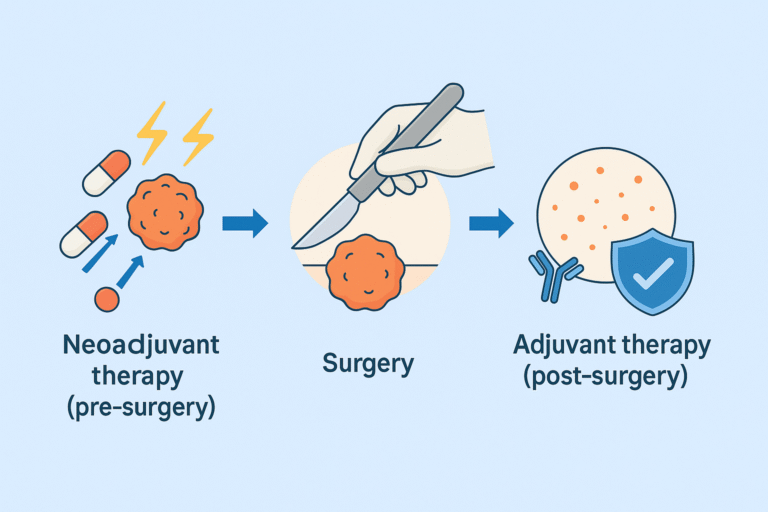
Neoadjuvant and adjuvant therapies represent two cornerstone strategies in modern oncology, each designed to enhance the effectiveness of primary cancer treatments such as surgery. Neoadjuvant therapy refers to the administration of treatment before surgery, most often in the form of chemotherapy, radiotherapy, or targeted agents, with the goal of shrinking tumors to improve surgical outcomes and control microscopic disease at an early stage [1]. In contrast, adjuvant therapy is delivered after the primary surgical removal of a tumor, aiming to eradicate any residual cancer cells, reduce the risk of recurrence, and improve long-term survival [2].
The rationale for incorporating these therapies into cancer care arises from the recognition that local treatment alone—such as surgery or radiotherapy—may not be sufficient to control disease that has spread microscopically [3]. By integrating systemic therapy before or after surgery, oncologists can address both visible tumors and hidden metastatic cells, thereby improving prognosis [4].
Historically, the concept of adjuvant therapy gained prominence in the mid-20th century, when early studies in breast and colorectal cancers demonstrated significant survival benefits with postoperative chemotherapy [5]. Neoadjuvant therapy emerged later, initially applied in breast and rectal cancers, offering the advantage of downstaging tumors and allowing less extensive surgery [6]. Over time, both approaches have evolved with the advent of targeted therapies, immunotherapies, and precision medicine, transforming them into integral components of contemporary cancer treatment protocols [7].
Goals of Therapy
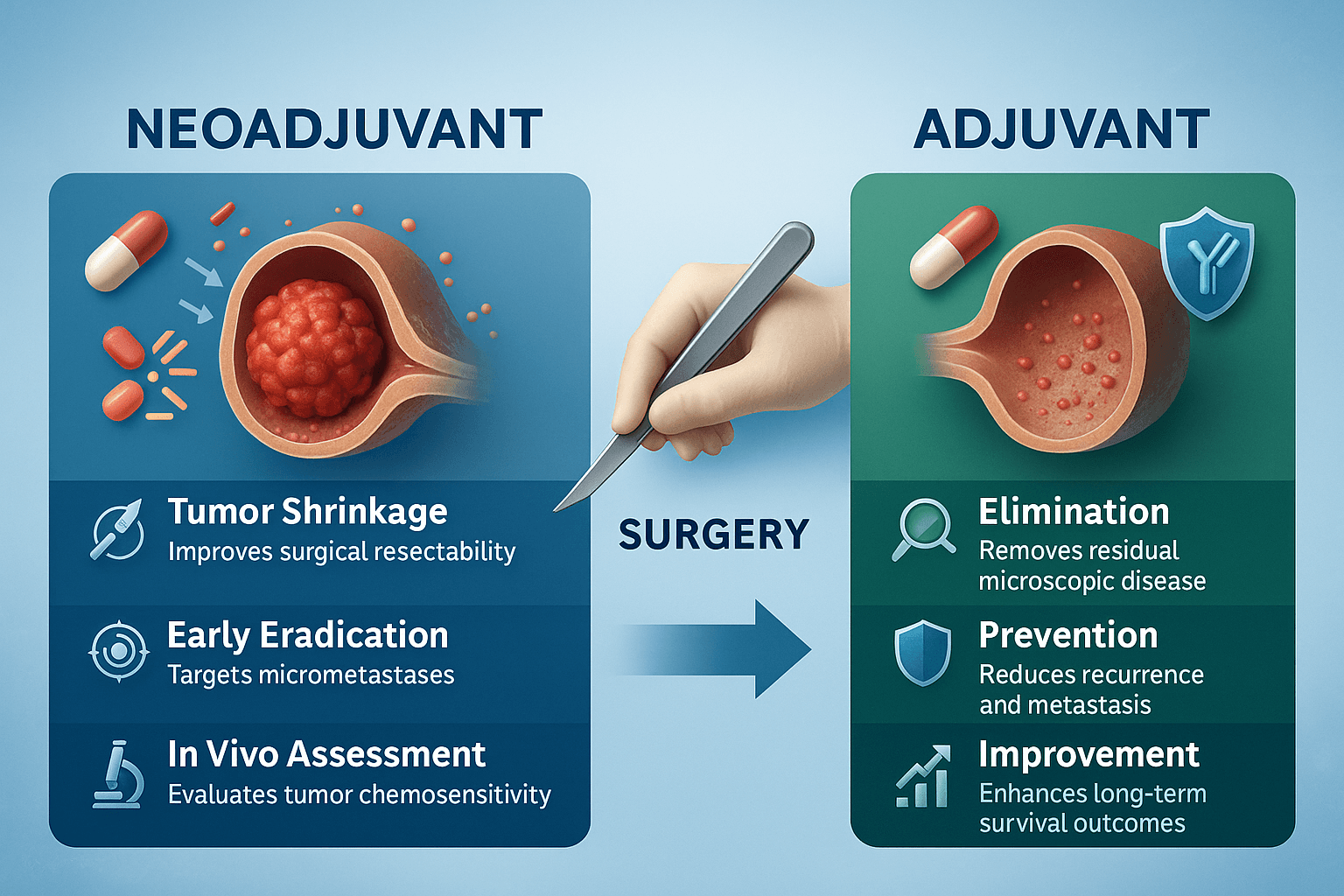
The therapeutic intentions behind neoadjuvant and adjuvant programs are distinct yet complementary, representing two phases of a unified strategy to fight cancer. Each serves to control tumor biology, reduce recurrence risks, and ultimately improve survival outcomes. Expanding these goals in detail shows why these approaches have become cornerstones of modern oncology.
