opportunity to stabilize the internal environment that interacts with the disease.
Key Signs That Require Immediate Breast Evaluation
Breast changes are common and many are benign. However, certain symptoms should always be evaluated promptly by a qualified healthcare professional. Early medical assessment helps determine whether changes are harmless or require further diagnostic investigation such as imaging or biopsy.
A New Lump in the Breast or Underarm
One of the most recognized warning signs is the appearance of a new lump in the breast or underarm region. Some lumps are benign conditions such as cysts or fibroadenomas, but any new mass should be evaluated through appropriate clinical examination and imaging.
Persistent Breast Pain in a Specific Area
Breast pain alone is often related to hormonal changes and is usually not associated with cancer. However, persistent localized pain that does not resolve over time should be assessed, particularly if accompanied by other changes in breast tissue.
Changes in Breast Shape or Size
Noticeable changes in breast contour, asymmetry, or swelling may indicate underlying structural changes. Although these changes can occur due to benign causes, they should be evaluated to rule out serious conditions.
Skin Changes on the Breast
Redness, thickening of the skin, dimpling, or a texture resembling orange peel can sometimes indicate deeper tissue changes. These signs warrant prompt clinical assessment.
Nipple Changes or Discharge
Inversion of the nipple, scaling of the skin around the nipple, or unexpected nipple discharge should be evaluated by a healthcare professional. These symptoms may require imaging or further diagnostic tests.
Unexplained Swelling or Enlarged Lymph Nodes
Swelling in the underarm or around the collarbone area may indicate lymph node involvement and should be examined by a physician.
Why Early Evaluation Matters
Many breast abnormalities are benign and can be managed with monitoring or simple treatment. However, early evaluation ensures that serious conditions are identified as quickly as possible.
Modern breast cancer screening programs emphasize early detection because treatment outcomes are generally better when cancer is identified at earlier stages.
What to Do If You Notice These Symptoms
If you experience any persistent breast changes, scheduling an evaluation with a qualified healthcare provider is the safest course of action. Clinical examination, mammography, ultrasound, or other imaging studies may be recommended depending on your age, medical history, and symptoms.
Prompt evaluation provides clarity and allows appropriate care decisions to be made based on accurate diagnosis.
Metabolic Dysfunction and Insulin Signaling in Cancer Biology

Cancer is not only a genetic condition. It is also deeply influenced by metabolic signaling. When we look at oncology through a modern scientific lens, we see that hormones such as insulin, adipokines, and growth factors interact with tumor cells in measurable ways.
If you are undergoing treatment in the United States, the United Kingdom, Australia, Singapore, or Canada, your oncologist may focus primarily on staging, receptor status, and histopathology. However, the metabolic environment in which the tumor exists is also biologically relevant.
Obesity and the Tumor Microenvironment
Research by Iyengar et al. [4] demonstrated that obesity alters the tumor microenvironment. Adipose tissue is not simply fat storage. It is metabolically active tissue that produces inflammatory mediators, estrogen in postmenopausal women, and growth promoting signals. These factors can influence cellular proliferation and immune behavior within the tumor environment.
If you are overweight or insulin resistant, the surrounding tissue environment may produce cytokines and inflammatory markers that create a more permissive setting for tumor progression. This does not mean weight alone causes cancer. It means that metabolic status can influence how cancer behaves.
Across advanced healthcare systems, including National Cancer Institute affiliated centers in the United States, NHS oncology services in the United Kingdom, Cancer Australia aligned pathways, National Cancer Centre Singapore, and Canadian provincial cancer programs, metabolic health is increasingly recognized as part of survivorship and prevention strategy. The scientific evidence supports this broader view [4].
Insulin Signaling and Tumor Growth
Insulin is a powerful anabolic hormone. When chronically elevated due to insulin resistance, it can activate pathways that stimulate cellular growth. Pollak [5] described how insulin and insulin like growth factors interact with neoplastic cells, potentially promoting proliferation and inhibiting apoptosis.
If your fasting insulin levels are persistently elevated, or if you have metabolic syndrome, those signaling pathways may remain active even during cancer treatment. This does not replace the importance of surgery or chemotherapy. It highlights that systemic metabolic signaling influences cellular behavior beyond the tumor itself.
You may not feel insulin resistance directly. It often presents silently. Yet biologically, it alters intracellular pathways linked to growth and survival.
The Clinical Position on Obesity in Oncology
The American Society of Clinical Oncology has formally addressed the relationship between obesity and cancer outcomes. The ASCO position statement [6] emphasizes that obesity is associated with increased risk of several cancers and can influence treatment outcomes and survivorship.
For you as a patient, this means metabolic health is not a cosmetic issue. It is a clinical variable. In countries such as the United States, Canada, Australia, and the United Kingdom, obesity prevalence is high, making metabolic correction an important component of comprehensive cancer care. Singapore, while having lower obesity rates overall, still recognizes metabolic syndrome as a public health concern.
Oncology guidelines focus on tumor directed therapy. Position statements such as ASCO’s [6] acknowledge that addressing metabolic dysfunction is part of responsible cancer care.
Why Metabolic Stabilization Matters During Waiting and Treatment Phases
Metabolic pathways continue to function regardless of treatment scheduling. If you are waiting for surgery or between chemotherapy cycles, insulin signaling and inflammatory mediators remain active. If you are in remission, these pathways may still influence recurrence risk.
Structured integrative support does not replace oncology protocols. Instead, it recognizes what modern research already confirms. Obesity related inflammation [4], insulin driven growth signaling [5], and clinical oncology consensus on metabolic health [6] together provide a scientific basis for metabolic stabilization strategies.
In advanced healthcare systems across the United States, United Kingdom, Australia, Singapore, and Canada, integrating metabolic awareness into cancer care is not oppositional. It is aligned with emerging oncology understanding.
When you view cancer through this lens, you begin to see that biological terrain matters. Insulin signaling and metabolic dysfunction are not peripheral issues. They are part of the broader oncologic environment that influences disease behavior and long term outcomes.
Hormonal Regulation in Breast Cancer Biology

Breast cancer is not only a cellular disorder. In many patients, it is also hormonally influenced. When you review your pathology report, you may see terms such as estrogen receptor positive or progesterone receptor positive. These labels are not minor technical details. They indicate that the tumor responds to hormonal signaling within your body.
Hormonal regulation therefore becomes a central biological consideration during diagnosis, treatment, and long term prevention.
Estrogen Signaling and Tumor Behavior
Estrogen plays a physiological role in breast tissue development and maintenance. However, in hormone receptor positive breast cancer, estrogen can stimulate cellular proliferation. Chlebowski et al. [7] examined the influence of estrogen exposure on breast cancer risk and outcomes, reinforcing the role of hormonal environment in disease behavior.
If you are diagnosed with estrogen receptor positive breast cancer in the United States, the United Kingdom, Australia, Singapore, or Canada, your oncology team may recommend endocrine therapies such as aromatase inhibitors or selective estrogen receptor modulators. These treatments are designed to reduce estrogen stimulation of tumor cells.
From a biological standpoint, this reflects an important principle. Hormonal signaling pathways are active drivers of tumor growth in certain subtypes of breast cancer.
The Systemic Hormonal Environment
Hormonal influence is not limited to tumor receptors alone. Estrogen levels are shaped by multiple systemic factors including:
Adipose tissue activity
Menopausal status
Insulin resistance
Inflammatory cytokines
Liver metabolism
Adipose tissue, particularly in postmenopausal women, can contribute to peripheral estrogen production. This is one reason why metabolic health and hormonal balance intersect. Research highlighted by Chlebowski et al. [7] underscores that cumulative estrogen exposure influences breast cancer risk and recurrence patterns.
If you are undergoing care within NHS oncology services, an NCI affiliated center in the United States, Cancer Australia programs, National Cancer Centre Singapore, or Canadian provincial cancer agencies, endocrine therapy will follow evidence based protocols. At the same time, your broader hormonal environment continues to interact with treatment response.
Hormonal Modulation During Active Treatment
Endocrine therapy is often prescribed for several years after initial treatment. This means hormonal regulation is not a short term issue. It is a long term biological consideration.
If you are taking aromatase inhibitors, tamoxifen, or other endocrine therapies, your hormonal signaling pathways are being medically modified. However, factors such as body weight, insulin levels, liver function, and inflammatory state still influence the systemic hormonal milieu.
Hormonal regulation during this phase is not about replacing prescribed medication. It is about understanding the broader biological context in which those medications operate.
Why Hormonal Stability Matters in Parallel Support
In hormone receptor positive breast cancer, estrogen driven proliferation is a known pathway [7]. Standard oncology addresses this through endocrine therapy. Parallel supportive strategies focus on stabilizing metabolic and inflammatory contributors that influence hormonal dynamics.
If you are in the waiting phase before surgery, starting endocrine therapy, or transitioning into survivorship, the hormonal environment remains biologically active. Recognizing this allows you to view breast cancer management as a comprehensive process that includes tumor directed therapy and systemic hormonal awareness.
In advanced healthcare systems across the United States, United Kingdom, Australia, Singapore, and Canada, endocrine therapy remains foundational for hormone receptor positive disease. At the same time, acknowledging the influence of estrogen signaling on tumor biology [7] provides a rational basis for structured hormonal stabilization within an integrative, parallel care model.
Hormonal regulation is not an alternative concept. It is embedded in modern oncology science. Understanding its role allows you to approach treatment with greater clarity about how internal biological signaling interacts with clinical interventions.