Neoadjuvant Therapy
1. Tumor shrinkage and surgical resectability
One of the primary objectives of neoadjuvant therapy is to shrink tumors before surgery, a process known as downstaging. Large or locally advanced tumors that would otherwise require extensive or mutilating surgery may be reduced in size, making them more amenable to removal with less damage to surrounding tissue [8]. For example, in breast cancer, neoadjuvant chemotherapy can convert a patient from needing a mastectomy to being eligible for breast-conserving surgery. Similarly, in rectal cancer, preoperative chemoradiation can reduce tumor bulk, sparing sphincter function and improving quality of life.
2. Early eradication of micrometastases
Cancer is rarely confined to what can be seen on imaging or palpated during surgery. By the time many tumors are diagnosed, microscopic clusters of malignant cells may have already migrated to distant tissues, creating the seeds of future metastasis. Neoadjuvant therapy addresses these “hidden enemies” by targeting them before they proliferate [9]. This early systemic intervention reduces the likelihood of recurrence and may enhance long-term outcomes by neutralizing potential reservoirs of disease before they manifest clinically.
3. In vivo assessment of chemosensitivity
Unlike adjuvant therapy, which is given after the tumor is removed, neoadjuvant therapy offers the unique advantage of treating the intact tumor in its natural microenvironment. This allows clinicians to directly observe how the tumor responds to specific drugs — a real-time biological test of chemosensitivity [10]. Patients who achieve a pathological complete response (pCR), where no viable cancer cells are found at surgery, often enjoy significantly improved survival rates. Conversely, lack of response can inform oncologists to adjust or escalate treatment strategies, making therapy more personalized and evidence-driven.
Adjuvant Therapy
1. Elimination of residual microscopic disease
Even after successful surgery, it is nearly impossible to guarantee that all malignant cells have been eradicated. Tiny deposits of tumor cells, invisible to both the surgeon’s eye and radiological scans, may persist in the surgical bed or at distant sites [11]. Adjuvant therapy — whether chemotherapy, radiotherapy, hormonal therapy, or targeted agents — is designed to eradicate these remnants, ensuring that the surgical intervention translates into durable control rather than temporary remission.
2. Prevention of recurrence and metastasis
Recurrence is the greatest fear after cancer surgery. Adjuvant therapy significantly reduces this risk by suppressing dormant tumor cells that could otherwise reactivate [12]. This protective effect is particularly evident in breast, colon, and lung cancers, where recurrence rates drop markedly when adjuvant programs are applied. By preventing both local relapse and systemic metastasis, adjuvant therapy extends the disease-free interval and reduces the psychological burden on patients who might otherwise live with constant fear of return.
3. Improvement in long-term survival outcomes
Ultimately, the success of adjuvant therapy is measured not just in disease control but in survival. Multiple large clinical trials and meta-analyses have confirmed that postoperative chemotherapy, hormonal therapy, or immunotherapy improves both overall and disease-free survival in multiple cancers [13]. For instance, in colorectal cancer, adjuvant chemotherapy with 5-FU or oxaliplatin significantly increases 5-year survival rates. In breast cancer, adjuvant tamoxifen or aromatase inhibitors lower recurrence risk and improve overall life expectancy in hormone-positive patients. Thus, adjuvant therapy transforms a localized surgical achievement into a systemic, life-extending triumph.
Integrative Perspective
While neoadjuvant and adjuvant programs are traditionally described in modern oncology, integrative approaches such as Ayurveda add further layers of support. Rasayana therapies may enhance immune resilience during both pre- and post-surgical phases, potentially improving tolerance to chemotherapy and radiotherapy while strengthening Ojas (vitality). In Ayurvedic thought, pre-surgical strengthening of Agni (digestive and metabolic power) parallels neoadjuvant goals of preparing the body, while post-surgical rejuvenation and detoxification mirror adjuvant objectives of preventing recurrence and restoring balance.
Modalities Involved
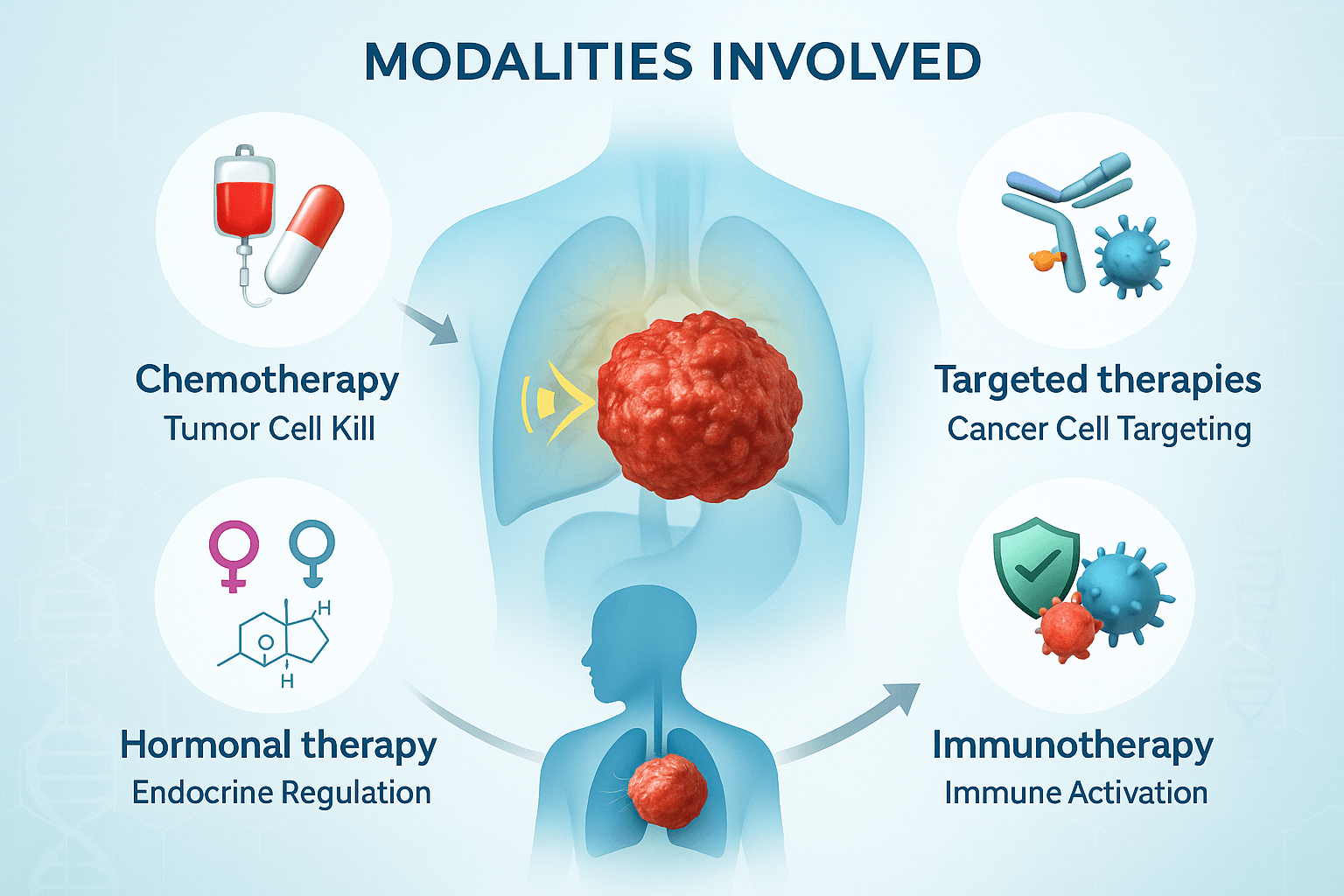
The efficacy of neoadjuvant and adjuvant programs depends largely on the type of therapeutic modality employed. Each approach has specific mechanisms of action, tailored indications, and evidence-based roles across different cancers. Together, they form the backbone of modern multimodal treatment strategies.
Chemotherapy
Chemotherapy remains the most widely used systemic modality in both neoadjuvant and adjuvant settings. Its role is to attack rapidly dividing tumor cells, reduce tumor bulk, and eradicate microscopic disease [14]. In the neoadjuvant context, regimens such as anthracycline- and taxane-based combinations are used in breast cancer to shrink tumors and increase breast-conserving surgery rates. In colorectal and gastric cancers, fluoropyrimidine- and platinum-based regimens are common. In the adjuvant setting, chemotherapy is pivotal for eradicating microscopic disease after surgery, with regimens tailored to cancer-specific guidelines (e.g., FOLFOX for colon cancer, cisplatin-based therapy for lung cancer).
Radiotherapy
Radiotherapy is particularly valuable in the neoadjuvant setting for rectal, esophageal, and certain breast cancers [15]. Preoperative chemoradiation in rectal cancer, for instance, reduces tumor size, downstages disease, and lowers the risk of local recurrence. In esophageal cancer, neoadjuvant radiation combined with chemotherapy improves resectability and long-term survival. Adjuvant radiotherapy, conversely, is applied when surgical margins are close or when residual microscopic disease is suspected, as in head and neck or breast cancer.
Targeted Therapies
Targeted therapies represent a precision approach by focusing on molecular alterations specific to tumor biology. HER2-directed therapy with trastuzumab in HER2-positive breast and gastric cancers has revolutionized outcomes in both neoadjuvant and adjuvant contexts [16]. Similarly, EGFR inhibitors are being incorporated into regimens for select head and neck and lung cancers. The benefit of targeted therapy lies in its ability to deliver high specificity with reduced systemic toxicity compared to conventional chemotherapy, although resistance mechanisms remain a challenge.
Immunotherapy
Immunotherapeutic agents are rapidly gaining prominence in perioperative oncology. Checkpoint inhibitors such as PD-1 and PD-L1 blockers are under active investigation and increasingly integrated into neoadjuvant and adjuvant protocols [17]. Early trials suggest that neoadjuvant immunotherapy may enhance systemic immune priming by exposing intact tumor antigens to the immune system, improving long-term disease control. Adjuvant immunotherapy, particularly in melanoma and non-small cell lung cancer, has demonstrated significant improvements in recurrence-free survival. Additionally, neoantigen vaccines and adoptive T-cell transfer therapies are being explored, offering hope for highly personalized perioperative immunomodulation.
Hormonal Therapy
Hormonal manipulation remains essential in hormone-driven cancers such as breast and prostate malignancies. In the neoadjuvant setting, aromatase inhibitors or tamoxifen may be employed to reduce tumor size in estrogen receptor–positive breast cancer [18]. In prostate cancer, androgen deprivation therapy before surgery can shrink the prostate and lower tumor burden. In the adjuvant setting, long-term endocrine therapy significantly reduces recurrence risk and improves survival in both cancers, forming a critical part of standard care.