Immune Surveillance and the Tumor Microenvironment
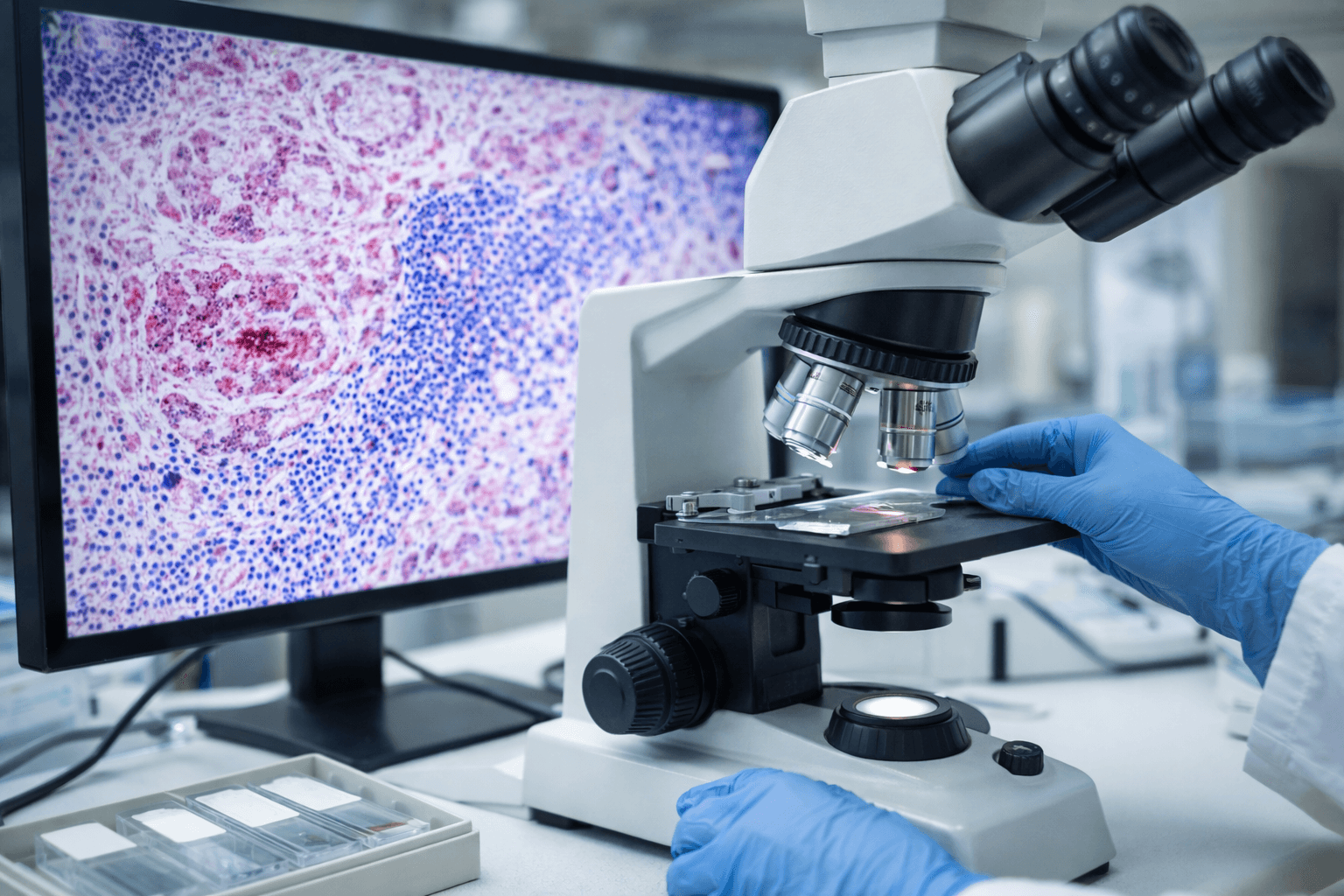
When you think about cancer, you may picture a tumor as an isolated mass. Modern oncology shows a more complex reality. Cancer exists within a dynamic biological environment known as the tumor microenvironment. This environment includes immune cells, blood vessels, connective tissue, inflammatory mediators, and signaling molecules.
Your immune system is not passive in this setting. It constantly monitors abnormal cells through a process known as immune surveillance. In early stages, immune cells may recognize and eliminate malignant cells. However, as cancer progresses, tumors can develop mechanisms to evade immune detection.
Immune Evasion in Modern Oncology
Hanahan and Weinberg [8] identified immune evasion as a fundamental hallmark of cancer. This framework changed how clinicians understand disease progression. Instead of viewing cancer purely as uncontrolled growth, it is now recognized that tumors actively adapt to avoid immune destruction.
Cancer cells can alter antigen presentation, recruit suppressive immune cells, and create a microenvironment that dampens cytotoxic responses. If you are receiving care in the United States, the United Kingdom, Australia, Singapore, or Canada, your oncology team may consider immunotherapy as part of treatment in specific subtypes. This reflects the growing recognition that restoring immune function is central to cancer control.
From your perspective as a patient, this means that immune strength and immune balance are biologically relevant. The immune system is not separate from the tumor. It interacts continuously with it.
The Tumor Microenvironment
The tumor microenvironment includes:
Immune cells such as T lymphocytes and macrophages
Fibroblasts and stromal tissue
Blood vessels supplying nutrients
Inflammatory mediators
This environment can either support immune attack or promote tumor survival. In certain contexts, chronic inflammation and metabolic dysfunction can shift the microenvironment toward a more permissive state.
If you are in a waiting phase before surgery or between chemotherapy cycles, this microenvironment remains active. It continues to influence tumor behavior even when direct treatment is not being administered.
Understanding this concept does not replace guideline directed oncology. Instead, it reinforces why systemic stabilization strategies are scientifically rational alongside standard care.
The Ayurvedic Perspective: Rasayana and Ojas
Long before the modern language of immune surveillance emerged, classical Ayurveda described a concept known as Ojas. In the Charaka Samhita [16], Ojas is described as the essence of all bodily tissues, responsible for vitality, resilience, and resistance to disease. When Ojas is strong, the body maintains equilibrium. When depleted, susceptibility increases.
Rasayana therapy in classical texts is designed to nourish tissues and enhance resilience. While the terminology differs from modern immunology, the conceptual parallel is notable. Immune competence and systemic vitality are central themes in both frameworks.
If you are undergoing oncology care within advanced healthcare systems, immunotherapy and targeted treatments are guided by evidence based protocols. At the same time, the recognition that immune surveillance influences cancer progression [8] aligns with the broader principle that systemic resilience matters.
Integrating Immune Understanding Into Parallel Support
You continue to follow your oncologist’s recommendations. Surgery, chemotherapy, radiation, and immunotherapy remain foundational. However, understanding immune evasion as a hallmark of cancer [8] and recognizing the classical emphasis on systemic vitality described in Charaka Samhita [16] provides a scientific and philosophical bridge.
Immune surveillance does not stop during waiting periods. The tumor microenvironment does not pause between treatment cycles. Supporting systemic balance during these phases is not oppositional to modern oncology. It reflects an integrative understanding that cancer exists within a living, responsive biological system.
When you approach breast cancer care through this lens, you begin to see that treatment is not only about eliminating malignant cells. It is also about strengthening the internal environment in which those cells exist.
Stress and Neuroendocrine Pathways in Breast Cancer Biology
When you receive a breast cancer diagnosis, the emotional impact is immediate. Uncertainty about surgery, chemotherapy, recurrence risk, and family implications activates powerful psychological stress responses. What many patients do not realize is that stress is not only emotional. It is also biological.
Your body responds to stress through the neuroendocrine system. This includes activation of the hypothalamic pituitary adrenal axis and the sympathetic nervous system. Cortisol, adrenaline, and other stress mediators are released into circulation. These hormones influence immune function, inflammation, angiogenesis, and cellular signaling.
Bio Behavioral Influence on Tumor Biology
Antoni et al. [9] examined how psychological stress and behavioral factors can influence tumor related biological processes. Their work demonstrated that chronic stress can alter immune function and may affect pathways related to tumor progression. Stress related neuroendocrine signaling can modulate cytokine production, angiogenic factors, and cellular proliferation dynamics.
If you are navigating care in the United States, the United Kingdom, Australia, Singapore, or Canada, your oncology team will focus primarily on tumor directed treatment. However, the scientific literature recognizes that psychological stress has measurable biological correlates.
This does not mean stress alone causes cancer. It means that persistent activation of stress pathways may influence the tumor microenvironment and immune surveillance processes.
The Neuroendocrine Immune Connection
When stress hormones remain elevated over prolonged periods, several changes can occur:
Altered immune cell distribution
Reduced cytotoxic T cell activity
Increased inflammatory signaling
Enhanced vascular support to tissues
From a physiological perspective, the body prioritizes survival responses. Unfortunately, chronic activation of these pathways can create internal conditions that are less favorable for immune mediated tumor control.
If you are waiting for surgery, receiving chemotherapy, or transitioning into survivorship, the psychological burden can be significant. The waiting phase in particular often intensifies uncertainty. During this time, neuroendocrine signaling remains active.
Understanding this biological link helps reframe stress management as more than emotional comfort. It becomes part of systemic stabilization.
Why Stress Regulation Matters in Parallel Care
Standard oncology care in advanced healthcare systems is evidence driven and protocol based. Surgery removes tumors. Chemotherapy targets malignant cells. Endocrine therapy blocks hormonal stimulation. Immunotherapy enhances immune response.
Parallel supportive strategies do not replace these interventions. Instead, they acknowledge that the neuroendocrine environment interacts with immune and inflammatory pathways.
If you are experiencing ongoing anxiety about recurrence or treatment outcomes, that emotional state has physiological correlates. Structured stress regulation approaches aim to stabilize cortisol patterns, improve sleep, and support immune balance.
In countries such as the United States, the United Kingdom, Australia, Singapore, and Canada, psycho oncology services are increasingly integrated into cancer centers. This reflects growing recognition that mental health and biological resilience are interconnected.
The findings of Antoni et al. [9] reinforce that bio behavioral factors influence tumor biology. Recognizing this connection allows you to view stress management not as an optional wellness activity, but as a biologically relevant component of comprehensive cancer care.
When you approach breast cancer through this lens, you begin to see that healing involves not only targeting malignant cells but also stabilizing the neuroendocrine environment in which those cells exist.
When Patients Seek Additional Guidance
Many patients begin searching for additional guidance during specific moments in the breast cancer journey. This often happens after an abnormal mammogram, during the waiting period before surgery, or after completing chemotherapy or radiation.
At these stages patients frequently look for ways to support their overall health, manage stress, improve metabolic stability, and reduce modifiable risk factors. Structured support programs can be considered alongside hospital care while diagnostic monitoring and treatment continue under oncology supervision.
Understanding the stage of disease and following the recommended clinical pathway remain essential before beginning any supportive strategy.
Situations Where Structured Support May Be Considered
Patients commonly explore additional support during several situations.
Some are waiting for surgery or treatment scheduling after diagnosis. Others have completed hospital treatment and are entering the survivorship phase where recurrence prevention becomes important. Certain individuals are identified as high risk because of genetic mutations, family history, or atypical biopsy findings.
In these circumstances patients may seek programs that focus on lifestyle stabilization, metabolic health, stress management, and long term resilience while continuing standard medical care.
When to Seek Medical Consultation
Breast cancer management should always remain under the supervision of qualified oncology professionals. Any supportive approach must be coordinated carefully with the medical team to ensure safety and compatibility with treatment.
Patients who wish to explore structured support should first ensure that their diagnostic reports, pathology findings, and treatment plans are clearly understood. Consultation with experienced clinicians allows supportive strategies to be aligned appropriately with the stage of disease and the ongoing oncology plan.