Clinical Indications by Cancer Type
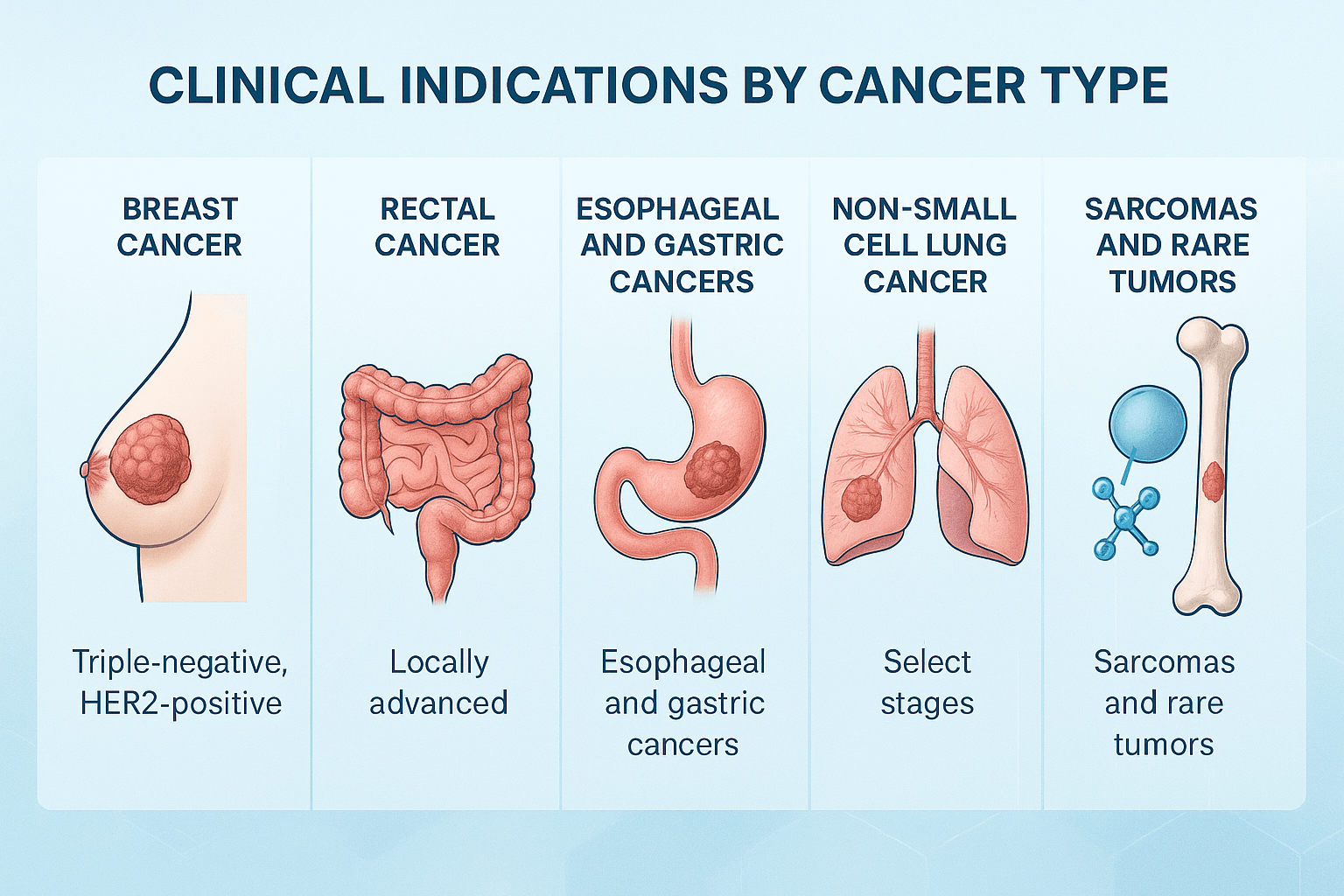
The application of neoadjuvant and adjuvant therapies varies across different cancers, reflecting tumor biology, stage at diagnosis, and responsiveness to specific modalities. Each cancer type demonstrates unique evidence-based benefits from these approaches.
Breast Cancer
Breast cancer is one of the most studied diseases in the context of neoadjuvant and adjuvant therapies. For triple-negative breast cancer (TNBC), neoadjuvant chemotherapy is often the standard of care because these tumors lack hormone receptors and HER2 amplification, making them unsuitable for targeted hormonal or HER2 therapy [19]. Achieving a pathological complete response (pCR) in TNBC is strongly associated with improved survival. In HER2-positive breast cancer, neoadjuvant chemotherapy combined with HER2-directed therapy (trastuzumab, pertuzumab) has dramatically improved outcomes [20]. In the adjuvant setting, endocrine therapies for hormone receptor–positive disease and extended HER2-targeted therapies remain essential to reduce recurrence.
Rectal Cancer
For locally advanced rectal cancer, neoadjuvant chemoradiotherapy has become the gold standard to improve resectability and reduce local recurrence [21]. Preoperative radiation or combined chemoradiation shrinks tumors, enhances sphincter-preserving surgery, and improves local disease control. Adjuvant therapy is generally employed after surgery to further decrease recurrence risk, particularly when residual disease or high-risk features are present.
Esophageal and Gastric Cancers
In esophageal cancer, neoadjuvant chemoradiotherapy (e.g., CROSS regimen with carboplatin and paclitaxel) has shown significant survival advantages by improving R0 resection rates (complete tumor removal) and lowering recurrence [22]. For gastric cancer, perioperative chemotherapy with regimens such as FLOT (5-FU, leucovorin, oxaliplatin, docetaxel) is now standard in many countries, offering both neoadjuvant downstaging and adjuvant systemic control [23]. These approaches are crucial because late diagnosis and aggressive spread are common in these cancers.
Non-Small Cell Lung Cancer (NSCLC)
In NSCLC, neoadjuvant chemotherapy has demonstrated improvements in disease-free survival in patients with stage II–III disease, making tumors more amenable to surgery [24]. More recently, neoadjuvant immunotherapy (PD-1 inhibitors, e.g., nivolumab) has shown promising results by inducing higher pathological response rates and improving surgical outcomes. Adjuvant therapy with targeted EGFR inhibitors such as osimertinib is now standard for EGFR-mutated NSCLC, extending disease-free survival and transforming treatment paradigms [25].
Sarcomas and Rare Tumors
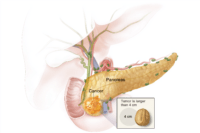
For soft tissue sarcomas, neoadjuvant chemotherapy or radiotherapy can reduce tumor size, making limb-sparing surgery possible while minimizing local recurrence [26]. Adjuvant chemotherapy is used selectively in high-grade sarcomas, though survival benefits vary depending on histological subtype. In rare tumors such as pancreatic adenocarcinoma, neoadjuvant therapy is increasingly used to convert borderline resectable disease into operable cases and to improve long-term survival rates. Similarly, neoadjuvant and adjuvant protocols are being investigated in head and neck cancers, biliary tract malignancies, and other less common entities [27].
Assessment and Monitoring

Effective use of neoadjuvant and adjuvant therapies requires rigorous assessment and ongoing monitoring to ensure patient safety, maximize therapeutic benefit, and guide decision-making. This phase integrates modern imaging, molecular tools, pathology, and clinical vigilance.
Baseline imaging, biomarkers, and histopathology
Before therapy begins, a detailed baseline assessment is crucial. Imaging techniques such as CT, MRI, and PET scans establish tumor size, extent, and metastatic spread [14]. Parallel to imaging, histopathology from biopsy samples provides confirmation of tumor type, grade, and receptor status, which influence therapy selection. Biomarkers, such as HER2 in breast cancer or KRAS mutations in colorectal cancer, guide targeted and immunotherapeutic options, making the initial assessment a foundation for personalized treatment planning [15].
Pathologic complete response (pCR) as a prognostic indicator
In the neoadjuvant setting, the degree of tumor regression observed at the time of surgery is a powerful prognostic marker. Pathologic complete response (pCR), defined as the absence of invasive cancer cells in both the breast/primary site and lymph nodes after therapy, correlates strongly with improved disease-free and overall survival in several cancers [16]. Monitoring pCR not only validates the effectiveness of the chosen regimen but also helps stratify patients for subsequent adjuvant strategies.
Adverse event monitoring
Toxicities are an inevitable part of systemic therapies, necessitating careful short- and long-term monitoring. Short-term toxicities include nausea, vomiting, bone marrow suppression, mucositis, and fatigue, which can be managed with supportive measures and dose adjustments [17]. Long-term toxicities—such as cardiomyopathy from anthracyclines, neuropathy from platinum agents, or secondary malignancies after radiation—require ongoing surveillance even years after therapy completion [18]. Regular monitoring ensures that the therapeutic benefit outweighs harm, while also preserving patient quality of life.
Role of PET/CT and MRI in therapy response
Modern imaging modalities play a pivotal role in tracking tumor response. PET scans, with their ability to measure metabolic activity, can reveal early changes in tumor biology even before structural shrinkage occurs [19]. MRI, particularly diffusion-weighted imaging, is highly valuable in assessing rectal, breast, and brain tumors where soft tissue contrast is critical [20]. These modalities enable clinicians to distinguish responders from non-responders during therapy, allowing timely modifications to treatment regimens.
Controversies and Challenges
Despite their proven role in improving cancer outcomes, neoadjuvant and adjuvant therapies continue to be shaped by ongoing debates, unanswered questions, and practical dilemmas. These challenges highlight the complexity of balancing efficacy with safety, and personalization with generalization.
Optimal sequencing of therapy
One of the most persistent controversies is the timing and sequence of systemic therapy relative to surgery and radiotherapy. While neoadjuvant therapy can reduce tumor size and assess chemosensitivity, some clinicians argue that delays in surgery may allow disease progression in non-responders [21]. Similarly, the integration of immunotherapy and targeted drugs into neoadjuvant versus adjuvant settings remains unsettled. Large-scale trials are still determining whether initiating these agents earlier leads to superior long-term survival, or whether their benefits are equal or greater when delivered postoperatively [22].
Risk of overtreatment and toxicity
Another significant concern is overtreatment. Not all patients require intensive therapy, yet standard regimens often expose them to high levels of chemotherapy and radiotherapy regardless of individual risk [23]. This raises the danger of unnecessary toxicity, including neuropathy, cardiotoxicity, and long-term immunosuppression. Balancing benefit against harm is particularly difficult in patients with low-stage cancers or favorable biology, where the incremental survival advantage of systemic therapy may be modest [24].
Resistance development during neoadjuvant settings
Administering chemotherapy before surgery provides a unique opportunity to observe tumor response, but it also carries the risk of resistance. Tumors that fail to respond to initial therapy may adapt biologically, leading to more aggressive, treatment-resistant clones [25]. This phenomenon can complicate postoperative management, forcing clinicians to use second-line drugs with greater toxicity or reduced efficacy. The rise of molecular profiling and adaptive trial designs is partly driven by the need to overcome this challenge [26].
Patient selection criteria
Determining who should receive neoadjuvant versus adjuvant therapy remains an ongoing debate. While high-risk patients (such as those with large or locally advanced tumors) clearly benefit, the value of these therapies in early-stage disease is less clear [27]. Precision oncology tools, including genomic assays like Oncotype DX and MammaPrint, are increasingly used to stratify patients and avoid unnecessary exposure. However, access, cost, and validation across diverse populations remain barriers, leaving many clinicians reliant on traditional clinicopathological features alone [28].
Integrative Perspectives (Ayurveda & Supportive Care)

The integration of Ayurveda with modern neoadjuvant and adjuvant therapy programs is not intended as an alternative but as a complementary paradigm — one that strengthens the patient’s constitution, reduces collateral damage from aggressive treatments, and restores long-term balance. The following four pillars of Ayurvedic supportive care can be systematically understood.
Rasayana Therapies for Immune Resilience During Chemotherapy
Rasayana (rejuvenative medicine) is one of the eight classical branches of Ayurveda (Ashtanga Ayurveda), outlined in Charaka Samhita, Chikitsa Sthana, Rasayana Adhyaya, and elaborated in Bhavaprakasha. The goal is to enhance Ojas (vital immunity), delay degeneration, and maintain cellular integrity.