Active Oncology Support-Pre Surgical Optimization in Breast Cancer Care
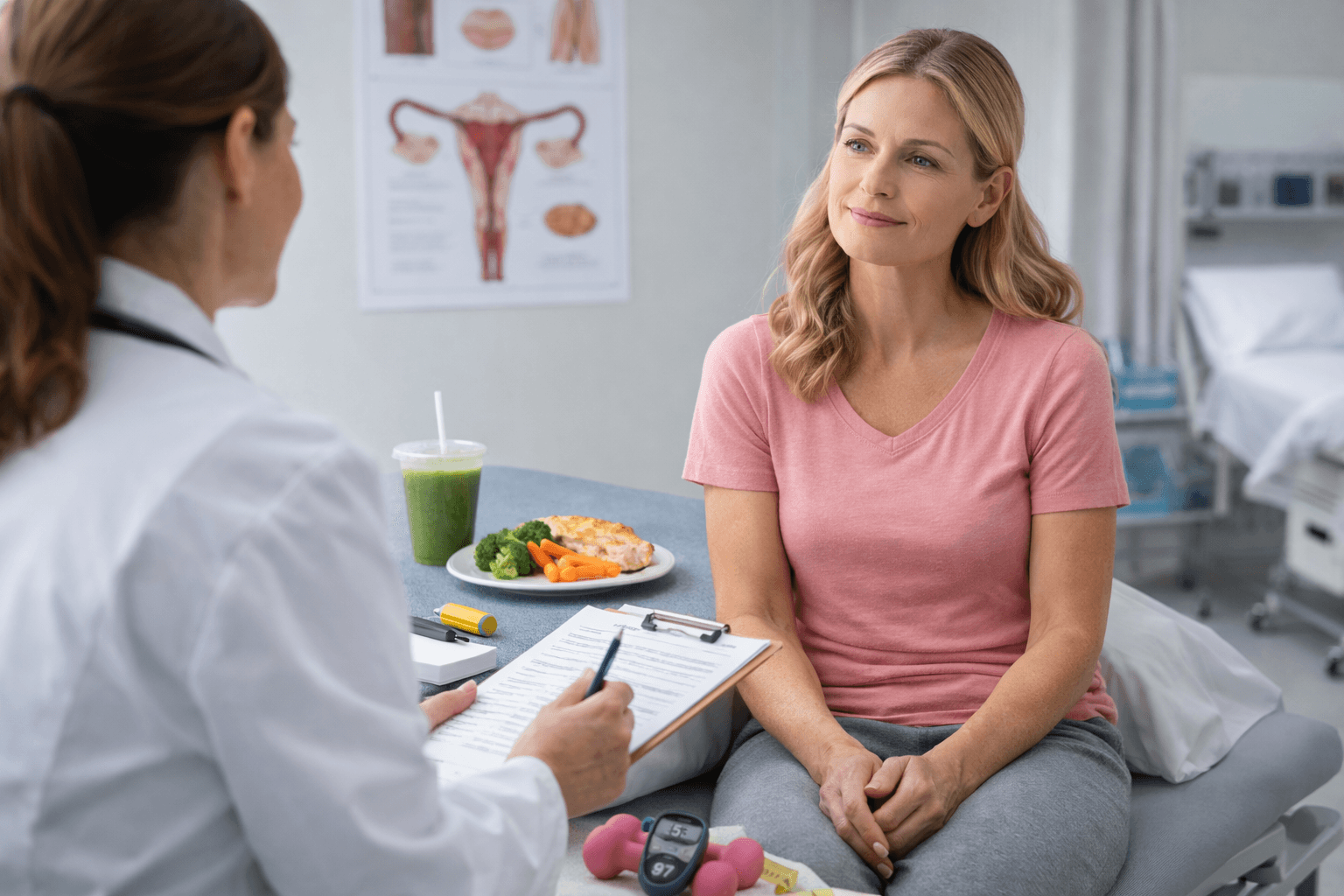
When you receive a surgical date for breast cancer, it can feel like a countdown. You may be waiting two weeks or several weeks depending on diagnostic review, operating room availability, or multidisciplinary planning. In advanced healthcare systems such as the United States, the United Kingdom, Australia, Singapore, and Canada, surgical pathways are structured and evidence based. However, the period before surgery is not biologically inactive.
This is where pre surgical optimization becomes clinically relevant.
Treatment pathways depend heavily on whether the diagnosis is ductal carcinoma in situ or invasive breast cancer. These two categories follow different oncology management strategies. A detailed explanation is provided in DCIS and Invasive Breast Cancer.
Link https://panaceayur.com/disease-cure/oncology/breast-cancer/dcis-and-invasive-cancer
Why Timing Matters Before Surgery
A comprehensive analysis by Hanna et al. [2] demonstrated that delays in cancer treatment, including surgery, are associated with increased mortality risk across several cancer types. The findings emphasize that timing is not merely administrative. It is biologically meaningful.
If your surgery is scheduled several weeks after diagnosis, this does not mean harm is inevitable. It means that tumor biology continues to operate during that interval. Inflammation, metabolic signaling, immune interaction, and stress pathways remain active.
You cannot change the operating room schedule in most cases. What you can influence is your internal biological environment during this waiting phase.
Pre surgical optimization does not replace surgery. It prepares your system for it.
Lifestyle and Systemic Stabilization Before Surgery
Rock et al. [11], representing major oncology nutrition and survivorship guidance, emphasize that lifestyle factors including nutrition, physical activity, and weight management influence cancer outcomes and overall resilience. While their guidance often focuses on survivorship, the principles apply across the treatment continuum.
If you are preparing for surgery, several areas deserve attention:
Nutritional adequacy to support wound healing
Stable blood glucose levels to reduce infection risk
Optimized protein intake for tissue repair
Moderate physical activity to maintain cardiovascular resilience
Adequate sleep to regulate immune and inflammatory balance
These are not alternative concepts. They are aligned with mainstream oncology survivorship and supportive care recommendations [11].
Reducing Surgical Complication Risk
In countries such as the United States and Canada, surgical teams often assess metabolic health before major procedures. In the United Kingdom and Australia, pre assessment clinics evaluate comorbidities before breast surgery. Singapore’s oncology centers similarly follow structured pre operative evaluation protocols.
Poor glycemic control, chronic inflammation, obesity, and high stress levels can influence surgical recovery. Optimizing these variables before surgery may support:
Improved wound healing
Reduced infection risk
Better tolerance to anesthesia
Faster functional recovery
The biological rationale is consistent with the understanding that treatment timing matters [2] and systemic health influences outcomes [11].
What Pre Surgical Optimization Is Not
Pre surgical optimization is not about delaying hospital treatment. It is not about replacing guideline directed surgery. It is not about promising outcome guarantees.
You continue to follow your oncology team’s recommendations. Surgery remains the cornerstone of treatment for many breast cancer cases.
Parallel support focuses on preparing your physiology to enter surgery in the most stable condition possible. It respects established oncology pathways in advanced healthcare systems while acknowledging that internal biological terrain influences recovery.
Viewing the Waiting Period as an Active Phase
If you are waiting for surgery, you may feel powerless. Pre surgical optimization reframes that waiting period as active preparation.
Hanna et al. [2] reinforce that time matters in oncology. Rock et al. [11] reinforce that lifestyle factors influence resilience and survivorship. Together, these findings support a structured, responsible approach to active stabilization before definitive surgical intervention.
You are not replacing your surgeon.
You are not interrupting hospital care.
You are strengthening your internal environment while the healthcare system prepares for the next step.
In advanced oncology systems across the United States, United Kingdom, Australia, Singapore, and Canada, this integrative preparation model complements surgical excellence rather than competing with it.
Active Oncology Support- Chemotherapy Phase Support in Breast Cancer Care

Chemotherapy is one of the most structured and protocol driven phases of breast cancer treatment. Whether you are receiving care in the United States, the United Kingdom, Australia, Singapore, or Canada, chemotherapy regimens are guided by standardized oncology pathways. Drug dosing, cycle intervals, toxicity monitoring, and response assessment are carefully regulated.
During this phase, your primary oncology team remains central. Chemotherapy drugs directly target rapidly dividing malignant cells. Parallel support does not replace these drugs. It focuses on maintaining systemic resilience while treatment proceeds.
Safe Integration During Chemotherapy
The American Society of Clinical Oncology integrative oncology guidelines [14] emphasize that supportive therapies may be used during cancer treatment when they do not interfere with chemotherapy mechanisms or safety. The key principle is coordination and transparency. Any parallel intervention must be disclosed to and aligned with your oncology team.
If you are undergoing chemotherapy, safety becomes the primary concern. Certain supplements, high dose antioxidants, or unregulated products may interact with cytotoxic drugs. Evidence based integrative oncology guidelines [14] stress the importance of avoiding therapies that could reduce treatment efficacy or increase toxicity risk.
This means structured chemotherapy phase support must be:
Medically supervised
Non interfering with cytotoxic pathways
Focused on symptom stabilization
Aligned with standard oncology timing
In advanced healthcare systems, multidisciplinary oncology teams increasingly recognize the value of supportive care when delivered responsibly.
Nutritional Stabilization During Chemotherapy
Rock et al. [11] provide evidence based nutrition guidance for cancer patients across treatment phases. During chemotherapy, nutritional goals shift toward maintaining adequate caloric intake, preserving lean body mass, and preventing treatment related malnutrition.
You may experience nausea, taste changes, fatigue, mucositis, or reduced appetite. These effects can compromise nutritional intake. Structured nutritional planning during chemotherapy focuses on:
Maintaining protein adequacy
Preventing unintended weight loss
Stabilizing blood glucose
Ensuring hydration
Reducing gastrointestinal distress
This approach is not alternative medicine. It is supportive oncology care grounded in mainstream guidance [11].
Supporting Treatment Tolerance
Chemotherapy places metabolic and inflammatory stress on the body. The immune system may be temporarily suppressed. Fatigue may increase. Emotional strain may intensify.
Responsible chemotherapy phase support aims to:
Preserve functional status
Maintain nutritional reserves
Support sleep quality
Stabilize stress responses
Encourage gentle physical activity when appropriate
ASCO integrative oncology guidance [14] highlights that symptom management strategies such as mindfulness, gentle exercise, and nutrition counseling may improve quality of life without interfering with medical treatment.
In countries such as the United States and Canada, comprehensive cancer centers often include integrative services. In the United Kingdom, psycho oncology and dietetic services are increasingly incorporated into NHS cancer programs. Australia and Singapore similarly integrate supportive care within oncology frameworks.