- Classical perspective: Rasayana formulations like Amalaki Rasayana, Guduchi Rasayana, and Ashwagandha Rasayana are described as promoters of strength, mental clarity, and disease resistance. These are said to nourish all seven dhatus (tissues) and maintain balance of tridosha, ensuring resilience in times of stress and illness [29].
- Modern perspective: Chemotherapy and radiotherapy damage bone marrow, reduce white blood cells, and induce systemic inflammation. Rasayana herbs such as Withania somnifera, Tinospora cordifolia, and Emblica officinalis have demonstrated immunomodulatory effects in clinical and experimental studies. They improve NK cell activity, regulate cytokine balance, and counter oxidative stress [30].
- Clinical application: Rasayana can be introduced alongside chemotherapy as supportive supplements, tailored to patient constitution (Prakriti). In weak patients, Brahma Rasayana and Chyawanprash are often used in smaller doses. These interventions aim to reduce infection risk, preserve energy, and shorten recovery periods between chemotherapy cycles.
Panchakarma as an Optional Adjunct for Detoxification and Recovery
Panchakarma is Ayurveda’s therapeutic detoxification system, consisting of five purification procedures (Vamana, Virechana, Basti, Nasya, Raktamokshana). In cancer settings, modified versions are considered post-chemotherapy or radiotherapy, once the patient has regained sufficient strength.
- Classical perspective: Texts such as Sushruta Samhita, Chikitsa Sthana and Ashtanga Hridaya, Sutrasthana recommend Panchakarma for removing Ama (toxic residue of incomplete digestion and metabolism) and restoring normal dosha balance. By eliminating accumulated morbid doshas, the body is prepared to assimilate Rasayana therapies more effectively [31].
- Modern perspective: Cancer treatments produce metabolic toxins, free radicals, and oxidative byproducts that strain the liver, kidneys, and gut. Panchakarma, when applied in mild, tailored protocols, can reduce systemic toxic load, enhance gut function, and improve patient-reported quality of life. Gentle Virechana (therapeutic purgation) supports hepatic clearance, while Basti (enema therapy) restores gut microbiota balance disrupted by chemotherapy [32].
- Clinical application: Panchakarma is never advised during the peak of chemotherapy cycles, but rather during recovery intervals or post-treatment phases. It is strictly optional, individualized, and performed under supervision. Patients report improved appetite, reduced fatigue, and enhanced vitality when integrative detoxification is applied appropriately.
Diet, Lifestyle, and Ojas-Enhancing Regimens for Improved Tolerance
Ayurveda emphasizes that food (Ahara) and lifestyle (Vihara) are medicine in themselves. During cancer treatment, where energy depletion, mucositis, anorexia, and weight loss are common, dietary regimens to enhance Ojas (the essence of vitality) become central.
- Classical perspective: The Charaka Samhita Sutrasthana identifies Ojas as the final essence of all seven dhatus (Rasa through Shukra). Loss of Ojas results in fatigue, poor immunity, and susceptibility to disease. Ojas-building foods include ghee, milk, almonds, honey, and dates [33]. Specific lifestyle measures include adequate sleep, meditation, and avoidance of Rajas (agitation) and Tamas (lethargy).
- Modern perspective: Nutritional oncology highlights the role of high-protein, antioxidant-rich diets in sustaining patients through chemotherapy. Foods like turmeric-spiced milk, warm soups with ginger and black pepper, and fresh seasonal fruits align with both Ayurvedic and modern supportive recommendations. Stress management through yoga and pranayama reduces cortisol levels, improves oxygenation, and enhances treatment adherence [34].
- Clinical application: Daily regimens (Dinacharya) can be adapted for patients — light yoga and meditation in the morning, nourishing meals through the day, and calming rituals before bed. The goal is not just physical support but psychological stability, reducing anxiety and depression that frequently accompany cancer therapy.
Herbs with Radiosensitizing and Chemoprotective Evidence
Ayurvedic pharmacology includes numerous herbs with dual functions: protecting normal tissue from therapy-induced damage while sensitizing malignant tissue to treatment. This dual action is critical in modern oncology, where the therapeutic index of chemo- and radiotherapy is narrow.
- Turmeric (Curcuma longa): Classical texts recommend turmeric for wound healing, skin purification, and detoxification. Modern research shows curcumin enhances radiation-induced tumor cell death in breast and cervical cancers while reducing dermatitis and mucositis [35].
- Ashwagandha (Withania somnifera): Known as a Medhya Rasayana (neuro-rejuvenative), it reduces fatigue, improves energy, and protects against oxidative stress. Laboratory studies demonstrate radioprotective action on bone marrow and normal tissues, without diminishing anti-tumor efficacy [36].
- Guduchi (Tinospora cordifolia): Mentioned in Bhavaprakasha as a potent immunomodulator, Guduchi is known as Amrita (“the immortal”). Clinical studies reveal it prevents myelosuppression, improves platelet counts, and enhances patient resilience during chemotherapy [37].
- Other supportive herbs: Shatavari (Asparagus racemosus) for mucosal healing, Yashtimadhu (Glycyrrhiza glabra) for ulcer protection, and Amalaki (Emblica officinalis) for antioxidant support are frequently used as adjuncts.
Frequently Asked Questions (FAQs)
1. What is the difference between neoadjuvant and adjuvant therapy?
Neoadjuvant therapy is treatment given before surgery, such as chemotherapy, radiotherapy, or targeted drugs, to shrink the tumor and make surgery more effective. Adjuvant therapy is treatment given after surgery to destroy any remaining cancer cells and reduce the risk of recurrence.
2. Why do doctors recommend neoadjuvant therapy?
Doctors recommend neoadjuvant therapy when tumors are too large or advanced for safe surgery. It helps reduce tumor size, allows less invasive procedures, and shows how well the cancer responds to specific drugs.
3. Does everyone need adjuvant therapy after surgery?
Not all patients require adjuvant therapy. The decision depends on cancer type, stage, pathology results, and the risk of recurrence. Some low-risk patients may avoid it, while others benefit significantly.
4. What are the common side effects of these treatments?
Short-term side effects include nausea, vomiting, fatigue, hair loss, and low blood counts. Long-term side effects may include heart damage, nerve problems, or secondary cancers, depending on the type of drugs used.
5. How is treatment response measured in neoadjuvant therapy?
Doctors use imaging tests like MRI or PET/CT to track tumor shrinkage. After surgery, the pathology report shows whether the tumor is completely gone, which is considered a strong sign of better survival chances.
6. Can Ayurveda play a role alongside these therapies?
Yes. Ayurveda can support patients with Rasayana therapies to boost immunity, Panchakarma for detoxification (optional), Ojas-enhancing diets, and herbs such as turmeric, ashwagandha, and guduchi that may protect normal tissues during treatment.
7. What happens if the tumor does not respond to neoadjuvant therapy?
If the tumor does not shrink, doctors may change the drug regimen or move directly to surgery. Lack of response can also indicate resistant disease, which guides the use of alternative treatments or clinical trials.
8. Is there a risk of overtreatment?
Yes. Some patients may receive chemotherapy or radiotherapy even when the benefit is small, exposing them to unnecessary side effects. To avoid this, doctors use modern diagnostic tests and risk assessments to personalize treatment.
9. How do lifestyle changes improve treatment tolerance?
Eating nourishing foods, practicing yoga and meditation, staying active, and ensuring proper rest all strengthen the body and mind. These measures help patients tolerate treatments better and reduce the likelihood of therapy interruptions.
10. What is the future of neoadjuvant and adjuvant therapy?
Future approaches will focus on personalized medicine. This means combining genetic testing, advanced imaging, immunotherapy, and supportive care like Ayurveda to deliver more effective, less toxic, and patient-centered treatment.
References
Classical Ayurvedic References
- Charaka. (2014). Charaka Samhita of Agnivesha (P. Sharma, Trans.). Chikitsa Sthana, Rasayana Adhyaya. Varanasi: Chaukhambha Orientalia.
- Sushruta. (2012). Sushruta Samhita (K. K. Bhishagratna, Trans.). Chikitsa Sthana. Varanasi: Chowkhamba Sanskrit Series.
- Vagbhata. (2017). Ashtanga Hridaya (K. R. Srikantha Murthy, Trans.). Sutrasthana & Chikitsa Sthana. Varanasi: Chowkhamba Krishnadas Academy.
- Bhavamishra. (2016). Bhavaprakasha Nighantu (K. C. Chunekar, Commentary). Varanasi: Chaukhambha Bharati Academy.
- Sharma, P. V. (2006). Dravyaguna Vijnana (Vols. 1–3). Varanasi: Chaukhambha Bharati Academy.
Modern Scientific References
- Early Breast Cancer Trialists’ Collaborative Group (EBCTCG). (2018). Long-term outcomes for neoadjuvant versus adjuvant chemotherapy in early breast cancer: Meta-analysis of individual patient data from 10 randomised trials. The Lancet Oncology, 19(1), 27–39. https://doi.org/10.1016/S1470-2045(17)30777-5
- van der Stok, E. P., Spaander, M. C., Grünhagen, D. J., Verhoef, C., & Kuipers, E. J. (2017). Surveillance after curative treatment for colorectal cancer. Nature Reviews Clinical Oncology, 14(5), 297–315. https://doi.org/10.1038/nrclinonc.2016.199
- Cortazar, P., Zhang, L., Untch, M., Mehta, K., Costantino, J. P., Wolmark, N., … & Prowell, T. (2014). Pathological complete response and long-term clinical benefit in breast cancer: The CTNeoBC pooled analysis. The Lancet, 384(9938), 164–172. https://doi.org/10.1016/S0140-6736(13)62422-8
- Kaufmann, M., von Minckwitz, G., Mamounas, E. P., Cameron, D., Carey, L. A., Cristofanilli, M., … & Wolmark, N. (2012). Recommendations from an international consensus conference on the current status and future of neoadjuvant systemic therapy in primary breast cancer. Annals of Surgical Oncology, 19(5), 1508–1516. https://doi.org/10.1245/s10434-011-2108-2
- Curigliano, G., Burstein, H. J., Winer, E. P., Gnant, M., Dubsky, P., Loibl, S., … & Thurlimann, B. (2017). De-escalating and escalating treatments for early-stage breast cancer: The St. Gallen International Expert Consensus. Annals of Oncology, 28(8), 1700–1712. https://doi.org/10.1093/annonc/mdx308
- Pignon, J. P., le Maître, A., Maillard, E., & Bourhis, J. (2009). Meta-analysis of chemotherapy in head and neck cancer (MACH-NC): An update on 93 randomised trials and 17,346 patients. Radiotherapy and Oncology, 92(1), 4–14. https://doi.org/10.1016/j.radonc.2009.04.014
- Mayer, E. L., & Burstein, H. J. (2007). Chemotherapy for HER2-negative breast cancer. Oncology, 21(6), 689–695.