What Chemotherapy Phase Support Is Not
Chemotherapy phase support is not a substitute for cytotoxic treatment. It does not claim to replace chemotherapy. It does not promise outcome guarantees. It does not advise stopping hospital based care.
You continue your chemotherapy exactly as prescribed. Blood counts are monitored. Imaging follow up is performed. Dose adjustments occur when medically necessary.
Parallel support respects oncology protocols and focuses on maintaining systemic resilience during treatment cycles.
Viewing Chemotherapy as a Dual Phase
Chemotherapy targets malignant cells directly. At the same time, your body undergoes significant physiological stress. Evidence based nutrition guidance [11] and safe integrative oncology principles [14] provide a framework for structured support that aligns with hospital care.
In advanced healthcare systems across the United States, United Kingdom, Australia, Singapore, and Canada, the goal is not to oppose standard treatment. The goal is to enhance tolerance, maintain strength, and reduce treatment related burden while adhering strictly to established oncology protocols.
When approached responsibly, chemotherapy phase support becomes a parallel stabilization strategy rather than an alternative path.
Active Oncology Support-Radiation Phase Support in Breast Cancer Care
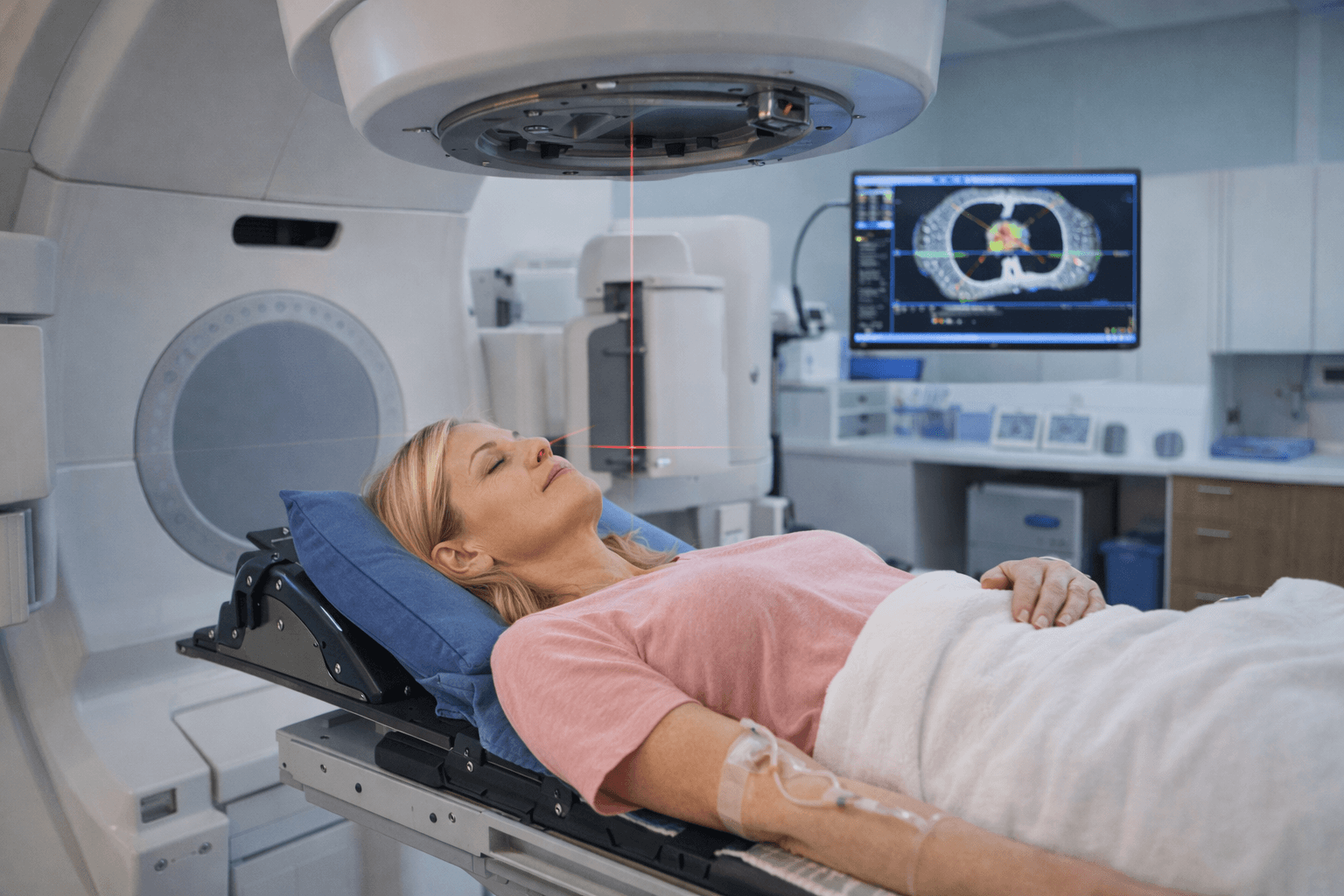
Radiation therapy is a highly targeted and technology driven phase of breast cancer treatment. Whether you are receiving care in the United States, the United Kingdom, Australia, Singapore, or Canada, radiation protocols are delivered within structured oncology pathways using precise imaging and dose planning systems.
Radiation aims to eliminate residual microscopic cancer cells after surgery or reduce recurrence risk in specific clinical scenarios. It is a local treatment. However, the body still responds systemically to radiation exposure.
Parallel support during radiation is not about altering the prescribed dose. It is about maintaining physiological stability while treatment proceeds according to oncology guidelines.
Safety First During Radiation Therapy
The ASCO integrative oncology guidelines [14] emphasize that any complementary support used during cancer treatment must be safe, evidence informed, and non interfering with conventional therapy.
During radiation, certain topical agents, supplements, or high dose antioxidants may theoretically affect treatment response. This is why integrative approaches must be carefully structured and coordinated with your oncology team.
If you are undergoing radiation, the primary rules are:
Do not apply unapproved topical substances to irradiated skin
Do not use high dose supplements without oncology approval
Maintain open communication with your radiation oncologist
Responsible support aligns with established treatment plans rather than competing with them [14].
Common Radiation Related Challenges
Radiation therapy may cause:
Skin irritation or dermatitis
Fatigue
Localized swelling
Breast tenderness
Sleep disturbance due to treatment scheduling
These effects vary depending on radiation field, dose, and individual physiology.
Supportive care during radiation focuses on maintaining hydration, skin integrity, nutritional adequacy, and stress stability. Oncology teams in advanced healthcare systems already provide guidance on gentle skin care and symptom monitoring.
Parallel support reinforces these principles while respecting hospital protocols.
Maintaining Tissue Resilience
Radiation affects targeted tissues through controlled DNA damage mechanisms. While this is necessary for tumor control, surrounding healthy tissue must recover efficiently.
Physiological resilience during radiation depends on:
Adequate protein intake
Stable micronutrient status
Balanced inflammatory response
Proper sleep and circadian rhythm
Stress regulation
ASCO integrative oncology guidance [14] supports the use of non invasive approaches such as mindfulness, gentle physical activity, and medically supervised supportive strategies to improve quality of life during treatment.
These approaches are particularly relevant in healthcare systems across the United States, United Kingdom, Australia, Singapore, and Canada, where patient centered oncology models increasingly incorporate supportive care frameworks.
What Radiation Phase Support Is Not
Radiation phase support does not reduce radiation dose.
It does not replace radiation therapy.
It does not advise discontinuation of hospital treatment.
It does not promise prevention of side effects.
You continue radiation exactly as prescribed by your oncology team.
Parallel support focuses on symptom stabilization and tissue recovery while treatment is delivered according to evidence based standards.
Viewing Radiation as a Coordinated Phase
Radiation therapy is precise and localized. Your systemic physiology, however, remains dynamic. Supporting hydration, nutrition, sleep, and emotional stability during this phase aligns with integrative oncology principles outlined in ASCO guidance [14].
In advanced oncology systems across the United States, United Kingdom, Australia, Singapore, and Canada, the goal is coordinated care. Radiation eliminates residual disease risk at the tissue level. Structured supportive care aims to maintain overall physiological stability during that process.
When managed responsibly, radiation phase support complements hospital treatment and reinforces resilience without interfering with oncologic efficacy.
Common Mistakes Patients Make After a Breast Cancer Diagnosis
Receiving an abnormal breast imaging result or a cancer diagnosis can be overwhelming. During this period, patients often search extensively for information and may encounter conflicting advice online. Understanding common mistakes can help patients make safer and more informed decisions during this critical time.
Assuming Every Breast Finding Means Invasive Cancer
Not all abnormal findings represent invasive breast cancer. Some patients receive biopsy results showing benign tumors, fibroadenoma, or atypical hyperplasia. These conditions may require monitoring or preventive strategies but are not the same as invasive disease. Proper classification of the pathology report is essential before making treatment decisions.
Delaying Medical Evaluation After Abnormal Imaging
Occasionally patients postpone follow-up tests because they feel well or believe symptoms will resolve. Breast cancer in its early stages may not cause pain or obvious symptoms. Prompt diagnostic evaluation following abnormal imaging or biopsy recommendations remains essential.
Relying on Unverified Online Treatment Claims
The internet contains a large volume of health information, but not all sources are reliable. Some websites promote treatments that claim to replace surgery, chemotherapy, or radiation therapy. Patients should be cautious about approaches that discourage hospital care or lack scientific evidence.
Ignoring Lifestyle and Metabolic Risk Factors
Cancer biology is influenced by many systemic factors including metabolic health, chronic inflammation, physical inactivity, and obesity. Lifestyle measures such as nutrition, physical activity, sleep regulation, and stress management may support overall health during treatment and survivorship.
Not Understanding the Difference Between DCIS and Invasive Cancer
Ductal carcinoma in situ and invasive breast cancer follow different treatment pathways. Misunderstanding these categories can create unnecessary fear or lead to incorrect expectations about treatment. Reviewing pathology results carefully with a medical professional helps clarify the appropriate care plan.
Attempting Multiple Therapies Without Medical Coordination
Some patients begin supplements, herbal preparations, or alternative therapies without informing their oncology team. Certain substances can interact with chemotherapy agents, hormone therapies, or surgical medications. Any supportive strategy should be coordinated with healthcare providers to ensure safety.
The Importance of Informed, Coordinated Care
Breast cancer management works best when patients understand their diagnosis, follow evidence-based treatment plans, and seek credible medical guidance. Hospital-based oncology care remains the foundation of treatment, while supportive strategies may be considered under appropriate supervision.
Clear information and structured medical guidance help patients move through diagnosis, treatment, and survivorship with greater confidence and safety.
Post Treatment Recurrence Prevention in Breast Cancer Care

Completing breast cancer treatment is a major milestone, but it is not the end of the journey. Many patients describe the post treatment phase as emotionally complex. During active treatment, you have a schedule and a medical team guiding each step. After treatment ends, follow up often becomes less frequent. Even in advanced healthcare systems across the United States, the United Kingdom, Australia, Singapore, and Canada, survivorship can feel like a quieter phase with a louder internal question. What can I do now to reduce recurrence risk.
Post treatment recurrence prevention is not about living in fear. It is about building a structured long term plan that supports biological stability, reduces modifiable risk drivers, and strengthens resilience.
Understanding Recurrence Dynamics
Breast cancer recurrence does not follow a single predictable timeline for every patient. Demicheli et al. [10] described recurrence dynamics, showing that recurrence risk can vary over time and may follow distinct patterns. This matters because it reframes recurrence prevention as a long term strategy rather than a short term effort.
If you are in surveillance follow up, you may feel anxious before scans or clinic visits. Understanding that recurrence patterns can be time dependent helps shift the focus toward building consistent long term stability instead of reacting only to checkups.
Inflammation as a Modifiable Risk Driver
The internal biological environment continues to matter after treatment ends. Chronic systemic inflammation is increasingly recognized as relevant to cancer outcomes. Roxburgh and McMillan [3] showed that systemic inflammatory response is associated with prognosis across cancers, reinforcing the clinical significance of inflammation as part of overall risk biology.
Inflammation is influenced by multiple variables including metabolic status, sleep quality, stress load, diet patterns, and physical activity. You may not feel chronic low grade inflammation as a clear symptom, yet it can remain active within the body.
Post treatment stabilization therefore includes addressing drivers that contribute to persistent inflammatory signaling. This is not about replacing oncology follow up. It is about supporting internal biology in a way that aligns with modern understanding of cancer terrain.
Lifestyle and Survivorship Guidance
Cancer survivorship guidance emphasizes that lifestyle factors play a role in long term outcomes. Rock et al. [11] provide evidence based nutrition and physical activity guidelines for cancer survivors. These recommendations reinforce that survivorship is not passive. It includes structured attention to weight management, dietary quality, activity, and overall health maintenance.
If you have completed surgery, chemotherapy, or radiation, your recovery phase is an opportunity to rebuild metabolic resilience. Nutrition becomes important not only for energy but also for maintaining stable glucose levels, supporting immune balance, and improving overall physiological recovery.
Your survivorship plan should not feel like vague advice such as eat healthy. It should feel structured and practical, especially for patients balancing modern life pressures in the United States, the United Kingdom, Australia, Singapore, and Canada.
Physical Activity and Cancer Outcomes
Physical activity is one of the most consistently supported lifestyle factors in cancer survivorship research. Ligibel et al. [12] reviewed evidence linking physical activity to improved cancer outcomes, including potential benefits related to recurrence and survival. The relationship is multifactorial. Exercise influences insulin sensitivity, inflammatory mediators, immune function, and body composition.
This does not mean you need intense workouts. It means you need a consistent, achievable activity plan suited to your treatment recovery stage and physical condition.
If you are still managing fatigue, neuropathy, or joint pain after treatment, activity planning must be individualized. The goal is long term consistency, not short term intensity.
A Structured Long Term Prevention Plan
Post treatment recurrence prevention becomes most effective when it is structured. A responsible plan typically includes:
Regular oncology follow up and imaging as advised
Nutrition guidance aligned with survivorship recommendations [11]
Physical activity planning aligned with evidence based outcomes [12]
Inflammation and metabolic stabilization strategies informed by prognostic research [3]
Stress and sleep stabilization to reduce neuroendocrine strain
This approach is not oppositional to oncology. It is supportive of survivorship.
If you are being followed in an NCI affiliated center in the United States, within NHS survivorship programs in the United Kingdom, under Cancer Australia aligned survivorship resources, through the National Cancer Centre Singapore, or within Canadian provincial cancer agency follow up systems, your hospital pathway remains essential.
Parallel prevention planning focuses on what patients often feel is missing. A structured roadmap for long term biological stabilization.
Why This Matters Emotionally and Clinically
After treatment, many patients experience recurrence anxiety. Every symptom can feel suspicious. Every scan can feel heavy. A structured plan does not eliminate uncertainty, but it reduces helplessness.
Demicheli et al. [10] provide a framework for understanding recurrence patterns over time. Rock et al. [11] and Ligibel et al. [12] provide evidence based survivorship guidance that transforms prevention into practical daily action. Roxburgh and McMillan [3] reinforce the significance of inflammation as a measurable biological factor connected to outcomes.
Together, these findings support a clinically responsible conclusion. Post treatment breast cancer care should include not only surveillance, but also structured long term risk reduction and resilience building.
If you are moving from treatment completion into survivorship, you deserve a plan that is as structured as your treatment was.
High Risk Prevention in Breast Cancer

High Risk Prevention
Patients with hereditary risk factors such as BRCA mutations or strong family history require specialized surveillance strategies. A detailed explanation of genetic risk pathways is explained in
High Risk and Genetic Breast Cancer.
Link https://panaceayur.com/disease-cure/oncology/breast-cancer/high-risk-and-genetics/
Hormonal and Metabolic Risk Modulation with Classical Ayurvedic Integration
High risk prevention applies to women with strong family history, atypical hyperplasia, dense breast tissue, prolonged hormonal exposure, obesity, metabolic syndrome, or prior breast cancer. In the United States, United Kingdom, Australia, Singapore, and Canada, high risk patients are monitored under structured screening frameworks. Screening detects. Prevention stabilizes. A structured high risk prevention model focuses on hormonal exposure, metabolic dysfunction, inflammation, and tissue resilience in parallel with hospital surveillance.
Hormonal Exposure and Breast Cancer Risk
Chlebowski et al. [7] demonstrated that cumulative estrogen exposure influences breast cancer incidence and recurrence risk. Prolonged hormonal stimulation, whether endogenous or therapy related, remains biologically significant. Adipose tissue contributes to peripheral estrogen production, particularly in postmenopausal women. Insulin resistance amplifies proliferative signaling pathways. Chronic inflammation interacts with hormone receptor activity. If you are categorized as high risk under NCCN in the United States, NHS risk stratification in the United Kingdom, Cancer Australia guidance, or Canadian provincial programs, hormonal exposure forms part of your risk profile. High risk prevention must therefore address hormonal stability alongside imaging surveillance.
Obesity, Metabolic Dysfunction, and the Tumor Microenvironment
Iyengar et al. [4] described how obesity alters the tumor microenvironment through inflammatory cytokines and metabolic signaling. Adipose tissue is metabolically active and contributes to estrogen synthesis and chronic inflammatory activity. The ASCO Obesity Position Statement [6] recognizes obesity as a modifiable oncology risk factor and supports weight management as part of prevention and survivorship care. If you carry central adiposity or metabolic syndrome, insulin mediated growth signaling and inflammatory activation may remain biologically active. High risk prevention therefore includes weight stabilization, insulin sensitivity improvement, and chronic inflammation reduction in alignment with modern oncology evidence [4], [6], [7].
Ayurvedic Integration in High Risk Prevention
Rasayana Concept in Sushruta Samhita
Reference [15] Sushruta Samhita Chikitsa Sthana Chapter 27 Rasayana Adhyaya
Shloka
“Rasayanam nama jaravyadhi nashanam ayuh prajna bala varna karanam.”
Translation
Rasayana therapy destroys degeneration and disease and promotes longevity, intellect, strength, and vitality.
In modern interpretation, Rasayana enhances systemic resilience. For high risk individuals, this corresponds to strengthening tissue stability and reducing internal vulnerability before pathology manifests. Rasayana is preventive terrain stabilization rather than symptomatic intervention.
Rasayana Formulations in Bhavaprakasha
Reference [17] Bhavaprakasha Madhyama Khanda Rasayana Prakarana
Shloka
“Yat jara vyadhi nashanam deha dhatu balapradam tat sarvam rasayanam proktam.”
Translation
That which prevents degeneration and strengthens body tissues is called Rasayana.
Bhavaprakasha describes formulations intended to nourish Dhatu, enhance vitality, and stabilize internal balance. In a high risk prevention context, Rasayana supports hormonal equilibrium, metabolic correction, and long term tissue nourishment.
Bridging Modern Oncology and Rasayana Science
Modern oncology identifies hormonal exposure risk [7], obesity driven microenvironment changes [4], and metabolic correction as clinically important [6]. Classical Ayurveda emphasizes Dhatu stability, Ojas preservation, Agni balance, and Rasayana therapy [15], [17]. These frameworks are conceptually complementary. Hospital based surveillance continues. Metabolic and hormonal stabilization is implemented. Rasayana based systemic strengthening supports long term resilience.
High Risk Prevention Model
High risk prevention is especially relevant for women with atypical hyperplasia, dense breast patterns with hormonal imbalance, strong family history, obesity related estrogen dominance, or post treatment recurrence risk. This model does not replace mammography, MRI, or oncology review. It strengthens internal biology while standard surveillance continues.
In the United States, United Kingdom, Australia, Singapore, and Canada, high risk patients are managed within structured screening systems. Integrative high risk prevention functions in parallel by focusing on hormonal modulation, metabolic stabilization, inflammation reduction, and tissue resilience supported by modern evidence [4], [6], [7] and classical Rasayana science [15], [17].
Structured Care Model

Parallel Positioning With Oncology Supervision
A structured care model must be clear, responsible, and aligned with established oncology standards. In the United States, the United Kingdom, Australia, Singapore, and Canada, cancer care follows regulated pathways that emphasize safety, timing, and multidisciplinary coordination. Any integrative model must function within this framework, not outside it.
Oncology Supervision as the Foundation
The NCCN Guidelines [13] represent one of the most widely referenced oncology care frameworks in the United States and influence clinical standards globally. These guidelines outline staging, surgery, systemic therapy, radiation, and follow up recommendations. They reinforce that cancer treatment must remain supervised by qualified oncology specialists.
Parallel integrative support does not replace these pathways. It operates under oncology supervision. Imaging, biopsy interpretation, surgical planning, chemotherapy cycles, endocrine therapy, and radiation scheduling remain directed by your oncology team.
If you are receiving care through NCI affiliated centers in the United States, NHS oncology pathways in the United Kingdom, Cancer Australia networks, National Cancer Centre Singapore, or Canadian provincial cancer agencies, your hospital protocol remains primary.