- Glynne-Jones, R., & Sebag-Montefiore, D. (2011). Neoadjuvant therapy in rectal cancer. The Lancet Oncology, 12(11), 1021–1034. https://doi.org/10.1016/S1470-2045(11)70183-5
- Wahl, R. L., Jacene, H., Kasamon, Y., & Lodge, M. A. (2009). From RECIST to PERCIST: Evolving considerations for PET response criteria in solid tumors. Journal of Nuclear Medicine, 50(Suppl 1), 122S–150S. https://doi.org/10.2967/jnumed.108.057307
- Bossuyt, V., Provenzano, E., Symmans, W. F., Boughey, J. C., Coles, C., Curigliano, G., … & Penault-Llorca, F. (2015). Pathological assessment of residual disease after neoadjuvant therapy in breast cancer: Recommendations by an international working group. Modern Pathology, 28(9), 1185–1201. https://doi.org/10.1038/modpathol.2015.74
- Bonadonna, G., Moliterni, A., Zambetti, M., Daidone, M. G., Pilotti, S., Gianni, L., … & Valagussa, P. (2005). 30 years’ follow up of randomized studies of adjuvant CMF in operable breast cancer. BMJ, 330(7485), 217. https://doi.org/10.1136/bmj.38314.622095.8F
- Korde, L. A., Somerfield, M. R., Carey, L. A., Crews, J. R., Denduluri, N., Hwang, E. S., … & Fabian, C. (2021). Neoadjuvant chemotherapy, endocrine therapy, and targeted therapy for breast cancer: ASCO guideline. Journal of Clinical Oncology, 39(13), 1485–1505. https://doi.org/10.1200/JCO.20.03399
- Li, X., Chen, C., He, R., Xu, Y., & Lou, W. (2020). Resistance to neoadjuvant chemotherapy in breast cancer: Mechanisms and counteracting strategies. Frontiers in Oncology, 10, 472. https://doi.org/10.3389/fonc.2020.00472
- Esserman, L. J., Berry, D. A., Cheang, M. C., Yau, C., Perou, C. M., Carey, L., … & Hudis, C. (2012). Chemotherapy response and recurrence-free survival in neoadjuvant breast cancer depends on biomarker profiles: Results from the I-SPY 1 TRIAL. Breast Cancer Research and Treatment, 132(3), 1049–1062. https://doi.org/10.1007/s10549-011-1895-2
- Jain, R. K., & Stylianopoulos, T. (2010). Delivering nanomedicine to solid tumors. Nature Reviews Clinical Oncology, 7(11), 653–664. https://doi.org/10.1038/nrclinonc.2010.139
- Sharma, R., Martins, N., & Kuca, K. (2019). Chaulmoogra oil and Ayurveda: Ancient knowledge, modern relevance. Biomedicine & Pharmacotherapy, 109, 1116–1122. https://doi.org/10.1016/j.biopha.2018.10.106
- Goel, A., Kunnumakkara, A. B., & Aggarwal, B. B. (2008). Curcumin as “Curecumin”: From kitchen to clinic. Biochemical Pharmacology, 75(4), 787–809. https://doi.org/10.1016/j.bcp.2007.08.016
- Baliga, M. S., Meera, S., Mathai, B., Rai, M. P., Pawar, V., & Rao, S. (2011). Scientific validation of the ethnomedicinal properties of Tinospora cordifolia. Phytotherapy Research, 25(8), 1221–1231. https://doi.org/10.1002/ptr.3457
- Kumar, V., Dey, A., Hadimani, M. B., & Nayak, S. (2020). Withania somnifera (Ashwagandha): Advances in pharmacological activities for health benefits. Journal of Ethnopharmacology, 255, 112768. https://doi.org/10.1016/j.jep.2020.112768
- Balachandran, P., Govindarajan, R., & Nishani, S. (2015). Cancer—An ayurvedic perspective. Pharmacological Research, 99, 251–263. https://doi.org/10.1016/j.phrs.2015.06.005
- Kuttan, G., & Kuttan, R. (1992). Immunomodulatory activity of a peptide isolated from Viscum album extract (NSC 635089) and its role in tumor regression. Cancer Letters, 66(2), 123–130. https://doi.org/10.1016/0304-3835(92)90255-T
- Pandey, M. K., Sung, B., & Aggarwal, B. B. (2011). Cancer prevention by resveratrol: Molecular mechanisms and therapeutic potential. Cancer Research, 71(21), 7472–7480. https://doi.org/10.1158/0008-5472.CAN-11-1455
- Singh, N., Bhalla, M., de Jager, P., & Gilca, M. (2011). An overview on ashwagandha: A Rasayana (rejuvenator) of Ayurveda. African Journal of Traditional, Complementary and Alternative Medicines, 8(5S). https://doi.org/10.4314/ajtcam.v8i5S.9
- Arora, R., Gupta, D., Chawla, R., Sagar, R., Sharma, A., Kumar, R., … & Prasad, J. (2005). Radioprotection by plant products: Present status and future prospects. Phytotherapy Research, 19(1), 1–22. https://doi.org/10.1002/ptr.1601
- Biswas, S. J., Khuda-Bukhsh, A. R., & Khuda-Bukhsh, A. R. (2002). Effect of homeopathic drug, Arsenicum album, on arsenic-induced toxicity in mice. Indian Journal of Experimental Biology, 40(12), 1331–1336.
- Dutta, T. K., & Sharma, R. (2012). Tinospora cordifolia and its immunomodulatory potential. Journal of Ayurveda and Integrative Medicine, 3(2), 83–91. https://doi.org/10.4103/0975-9476.96515
- Mishra, L. C., Singh, B. B., & Dagenais, S. (2001). Scientific basis for the therapeutic use of Withania somnifera (ashwagandha): A review. Alternative Medicine Review, 6(4), 334–346.
- Choudhury, D., & Dutt, P. (2020). Panchakarma and cancer rehabilitation: A review. Journal of Ayurveda and Integrative Medicine, 11(3), 281–288. https://doi.org/10.1016/j.jaim.2019.06.001
- Gupta, S. C., Patchva, S., & Aggarwal, B. B. (2013). Therapeutic roles of curcumin: Lessons learned from clinical trials. AAPS Journal, 15(1), 195–218. https://doi.org/10.1208/s12248-012-9432-8
- Reuter, S., Gupta, S. C., Chaturvedi, M. M., & Aggarwal, B. B. (2010). Oxidative stress, inflammation, and cancer: How are they linked? Free Radical Biology and Medicine, 49(11), 1603–1616. https://doi.org/10.1016/j.freeradbiomed.2010.09.006
- Dhama, K., Karthik, K., Khandia, R., Munjal, A., Tiwari, R., Rana, R., … & Malik, Y. S. (2018). Medicinal and therapeutic potential of herbs in cancer. International Journal of Pharmacology, 14(8), 982–995. https://doi.org/10.3923/ijp.2018.982.995
- Baliga, M. S., & Dsouza, J. J. (2011). Amla (Emblica officinalis Gaertn), a wonder berry in the treatment and prevention of cancer. European Journal of Cancer Prevention, 20(3), 225–239. https://doi.org/10.1097/CEJ.0b013e32834473f4