The structured model acknowledges that oncology supervision is non negotiable [13].
Safe Parallel Integrative Use
The ASCO Integrative Oncology Guidelines [14] emphasize that complementary approaches may be used alongside standard cancer treatment when safety, transparency, and coordination are maintained. These guidelines stress that integrative support must not interfere with tumor directed therapies.
This creates a clear operational boundary. Integrative strategies must be:
Disclosed to the oncology team
Non interfering with chemotherapy or radiation
Evidence informed
Focused on symptom management and physiological resilience
Parallel positioning means the patient continues hospital based treatment exactly as prescribed while supportive measures aim to stabilize inflammation, metabolic balance, immune function, and stress regulation.
This is not alternative positioning. It is coordinated positioning consistent with ASCO principles [14].
Ayurvedic Theoretical Bridge Through Rasayana
Classical Ayurveda described systemic strengthening through Rasayana science. In the Charaka Samhita [16], Rasayana is associated with preservation of Ojas and enhancement of resistance to disease.
Shloka reference
Charaka Samhita Chikitsa Sthana Rasayana Adhyaya
“Rasayanani ayuh balam varnam smritim medham prajnam ca vardhayanti.”
Translation
Rasayana therapies promote longevity, strength, complexion, memory, intellect, and resilience.
In modern language, this concept parallels immune strengthening and systemic stabilization. It does not describe replacing surgery or chemotherapy. It describes enhancing internal resilience.
When placed within a structured care model, Rasayana functions as terrain support while oncologic supervision continues under established guidelines [13], [14].
Defining the Parallel Structure
The structured parallel model operates on three levels.
First
Hospital directed oncology care according to NCCN or national equivalent guidelines [13].
Second
Safe integrative support aligned with ASCO principles [14], focusing on symptom stabilization and quality of life.
Third
Long term systemic strengthening informed by Rasayana concepts described in Charaka Samhita [16], aimed at maintaining immune and metabolic resilience.
Each level has a defined boundary. None replaces the other.
Compliance and Safety Emphasis
Parallel positioning is essential for Western compliance. Patients must clearly understand:
Hospital care continues without interruption
No oncology treatment is discontinued
Integrative strategies are disclosed and coordinated
Emergency situations require immediate hospital evaluation
In healthcare systems across the United States, United Kingdom, Australia, Singapore, and Canada, legal and ethical standards require transparency. The structured care model reinforces these principles.
Safety and Red Flags
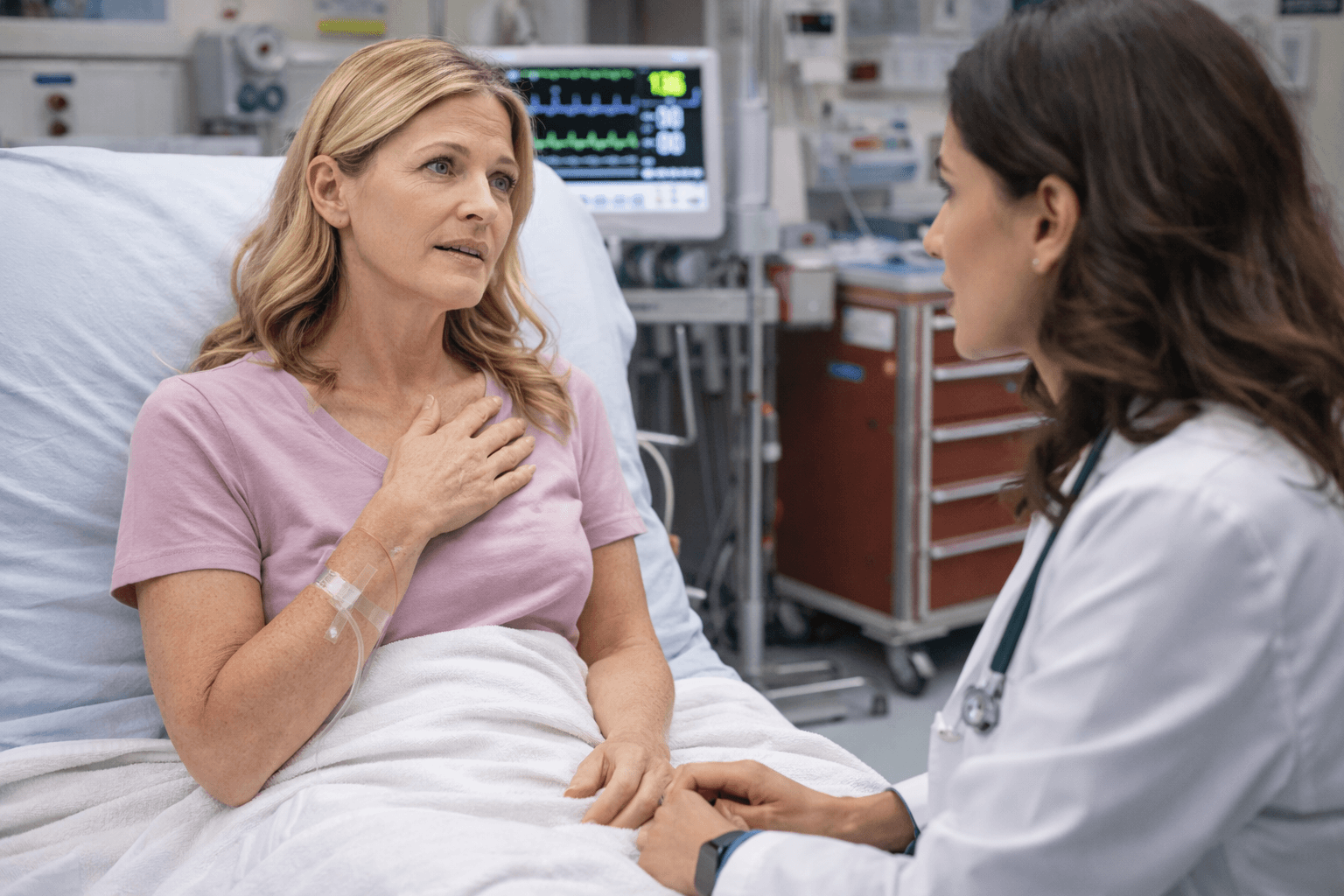
Clear Medical Boundaries in Parallel Care
A structured integrative model must clearly define what it is and what it is not. In the United States, the United Kingdom, Australia, Singapore, and Canada, oncology care follows regulated emergency protocols. Any supportive strategy must reinforce, not weaken, those safety standards.
The NCCN Guidelines [13] outline urgent oncologic situations that require immediate hospital based evaluation. The ASCO Integrative Oncology Guidelines [14] reinforce that complementary approaches must never delay or replace essential cancer treatment.
Parallel care exists within these boundaries.
Situations Requiring Immediate Hospital Evaluation
Certain symptoms must always be treated as medical priorities. If you experience any of the following, you must seek urgent oncology or emergency department care:
Rapidly enlarging breast mass
Sudden severe chest pain or shortness of breath
High fever during chemotherapy
Uncontrolled vomiting or dehydration
Neurological symptoms such as confusion or sudden weakness
Severe uncontrolled pain
Signs of infection at surgical site
Bleeding that does not stop
These are not situations for dietary adjustments, herbal strategies, or teleconsultation. These are oncologic emergencies defined within standard care frameworks [13].
During Chemotherapy or Radiation
If you are receiving chemotherapy, neutropenic fever is a medical emergency. If you are undergoing radiation and develop severe skin breakdown or unexpected systemic symptoms, immediate evaluation is required.
ASCO integrative oncology guidance [14] clearly states that supportive therapies must never interfere with active cancer treatment or delay emergency assessment.
Parallel care must pause when emergency care is required.
What Integrative Care Does Not Replace
Structured integrative oncology support does not replace:
Biopsy
Surgery
Chemotherapy
Radiation therapy
Endocrine therapy
Immunotherapy
Emergency management
Hospital based oncology supervision remains primary at all times [13].
Communication and Transparency
Safe parallel positioning requires:
Full disclosure of all supportive therapies to your oncology team
Avoidance of unregulated or unknown supplements
No unsupervised dose changes in prescribed medication
Immediate reporting of new or worsening symptoms
In high income healthcare systems, documentation and coordination are essential. Transparency protects the patient and strengthens trust.
Responsible Parallel Model
The structured care model follows three safety principles.
First
Oncology supervision remains central under NCCN aligned standards [13].
Second
Integrative strategies are used only when safe and non interfering according to ASCO guidance [14].
Third
Emergency red flags override all supportive plans.
This positioning reinforces that integrative care functions alongside hospital based oncology, not instead of it.
Why This Section Is Critical
Safety language builds credibility. It demonstrates that the model respects established medical authority and understands clinical risk.
If you are a patient in the United States, the United Kingdom, Australia, Singapore, or Canada, you deserve clarity. You deserve to know when to seek urgent care. You deserve to understand that parallel support never compromises essential treatment.
A responsible integrative oncology model must always begin and end with safety.
Lifestyle and Metabolic Correction

Structured Diet and Activity Framework for Oncology Support
Lifestyle and metabolic correction form a central pillar of structured oncology support. In the United States, the United Kingdom, Australia, Singapore, and Canada, survivorship care increasingly includes nutrition, weight management, and physical activity as part of comprehensive cancer management. These strategies are not alternative approaches. They are clinically endorsed components of risk reduction and long term stability.
Rock et al. [11] provide evidence based nutrition and lifestyle recommendations for cancer survivors. The guidance emphasizes maintaining a healthy body weight, prioritizing plant forward dietary patterns, limiting processed foods, and supporting metabolic balance during and after treatment. These recommendations apply across major oncology systems and are consistent with survivorship frameworks in high income countries.
Ligibel et al. [12] demonstrate that physical activity is associated with improved cancer outcomes, including recurrence risk modulation and overall survival in certain populations. Exercise influences insulin sensitivity, inflammatory signaling, and body composition. Structured physical activity is therefore not merely supportive. It is biologically relevant.
The ASCO Obesity Position Statement [6] recognizes obesity as a modifiable risk factor in oncology and encourages weight management interventions integrated within cancer care. Obesity influences hormonal exposure, inflammatory pathways, and tumor microenvironment signaling. Metabolic correction becomes a medical objective rather than a cosmetic goal.
Dietary Structure for Metabolic Stability
A structured oncology aligned dietary framework focuses on:
Balanced macronutrient intake
High fiber plant based foods
Adequate protein during treatment
Reduction of ultra processed sugars
Support of liver and metabolic pathways
This approach aims to stabilize insulin signaling and reduce chronic inflammatory load in alignment with survivorship guidance [11] and obesity related oncology evidence [6].
If you are undergoing chemotherapy, radiation, endocrine therapy, or post surgical recovery, nutritional adequacy becomes essential for treatment tolerance and tissue repair. If you are in survivorship phase, long term metabolic stability becomes central to recurrence prevention.
Exercise and Functional Recovery
Exercise is not restricted to athletic intensity. Ligibel et al. [12] highlight that moderate regular activity can influence cancer related outcomes. Walking programs, resistance training, and structured movement plans can improve muscle mass, reduce treatment related fatigue, and improve metabolic parameters.
In clinical oncology settings across the United States, United Kingdom, Australia, Singapore, and Canada, supervised exercise oncology programs are increasingly integrated into survivorship clinics. This reflects growing recognition of metabolic modulation as a therapeutic strategy.
Weight Management and Hormonal Influence
Adipose tissue contributes to peripheral estrogen production and inflammatory signaling. Excess adiposity therefore has both hormonal and metabolic implications. The ASCO statement [6] encourages clinicians to address weight management as part of comprehensive cancer care.
Metabolic correction includes:
Body composition optimization
Insulin sensitivity improvement
Reduction of systemic inflammation
Hormonal exposure modulation
These targets align with modern oncology understanding and reinforce structured prevention during waiting, treatment, and survivorship phases.
Integrative Implementation Within Structured Care
Lifestyle and metabolic correction operate within oncology supervision. They do not replace tumor directed therapy. They complement it.
Hospital guided treatment continues. Imaging and oncology review remain central. Diet and exercise strategies are layered on top to improve internal resilience and metabolic control.
This structured approach respects NCCN and ASCO aligned care pathways while implementing survivorship recommendations [11], exercise evidence [12], and obesity related oncology guidance [6].
Lifestyle correction becomes an active intervention rather than passive advice. It transforms waiting periods and survivorship phases into structured biological optimization periods under medical oversight.
Stress Stabilization and Psychological Support

Neuroendocrine Balance in Structured Oncology Care
Stress is not only emotional. It is biological. During cancer diagnosis, treatment, waiting periods, and survivorship, stress activates measurable neuroendocrine pathways. In modern oncology, this is no longer considered purely psychological. It is physiologically relevant.
Antoni et al. [9] demonstrated that chronic stress can influence tumor biology through neuroendocrine mechanisms. Stress hormones such as cortisol and catecholamines may affect inflammatory signaling, immune modulation, and tumor microenvironment dynamics. This does not mean stress causes cancer in isolation. It means sustained stress may influence biological processes relevant to cancer progression.
In healthcare systems across the United States, United Kingdom, Australia, Singapore, and Canada, psycho oncology services are increasingly integrated within cancer centers. Stress stabilization is recognized as part of comprehensive care.
Biological Impact of Chronic Stress
Chronic stress may contribute to:
Elevated cortisol patterns
Sympathetic nervous system activation
Immune surveillance disruption
Sleep disturbance
Inflammatory amplification
When a patient is in a waiting phase before surgery, undergoing chemotherapy, or transitioning into survivorship, prolonged uncertainty can activate persistent stress responses.
Antoni et al. [9] provide evidence that bio behavioral interventions can influence physiological markers associated with tumor biology. This supports structured psychological care as medically relevant, not optional.
Structured Psychological Support Model
Stress stabilization in oncology should include:
Guided counseling or psycho oncology consultation
Sleep regulation planning
Breathing and relaxation training
Mind body techniques under supervision
Social support reinforcement
This is not abstract advice. It is structured neuroendocrine stabilization.
In advanced oncology systems, multidisciplinary care increasingly incorporates mental health professionals as part of cancer care teams.
Ayurvedic Bridge Through Ojas Concept
Classical Ayurveda described systemic resilience through the concept of Ojas. In the Charaka Samhita [16], Ojas is described as the essence of all Dhatu and the foundation of vitality and resistance.
Shloka reference
Charaka Samhita Sutra Sthana
“Ojah sarva dhatu saram.”
Translation
Ojas is the essence of all body tissues.
Another reference in Rasayana context describes that preservation of Ojas strengthens resilience and longevity.
In modern interpretation, Ojas parallels the concept of integrated immune and neuroendocrine resilience. When stress depletes vitality, Ojas is considered weakened. When resilience is restored, Ojas is supported.
This framework does not replace psycho oncology. It complements it conceptually by emphasizing internal stability and systemic strength.
Integrating Stress Stabilization Within Structured Care
Stress stabilization operates within oncology supervision. It does not replace tumor directed therapy. It supports physiological balance during waiting periods, treatment cycles, and survivorship.
The model follows three principles.
First
Hospital based oncology treatment continues under established guidelines.
Second
Psychological support is structured and supervised.
Third
Resilience building strategies aim to stabilize neuroendocrine signaling and immune function.
Antoni et al. [9] provide the modern scientific foundation. Charaka Samhita [16] provides the classical theoretical bridge.
Why This Matters in Global Oncology Care
In the United States, United Kingdom, Australia, Singapore, and Canada, patients frequently report that emotional strain is one of the most difficult aspects of cancer care. Stress stabilization reduces psychological burden and may positively influence biological resilience.
Structured stress management is not an alternative therapy. It is an evidence informed supportive strategy integrated within comprehensive oncology care.
When implemented responsibly, stress stabilization strengthens the patient without compromising medical treatment, reinforcing both modern psycho oncology evidence [9] and classical resilience principles described in Charaka Samhita [16].
Integrative Biological Stabilization Theory
Bridging Modern Oncology and Rasayana Science
Cancer biology is not defined by a single mutation. It is defined by a pattern of biological behaviors. Modern oncology describes these behaviors through the framework of the hallmarks of cancer. Hanahan and Weinberg [8] outlined fundamental capabilities acquired during tumor development, including sustained proliferative signaling, evasion of growth suppressors, resistance to cell death, angiogenesis, immune evasion, and inflammation related progression.
This framework helps explain why cancer is dynamic rather than static. It also clarifies that stabilization must address systemic biological processes, not only tumor mass.
Integrative biological stabilization theory does not attempt to replace tumor directed treatment. It aims to support internal physiological balance while standard oncology therapy continues under supervision.
Hallmarks of Cancer and Systemic Context
Hanahan and Weinberg [8] emphasize that tumor progression interacts with the surrounding microenvironment. Inflammation, immune evasion, metabolic adaptation, and angiogenesis are not isolated events. They are interconnected processes.
From a systemic perspective, this means:
Chronic inflammation influences tumor behavior
Metabolic dysregulation alters signaling pathways
Immune surveillance may be suppressed
Stress pathways may modify microenvironment dynamics
Modern oncology targets specific pathways through surgery, chemotherapy, radiation, endocrine therapy, and immunotherapy. Integrative biological stabilization focuses on supporting host physiology during these interventions.
Rasayana in Sushruta Samhita
Reference [15] Sushruta Samhita Chikitsa Sthana Rasayana Adhyaya
Shloka
“Rasayanam nama jaravyadhi nashanam ayuh prajna bala varna karanam.”
Translation
Rasayana destroys degeneration and disease and promotes longevity, intellect, strength, and vitality.
In a modern interpretive framework, Rasayana represents systemic strengthening and tissue stabilization. While ancient texts did not describe oncogenes or tumor microenvironment, they emphasized preserving vitality and resisting degeneration.
Rasayana and Ojas in Charaka Samhita
Reference [16] Charaka Samhita Sutra Sthana
Shloka
“Ojah sarva dhatu saram.”
Translation
Ojas is the essence of all body tissues.
Charaka further describes that preservation of Ojas supports resistance to disease and maintains systemic stability. In contemporary language, Ojas may be viewed as integrated immune and metabolic resilience.
Integrative stabilization therefore aligns conceptually with immune support and reduction of systemic stress that may otherwise weaken biological resistance.
Rasayana Formulations in Bhavaprakasha
Reference [17] Bhavaprakasha Madhyama Khanda Rasayana Prakarana
Shloka
“Yat jara vyadhi nashanam deha dhatu balapradam tat sarvam rasayanam proktam.”
Translation
That which prevents degeneration and strengthens body tissues is called Rasayana.
Bhavaprakasha elaborates formulations designed to nourish Dhatu and promote long term vitality. These formulations were historically used for preventive and strengthening purposes.
Conceptual Integration
Modern oncology identifies hallmark processes such as immune evasion and inflammation [8]. Classical Ayurveda emphasizes Rasayana and Ojas preservation [15], [16], [17]. Though articulated in different scientific languages, both systems recognize the importance of systemic terrain.
Integrative biological stabilization therefore operates at three coordinated levels.
First
Oncology directed tumor management according to established clinical guidelines.
Second
Host environment stabilization through metabolic correction, inflammation control, and stress regulation.
Third
Long term resilience building informed by Rasayana principles.
This model does not claim that Rasayana replaces surgery, chemotherapy, or radiation. It positions Rasayana as a theoretical bridge supporting systemic stability while modern oncology addresses tumor specific pathways.
Global Clinical Relevance
In the United States, United Kingdom, Australia, Singapore, and Canada, oncology care is structured around evidence based protocols. Integrative biological stabilization theory aligns with this framework by reinforcing that supportive care must complement, not compete with, hospital treatment.
Hanahan and Weinberg [8] provide the molecular foundation. Sushruta Samhita [15], Charaka Samhita [16], and Bhavaprakasha [17] provide the classical resilience framework.
Together, they support a coordinated vision in which tumor directed therapy and systemic strengthening coexist within a responsible, supervised model of care.
FAQ
Can integrative care be used during chemotherapy?
Yes. Integrative support can be used during chemotherapy when it is structured as parallel care and does not interfere with oncology protocols. Any supplements, herbs, or intensive therapies must be reviewed for safety and coordinated with your oncology team to avoid treatment interaction.
Is it safe before surgery?
It can be safe before surgery when the focus is on nutrition optimization, metabolic stability, sleep regulation, and stress reduction. Substances that may increase bleeding risk or interfere with anesthesia should only be used if approved by your surgical team.
Do I stop hospital treatment?
No. This model does not replace hospital treatment. Imaging, biopsy, surgery, chemotherapy, radiation, and endocrine therapy continue exactly as prescribed. Integrative care functions alongside oncology supervision.
Are there drug interactions?
Some supplements, herbs, and nutraceuticals can interact with chemotherapy, endocrine therapy, anticoagulants, or anesthesia medications. Full disclosure and medical coordination are essential to prevent interaction risks.
How soon can I start?
Most patients can begin once diagnostic reports and treatment plans are available. Support during the waiting period before surgery or chemotherapy can often start immediately while hospital scheduling proceeds.
Can this reduce recurrence risk?
A structured program may help address modifiable risk factors such as chronic inflammation, metabolic dysfunction, obesity, and physical inactivity. It does not guarantee prevention, but it can support long term biological stability as part of survivorship care.
Is this suitable for BRCA carriers?
It may be used as parallel supportive care, but BRCA carriers require specialist genetic counseling and oncology supervision. Surveillance and preventive decisions remain guided by medical teams, while supportive strategies focus on metabolic and systemic resilience.
Is this evidence informed?
Yes. The model aligns with established oncology care pathways and evidence informed supportive care principles. It is designed to complement hospital treatment rather than replace it.
Understanding your diagnosis category is essential before beginning any structured support program. The following clinical guides explain how different breast conditions are classified and managed within modern oncology systems.
High Risk and Genetic Breast Cancer
If you have a strong family history, BRCA mutation, or hereditary cancer risk, your prevention strategy must be aligned with genetic counseling and oncology supervision. Structured metabolic and hormonal stabilization may complement surveillance plans.
Read more:
https://panaceayur.com/disease-cure/oncology/breast-cancer/high-risk-and-genetics
Benign Conditions and Atypical Hyperplasia
Not all abnormal findings are cancer. Benign breast lumps, fibroadenoma, and atypical hyperplasia carry different levels of risk. Understanding your pathology report helps determine whether prevention, surveillance, or intervention is appropriate.
Read more:
https://panaceayur.com/disease-cure/oncology/breast-cancer/benign-and-atypia
DCIS and Invasive Breast Cancer
Ductal carcinoma in situ and invasive breast cancer require different treatment pathways under oncology supervision. Structured parallel support may be considered during surgery planning, chemotherapy, radiation, or survivorship.
Read more:
https://panaceayur.com/disease-cure/oncology/breast-cancer/dcis-and-invasive-cancer
How Breast Cancer Care Works in Different Countries
Breast cancer care is organized through structured oncology systems across most developed healthcare environments. Although the healthcare models differ between countries, the core treatment pathway remains similar. Diagnosis is followed by staging, multidisciplinary review, and treatment planning that may include surgery, chemotherapy, radiation therapy, or hormonal therapy.
Understanding how these systems operate can help patients navigate care more confidently and make informed decisions during diagnosis and treatment.
Breast Cancer Care in the United States
In the United States, breast cancer care is typically delivered through specialized oncology centers and hospital networks. Multidisciplinary teams often include surgical oncologists, medical oncologists, radiation oncologists, radiologists, and pathologists. Treatment planning frequently follows national oncology guidelines and tumor board discussions.
Major cancer centers such as the MD Anderson Cancer Center, Memorial Sloan Kettering Cancer Center, and Mayo Clinic provide comprehensive cancer programs that include screening, treatment, survivorship care, and genetic counseling.
Breast Cancer Care in the United Kingdom
The United Kingdom provides breast cancer treatment through the National Health Service. Patients are usually referred to specialized breast clinics where multidisciplinary teams evaluate imaging, biopsy results, and pathology reports before recommending treatment.
Institutions such as the Royal Marsden Hospital and NHS Cancer Centers play key roles in delivering coordinated oncology care across the country.
Breast Cancer Care in Australia
Australia operates a mixed public and private healthcare system. Public hospital oncology programs deliver cancer care through multidisciplinary cancer centers, while private hospitals provide additional treatment access for patients with private insurance.
Organizations such as the Peter MacCallum Cancer Centre and Cancer Australia support national cancer treatment strategies and research programs.
Breast Cancer Care in Singapore
Singapore maintains a highly structured oncology system with centralized cancer institutes. Breast cancer patients often receive treatment through specialized cancer centers where multidisciplinary teams coordinate diagnosis and therapy.
The National Cancer Centre Singapore and major academic hospitals provide advanced imaging, surgical oncology, radiation therapy, and survivorship programs.
Breast Cancer Care in Canada
Canada provides cancer treatment through provincial healthcare systems supported by national cancer agencies. Breast cancer patients are usually managed through regional cancer centers where multidisciplinary teams coordinate diagnosis and treatment.
Institutions such as Princess Margaret Cancer Centre and provincial cancer programs deliver integrated oncology services across the country.
Why Patients Often Search for Additional Support
Even within advanced healthcare systems, patients may experience periods of uncertainty during diagnosis, treatment scheduling, or survivorship planning. Waiting periods before surgery, chemotherapy, or imaging appointments can create anxiety and lead patients to seek additional information about lifestyle support, metabolic health, and long-term recovery.
During these phases many patients look for credible guidance that can support overall health while continuing hospital-based oncology care.
The Importance of Coordinated Oncology Care
Regardless of the country, breast cancer management works best when treatment decisions are guided by qualified oncology professionals. Diagnostic imaging, pathology interpretation, surgery, chemotherapy, and radiation therapy remain central components of modern cancer care.
Supportive strategies should always be coordinated carefully with the treating medical team to ensure safety and compatibility with ongoing treatment plans.
Reference
[1] Sung, H., Ferlay, J., Siegel, R. L., et al. (2021). Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide. CA: A Cancer Journal for Clinicians, 71(3), 209–249. https://doi.org/10.3322/caac.21660
This global epidemiology report establishes cancer burden, incidence patterns, and mortality rates worldwide. It supports the need for structured oncology support systems and long-term prevention strategies across countries.
[2] Hanna, T. P., King, W. D., Thibodeau, S., et al. (2020). Mortality due to cancer treatment delay: Systematic review and meta-analysis. BMJ, 371, m4087. https://doi.org/10.1136/bmj.m4087
This meta-analysis shows that delays in cancer surgery and treatment are associated with increased mortality risk. It supports the concept that the waiting phase is clinically significant and not biologically inactive.
[3] Roxburgh, C. S., & McMillan, D. C. (2014). Role of systemic inflammatory response in predicting survival in patients with primary operable cancer. Future Oncology, 10(6), 989–1002. https://doi.org/10.2217/fon.14.16
Demonstrates how chronic systemic inflammation correlates with poorer cancer outcomes, supporting the section on inflammation and biological microenvironment stabilization.
[4] Iyengar, N. M., Gucalp, A., Dannenberg, A. J., & Hudis, C. A. (2016). Obesity and cancer mechanisms: Tumor microenvironment and inflammation. Journal of Clinical Oncology, 34(35), 4270–4276. https://doi.org/10.1200/JCO.2016.67.4283
Explains how metabolic dysfunction and adipose-driven inflammation influence tumor biology. Supports metabolic correction and insulin regulation sections.
[5] Pollak, M. (2008). Insulin and insulin-like growth factor signalling in neoplasia. Nature Reviews Cancer, 8(12), 915–928. https://doi.org/10.1038/nrc2536
Details the role of insulin and IGF pathways in tumor growth. Supports insulin resistance and metabolic stabilization discussion.
[6] Ligibel, J. A., et al. (2014). American Society of Clinical Oncology position statement on obesity and cancer. Journal of Clinical Oncology, 32(31), 3568–3574. https://doi.org/10.1200/JCO.2014.58.4680
Provides formal oncology guidance linking obesity, metabolism, and cancer outcomes. Supports lifestyle and metabolic regulation protocols.
[7] Chlebowski, R. T., et al. (2013). Influence of estrogen plus progestin on breast cancer incidence and mortality. JAMA, 304(15), 1684–1692. https://doi.org/10.1001/jama.2010.1500
Establishes hormonal influence on breast cancer risk and recurrence, supporting hormonal modulation and estrogen metabolism correction.
[8] Hanahan, D., & Weinberg, R. A. (2011). Hallmarks of cancer: The next generation. Cell, 144(5), 646–674. https://doi.org/10.1016/j.cell.2011.02.013
Foundational oncology framework defining tumor microenvironment, immune evasion, inflammation, and metabolic reprogramming. Supports biological rationale section.
[9] Antoni, M. H., et al. (2006). The influence of bio-behavioural factors on tumour biology. Nature Reviews Cancer, 6(3), 240–248. https://doi.org/10.1038/nrc1820
Demonstrates how stress, neuroendocrine pathways, and psychological distress influence tumor biology. Supports stress stabilization section.
[10] Demicheli, R., et al. (2008). Recurrence dynamics in breast cancer. Journal of Clinical Oncology, 26(21), 3511–3517. https://doi.org/10.1200/JCO.2007.14.6628
Shows patterns of recurrence over time, supporting long-term recurrence prevention strategy.
[11] Rock, C. L., et al. (2022). Nutrition and physical activity guidelines for cancer survivors. CA: A Cancer Journal for Clinicians, 72(3), 230–262. https://doi.org/10.3322/caac.21719
Evidence-based lifestyle guidelines for cancer survivors, supporting structured diet and lifestyle program.
[12] Ligibel, J. A., et al. (2018). Physical activity and cancer outcomes: A systematic review. Journal of Clinical Oncology, 36(9), 875–886. https://doi.org/10.1200/JCO.2017.76.6559
Demonstrates exercise benefits in recurrence reduction and survival improvement. Supports metabolic and immune stabilization protocols.
[13] NCCN Clinical Practice Guidelines in Oncology. (Current version). National Comprehensive Cancer Network. https://www.nccn.org
Authoritative oncology treatment guidelines. Supports compliance positioning and parallel-care framework.
[14] ASCO Integrative Oncology Guidelines Panel. (2018). Integrative therapies during and after breast cancer treatment. Journal of Clinical Oncology, 36(25), 2647–2655. https://doi.org/10.1200/JCO.2018.79.2721
Provides oncology-endorsed framework for safe integrative approaches during active treatment. Crucial for compliance section.
[15] Sushruta Samhita, Chikitsa Sthana, Rasayana Adhyaya. Classical Ayurvedic text.
Describes Rasayana therapy principles aimed at tissue rejuvenation, resilience, and systemic stability. Supports integrative biological stabilization model.
[16] Charaka Samhita, Chikitsa Sthana 1 (Rasayana Chapter). Classical Ayurvedic text.
Outlines Rasayana as method for enhancing Ojas, immune resilience, and tissue vitality. Used to bridge Ayurvedic theory with modern immune surveillance concepts.
[17] Bhavaprakasha, Madhyama Khanda, Rasayana Prakarana. Classical Ayurvedic text.
Details rejuvenative formulations aimed at systemic strengthening. Supports preventive framework positioning.